Abstract
Purpose
The incidence of head and neck squamous cell carcinomas (HNSCC) associated with papillomavirus (HPV) infection has increased over the past decades in the US. We aimed at examining the global impact of HPV-associated HNSCC, and whether the established key role of mTOR activation in HNSCC is also observed in HPV+ HNSCC lesions, thereby providing novel treatment options for HPV-associated HNSCC patients.
Experimental Design
An international HNSCC tissue microarray (TMA) was used to analyze the expression of p16INK4A, a surrogate for HPV infection, and Akt-mTOR pathway activation. Results were confirmed in a large collection of HPV− and HPV+ HNSCC cases and in a cervical cancer (CCSCC) TMA. Observations were validated in HNSCC and CCSCC-derived cell lines, which were xenografted into immunodeficient mice for tumorigenesis assays.
Results
Approximately 20% of all HNSCC lesions could be classified as HPV+, irrespective of their country of origin. mTOR pathway activation was observed in most HPV+ HNSCC and CCSCC lesions and cell lines. The pre-clinical efficacy of mTOR inhibition by rapamycin and RAD001 was explored in HPV+ HNSCC and CCSCC tumor xenografts. Both mTOR inhibitors effectively decreased mTOR activity in vivo, and caused a remarkable decrease in tumor burden. These results emphasize the emerging global impact of HPV-related HNSCCs, and indicate that the activation of the mTOR pathway is a widespread event in both HPV− and HPV-associated HNSCC and CCSCC lesions.
Conclusions
The emerging results may provide a rationale for the clinical evaluation of mTOR inhibitors as a molecular targeted approach for the treatment of HPV-associated malignancies.
Keywords: Oral cancer, cervical cancer Human papilloma virus, rapalogs, targeted therapy, mTOR
Introduction
The infection with human papillomavirus (HPV) is associated with benign and malignant tumors of the squamous epithelia (1). HPV types have been classified into low- or high-risk groups depending on their likelihood of inducing cervical cancer on persistent infection (1). Indeed, HPV is associated with nearly all cervical cancers; 70% of which are related to the high-risk HPV types 16 and 18 (1). HPV is also associated with other human malignancies, and is now considered to represent the viral etiological agent responsible for about 500,000 new annual human cancer cases worldwide, including those of the cervix, anus, vulva, penis and of the oral cavity and pharynx (1, 2). In particular for oral and pharyngeal cancers, these cancers, which are collectively referred to as head and neck squamous cell carcinomas (HNSCC) (3), have been traditionally associated with tobacco and alcohol consumption in the Western world, with the added risk factor of areca nut in Asia (4). A role for papillomavirus in oral carcinogenesis has been recognized since the mid 80’s (5, 6), but the perception of an emerging epidemic of HPV-associated HNSCC is relatively recent (7, 8), with a highly significant increase in the incidence of oropharyngeal tumors, the most likely to be HPV-associated, predominantly in younger patients (8). HPV-positive (HPV+) and HPV-negative (HPV−) oropharyngeal squamous-cell carcinomas might be inherently different, and hence have different causes, risk-factor profiles, and survival outcomes (9, 10). Indeed, there is a strong correlation between positive tumor HPV status and increased patient survival after surgical treatment (11), radiotherapy (12, 13), and combined treatment approaches (14–16). However, the overall global impact of HPV-associated HNSCC cases and the nature of the shared and distinct molecular mechanisms underlying the progression of HPV+ and HPV− HNSCC lesions are still unclear.
In this regard, the NIDCR-NCI spearheaded an international collaborative effort that resulted in the construction of a tissue microarray with hundreds of samples of HNSCC from patients from 8 different countries from Asia, Africa, and America (17). This array provided an opportunity to examine the distribution of possible HPV-associated cases in each participating country, and thus afforded the first glimpse at the possible impact of HPV-associated HNSCC worldwide. As a general screening approach, we used in this array the expression of protein product of the p16INK4A tumor suppressor gene, referred herein as p16, as a validated surrogate marker for HPV infections (18–20). Nearly 20% of all cases were positive, with a limited country-specific distribution.
Among the multiple dysregulated signaling mechanism in HNSCC, we have documented that the vast majority of the HNSCC lesions exhibit activation of the Akt-mTOR signaling route (17), a central component of multiple growth promoting and metabolic pathways. Thus, the availability of these HNSCC tissue arrays provided an opportunity to examine whether HPV-associated HNSCC also involve Akt-mTOR activation, in spite of their distinct etiology. We observed that most p16 positive HNSCC cases exhibited a remarkable activation of the mTOR pathway, similar to HNSCC associated with more traditional risk factors These observations were validated in a large collection of HNSCC cell lines and HNSCC cases of known HPV status and in a newly developed cervical cancer tissue array, thus confirming that HPV-associated SCCs display overactive mTOR pathway irrespective of their anatomical site of origin. These findings prompted us to explore the pre-clinical efficacy of mTOR inhibition in HPV+ oral and cervical tumor xenografts. We show here that two widely used mTOR inhibitors in the clinic, rapamycin and RAD001, effectively decrease mTOR activity in vivo, and promote the rapid collapse of HPV-associated oral and cervical tumors, resulting in a remarkable decrease in tumor burden. Collectively, the emerging results may provide a rationale for the clinical evaluation of mTOR inhibitors as a molecular targeted approach for the treatment of HPV-associated malignancies.
Materials and Methods
Chemical, Reagents, Cell Culture and Western Blot Analysis of Akt-mTOR Pathway
All chemicals and reagents were from Sigma-Aldrich (St Louis, MO, USA), unless indicated. HN12, HN13 and HN30 cells were obtained from Dr. J.F. Ensley (Wayne State University, Detroit, MI) (21). Cal33, and Cal27 and HeLa cells were from DSMZ (Braunschweig, Germany), and ATCC (VA, USA), respectively. UDSCC2, SCC47, SCC90 and 93VU147T cell lines were from Dr. Henning Bier, University of Duesseldorf, Dr. Thomas Carey, University of Michigan, Dr. Susan Gollin, University of Pittsburgh, and Dr. Steenbergen, VUMC, Netherlands, respectively. Spontaneously immortalized normal oral keratinocytes (NOKSI) were established as reported (22). All cells prior to experiments were authenticated by PCR amplification of short tandem repeats (Genetica DNA Laboratories, Inc., OH, USA), to ensure cell identity. Detailed information of cell culture conditions, antibodies use throughout the study, and Western Blot analysis of Akt-mTOR pathway in cell lysates (essentially as reported in (23)) are described in detail in Supplemental Material.
Assessment of Genomic HPV Status in HNSCC cell Lines and in HNSCC Lesions
DNA from exponentially growing cells was extracted and used as template to amplify by PCR the HPV genome as previously described (24). DNA was also isolated from tissue sections and used for HPV genotyping using a nested multiplex PCR protocol that combines degenerate E6/E7 consensus primers and type-specific primers, as previously reported (25). See Supplemental Material for details.
Animal Studies
All animal studies were carried out according to NIH-approved protocols (ASP# 10-569), in compliance with the NIH Guide for the Care and Use of Laboratory Animals. All cell and animal handling and tumor transplantation into the animal flanks are described in detail in Supplemental Material. Briefly, all animals bearing HNSCC tumor xenografts underwent biweekly evaluation for disease onset, and the volume of the tumor lesions determined as described (23). Animals for each experiment were randomized into three treatment arms (20 xenografts each) consisting of daily intra-peritoneal (ip) injections of rapamycin or RAD001 (5 mg/kg/day), and control diluent. Animals were euthanized at the indicated time points and tissues retrieved for histological and immunohistochemical evaluation. See Supplemental Materials for a detailed description of all procedures used.
Tissues and Immunohistochemistry (IHC) Analysis
For immunohistochemical analysis, samples from the Head and Neck Tissue Microarray (TMA) Initiative (17) were used, containing 317 evaluable cores from formalin-fixed, paraffin-embedded cases of HNSCC. Extensive information on the Akt and mTOR pathways were used based on prior analysis (17). A detailed description of the development of a cervical cancer TMA and immunostaining techniques for Akt and mTOR pathways and p16 in tissue cores and individual cases of HNSCC with known HPV infectious status is provided in Supplemental Materials.
Statistical analysis
Two-way ANOVA followed by Bonferroni’s test was used to analyze the differences in xenograft tumor volumes between the three experimental groups for each of the two cell lines. Covariation of the expression of the different Akt-mTOR markers was analyzed with the nonparametric Spearman correlation (Spearman’s rank correlation test). Differences in immunohistochemical reactivity between human normal tissues and HNSCC classified into different differentiation groups were analyzed using the Kruskall-Wallis test for non-Gaussian populations (nonparametric ANOVA) followed by the Dunn’s posttest to compare all pairs of columns (GraphPad Software). Data analysis was done using GraphPad Prism (GraphPad Software, La Jolla, CA, USA), and P values of <0.05 were considered statistically significant. See Supplemental Material for additional details. Fisher’s exact test was use to evaluate differences in the proportion of cases exhibiting 1–25, 26–50, 51–75, and 76–100% of cells stained positive for pS6 and pAktS473 between HPV− and HPV+ HNSCC human samples. Differences in the immunoreactivity in individual HPV− and HPV+ cases and non-neoplastic oral mucosal tissues were evaluated by 1 way ANOVA. The distribution of HPV cases among different countries was evaluated using Chi-square analysis.
Results
Incidence of HPV infection in HNSCC and Cervical Tumors
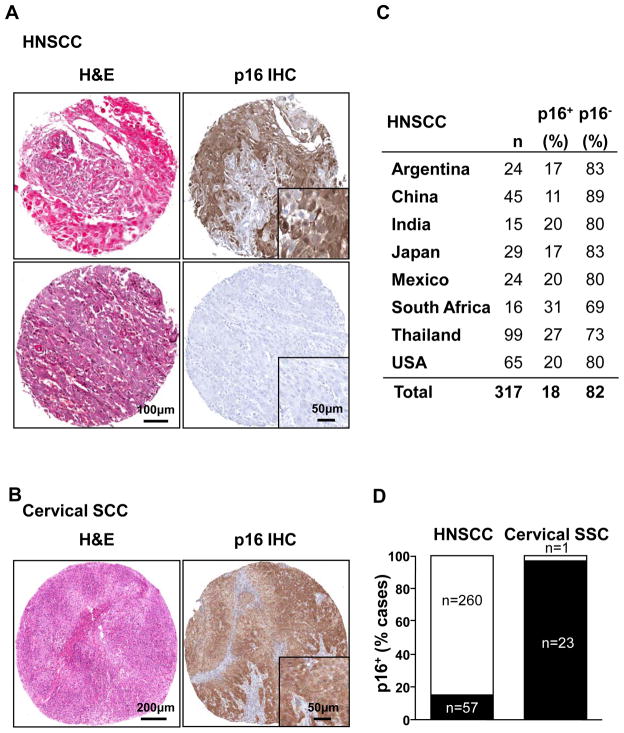
To begin exploring the worldwide impact of HPV-associated oral cancers, we evaluated the expression of p16, a validated surrogate marker for HPV infection in a HNSCC tissue microarray consisting of several hundred cores of formalin-fixed, paraffin-embedded cases of HNSCC from America (Argentina, Mexico, and the USA), Africa (South Africa), and Asia (China, India, Japan, and Thailand) (17). A total of 57 out of 317 cases (18%) showed a strong cytoplasmic and nuclear staining signal for p16, which was nearly homogenous throughout the malignant epithelium (Fig 1, top right). Higher number of p16 positive cases (p16+) were observed in Thailand and South Africa, and lower incidence was observed in China, albeit these differences were not statistically significant. In the USA, 20% of the HNSCC cases were found to be p16+, aligned with the current estimation of the incidence of HPV-associated HNSCC cases (26). As most cervical cancers are HPV-associated (1), we developed a cervical cancer TMA as a positive control. All but one of the cases were strongly positive for p16 (Fig 1, bottom left), with a pattern similar to that of the oral cancer lesions. The presence of SCC tissue was confirmed by H&E (Fig 1, left top and bottom images). The data are aligned with the use of p16 as a biomarker for the detection of HPV-associated cancers (20), and hence indicate that a subset of HNSCC lesions is associated with HPV infection, irrespective of the demographic distribution of the HNSCC cases.
Figure 1.
Distribution of p16 positive cases in squamous cell carcinoma of the head and neck (HNSCC) and uterine cervix (Cervical SCC). A, the upper figure shows representative cores of HNSCC lesions (HNSCC) stained with H&E and p16 (p16 IHC), with strong nuclear and cytoplasm staining (top panel). A negative HNSCC lesion is shown below. B, a uterine cervical carcinoma (Cervical SCC) TMA was used as positive control for p16 staining. C, about 18% of the HNSCC cases were positive for p16 by immunohistochemistry, while only 1 of the 24 cases of cervical SCC studied of was negative for p16. C, the distribution of p16+ HNSCC was similar in all 8 countries analyzed, although South Africa and Thailand had a relatively higher number of cases.
Aberrant mTOR pathway Activity in HPV-Associated HNSCC
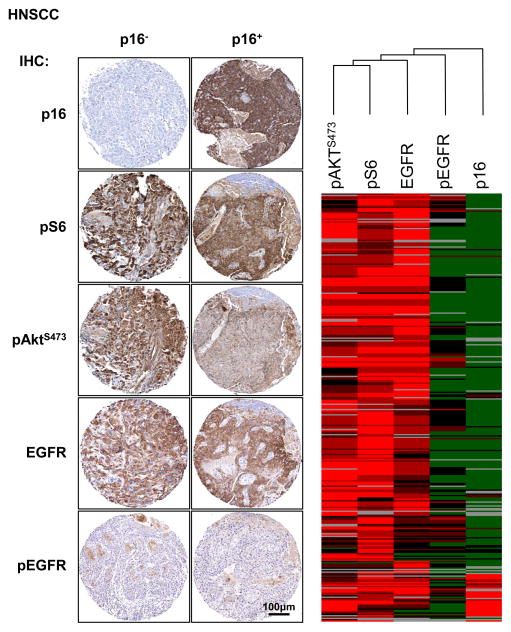
mTOR is a protein kinase involved in multiple cellular functions, including cell growth promotion, which can be found as part of two protein complexes, mTOR complex 1 (mTORC1) and mTORC2 (27). Thus, we next correlated the expression of p16 with that of pS6 and AktS473, which are downstream phosphorylation targets of mTORC1 and mTORC2, respectively (27). The quantification of the immunostainings was used for cluster analysis to generate a heat-map, as previously described (17). The p16+ cluster (positive cases in red, bottom, right) highly correlated with the positive expression of pAktS473 and pS6 (Figure 2, right). Most of the p16+ cases were also positive for EGFR, but negative for pEGFR. The left panel shows representative immunostaining of the proteins analyzed in p16− and p16+ lesions. These data demonstrate that most p16+ HNSCCs exhibit over activity of the mTOR pathway, similar to that of p16− HNSCC, likely independent from the activation of EGFR. A similar broad activation of the mTOR signaling network was observed in most cases of cervical SCC (see below, Figure 3), supporting the emerging notion that Akt-mTOR stimulation may represent an intrinsic feature of most HPV-related human carcinomas.
Figure 2.
The expression of p16, pAktS473, pS6, EGFR and pEGFR was evaluated by immunohistochemistry in HNSCC TMAs, and this information was used as input to generate a heat-map. The right panel shows a series of representative immunostaining in p16+ and p16− negative samples. As shown in the heat map, cases positive for p16 display an increased pAktS473 and pS6 expression. Elevated levels of EGFR but not of pEGFR were detected in p16+ samples.
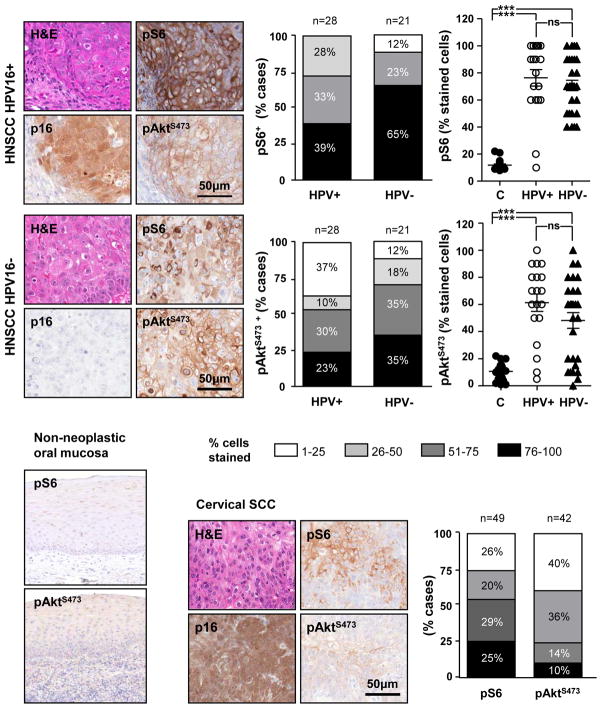
Figure 3.
HNSCC and cervical SCCs cases were analyzed by IHC for p16, pAktS473, and pS6 expression. The figure shows a representative squamous cell carcinoma (H&E) of each group. All cervical SCC and HNSCC HPV+ cases were strongly reactive for p16 in both nuclei and cytoplasm, and this was absent in the HNSCC HPV− cases (p16). Strong immunodetection of pS6 and pAktS473, with preferential cytoplasmic and membranous distribution, respectively, were observed in both cervical SCC and HNSCC HPV+ and HPV− cases, while non neoplastic oral mucosa shows very limited pS6 and pAktS473 immunostaining (bottom right). Quantification of IHC staining based on the % of stained cells in each tumoral lesion shows remarkable activation of the mTOR pathway, as reflected by the pS6 staining in a high percentage of the cervical cancers and HNSCC, the latter in both HPV+ and HPV− groups. For pAktS473, however, the fraction of immunostained cases is smaller in cervical SCC and HNSCC HPV+ lesions. The graphical representation of each individual HNSCC case (right graphs) demonstrates that both HPV+ (n=28) and HPV− (n=21) exhibit a highly significant increase in pS6 and pAktS473 with respect to the levels of the phosphorylated forms of S6 and Akt in non-neoplastic oral tissues (C) (n=19 and n=20, respectively). No significant differences were observed in the level of activation of Akt-mTOR pathway molecules when comparing HPV− and HPV+ HNSCC lesions (ns).
Analysis of the Akt-mTOR Pathway in HNSCC lesions with known HPV status
As our data suggested an aberrant activation of the Akt-mTOR pathway in patients with HPV-associated oral cancer, we validated these results by undertaking a molecular case-control study approach using a cohort of HNSCC cases that were clinically defined for presence (n=28) or absence (n=21) of HPV (+/−) by HPV E6 expression (25). When the cases in the two groups were classified based on their differentiation patterns (Supp. Table 1), the HPV− group had a higher number of well differentiated cases whereas the HPV+ cohort included a higher number of poorly differentiated cases, reflecting a trend often observed in prior studies (reviewed in (7). As expected, most HPV+ cases were distributed around the area of the base of the tongue or in the tonsil, whereas the HPV− cases originated from multiple anatomical locations throughout the oral cavity, tongue, an oropharynx (Supp. Table 2). Representative histological images illustrate morphological features of HPV+ and HPV− oral tumors and specific patterns of immunoreactivity of different target proteins (Fig 3). H&E staining show the typical features of moderately differentiated squamous cell carcinomas. In the HPV+ sample, p16 is strongly expressed in both nuclei and cytoplasm of the malignant cells, while pS6 shows essentially a cytoplasmic staining, and pAktS473 is strongly reactive in the cytoplasmic membrane. No evident differences in the distribution of the targeted Akt/mTOR pathway proteins were observed across HPV+ an HPV− groups (Fig 3, top panel). There was a near perfect correlation between the p16 staining and the presence of HPV DNA, with only one discordant case. In HPV+ lesions all cases gave positive reaction for pS6, whereas ~90% of HPV− cases were positive (Fig 3, bottom, left). Further indication of an active mTOR pathway, high levels of pAKTS473 were present in most HPV+ cases (Fig 3, lower right). Some variations were observed in Akt phosphorylation, being higher in HPV− (88%) than in HPV+ (60%) carcinomas, and S6 phosphorylation being higher in HPV+ cases. However, statistical analysis of the individual HPV− and HPV+ HNSCC cases indicate that there are no significant differences in pAKTS473 and pS6 staining when comparing both groups of HNSCC, with most HNSCC lesions displaying highly elevated mTOR signaling activity when comparing to non-neoplastic oral mucosal tissue samples. Overall, we can conclude that both HPV− and HPV-associated HNSCC exhibit an overactive mTOR pathway.
Activation of Akt-mTOR in HPV+ HNSCC cell lines; response to rapalogs
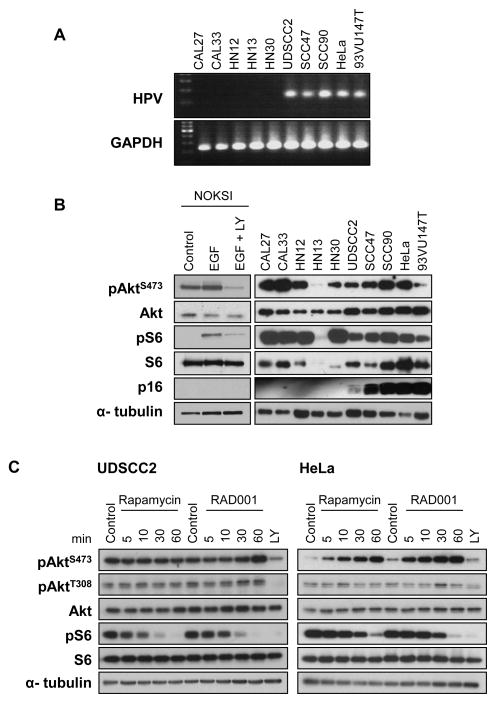
As the Akt-mTOR pathway was observed to be activated in HPV+ and HPV− HNSCC cases, we next investigated whether this was reflected in a representative panel of HPV+ and HPV− HNSCC derived cell lines in vitro. Initially, we analyzed the HPV status of a large collection of HNSCC cells by PCR, (Fig. 4A), and this enabled the identification of four oral cancer cell lines, UD-SCC2, SCC47, SCC90, and 93VU47T, which were HPV+ as judged by the amplification of a HPV-specific sequence, which was observed as a DNA band of the expected size (150 bp) when compared with the positive control (HeLa cells). GAPDH amplification was used to demonstrate intact DNA integrity across all samples. p16 was readily detectable in UD-SCC2, SCC47, SCC90, 93VU147T and HeLa cells, thus matching the detection of the HPV genome by PCR. pAktS473 and pS6 levels were elevated in all HPV+ and HPV− cell lines tested, except HN13, which we have used as a HNSCC premalignant models (28). As a control, immortalized normal oral keratinocyte cell line, NOKSI (22), which did not express p16, showed increased levels of pAktS473 and pS6 after EGF stimulation that was prevented by the treatment with a pan PI3K inhibitor, LY294002. We next chose two representative oral and cervical SCC HPV+ cell lines, UD-SCC2 and HeLa cells, respectively, both of which grow readily as tumor xenografts (see below) to examine the biochemical consequences of mTOR inhibition using two clinically relevant rapalogs, rapamycin and RAD001. Both rapalogs had a marginal effect on Akt activity in UDSCC2 cells, while in contrast, HeLa cells showed a notable increase in pAktS473 (Fig. 4C). However, we did not observe an increase in the level of pAktT308, the active form of Akt (23, 29), which was abolished by the use of the PI3K inhibitor. Thus, while the differences in the impact of rapamycin and RAD001 on pAktS473 in oral and cervical cancer cells require further examination, most likely these cells do not exhibit an increase in Akt activity after mTOR inhibition. In any case, both rapamycin and RAD001 caused a time dependent reduction in pS6 activity, with complete ablation occurring after 60 min of treatment, suggesting that HPV+ oral and cervical cancer cells lines are sensitive to clinically relevant rapalogs.
Figure 4.
A, A representative series of HNSCC cell lines were assessed by PCR for the presence of HPV DNA; 4 out of 9 HNSCC cell lines were positive, using cervical cancer HeLa cells as a positive control. B, cells were evaluated in parallel by Western blot for expression levels of Akt, pAktS473, S6, pS6, and p16, as indicated. α-Tubulin was used as a loading control. The Akt and mTOR pathways were significantly activated in the vast majority of the cell lines investigated, irrespective of their p16 status. Immortalized oral keratinocytes, NOKSI cells, representative of a normal phenotype, were used as control cells to simulate (EGF) and inhibit (EGF + LY) the Akt-mTOR pathway. C, two HPV-associated HNSCC and cervical cancer-derived cell lines, UDSCC2 and HeLa, respectively, were used to validate the biochemical activity of clinically relevant inhibitors allosteric mTOR inhibitors, rapamycin and RAD001, on the Akt-mTOR pathway. As seen, exposure of cells to 100 nM of these inhibitors caused a time dependent reduction in the levels of pS6, while they increased the levels of pAktS473 in HeLa but not in HNSCC cells.
Rapamycin and RAD001 Inhibits the growth of HPV-Associated Tumor Xenografts
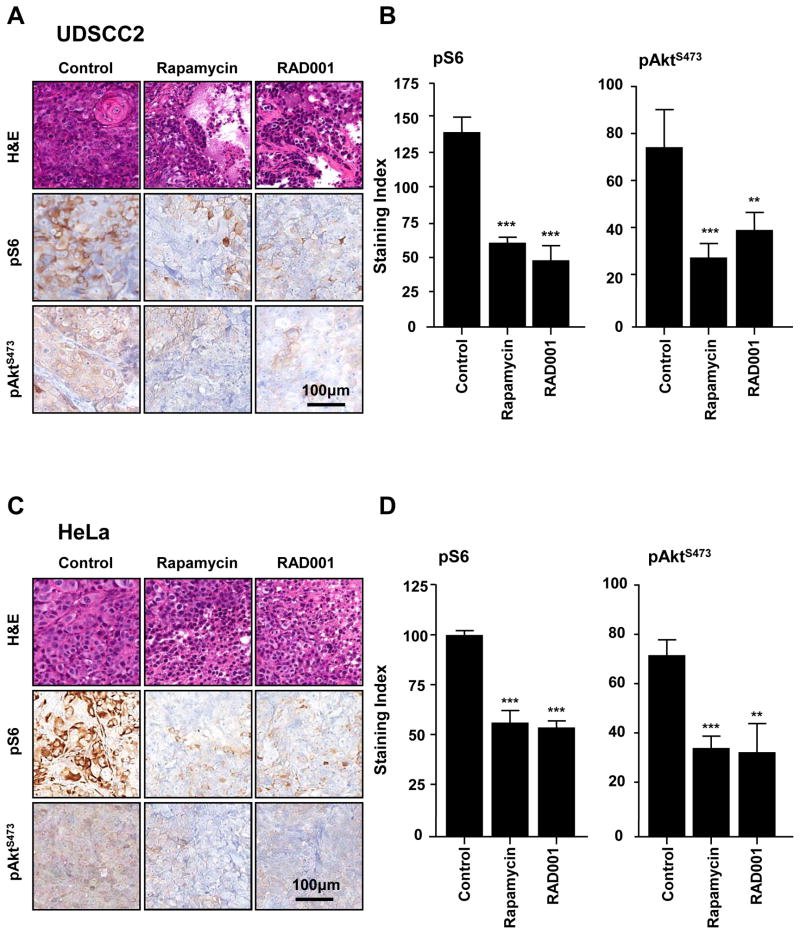
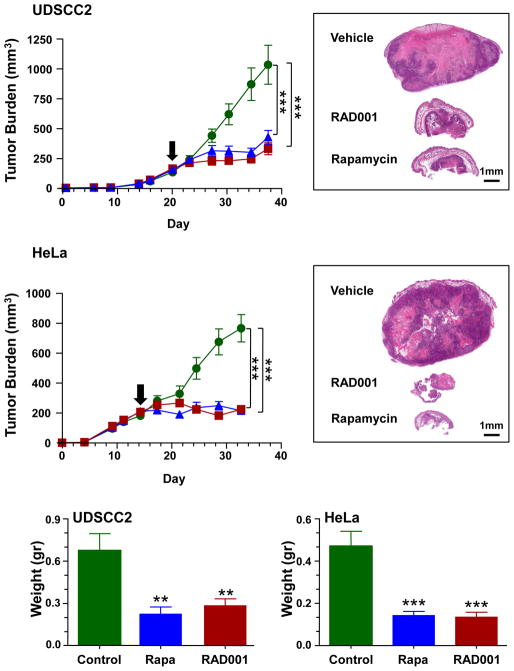
We next sought to examine if the aberrant activation of Akt-mTOR in HPV+ oral and cervical cancers could be exploited for therapeutic purposes. For these studies we choose UDSCC2 and HeLa cells, as representative HPV+ oral and cervical squamous cancers, respectively, which grow efficiently as tumor xenografts in athymic nude mice. We first confirmed that administration of the rapamycin and RAD001 would effectively result in the inhibition of the targeted pathways. Morphologically UDSCC2 is a moderately differentiated squamous cell, whereas HeLa tumors display the features of a poorly differentiated cancer (Fig. 5). In both UDSCC2 and HeLa, treatment for three days with rapamycin and RAD001 reduced the expression levels of pS6 and pAktS473, (Fig 5B and D). Thus, both rapalogs were able to hit their expected therapeutic molecular targets in vivo, including mTOR in its mTORC1 complex, as judged by the decrease in pS6, and likely indirectly on mTORC2, as it can be inferred from the decrease in pAktS473. Next, tumor bearing animals were randomized and treated with vehicle control, rapamycin, or RAD001 when the average volume was ~200–250 mm3. There was a remarkable response to rapamycin and RAD001 in UMDSCC2 and HeLa cells, when compared with the corresponding control groups (Fig 6). Indeed, statistically significant differences in tumor burden were observed as early as few days after treatment initiation. The dramatic effects of both inhibitors, as depicted in the tumor pictures, was further supported by the highly significant differences in the tumor weights of the control and treated groups at the end of the observation period (Fig 6, bottom panel). Similar results were also obtained in multiple human HNSCC xenograft models derived from HPV− HNSCC cells (23). HPV+ treated tumors are small and quiescent, with necrotic areas and strands of tumor cells without evidence of mitosis embedded into a fibrous collagen matrix. Overall, these findings suggest that similar to HPV− HNSCC, HPV-associated squamous tumors may respond favorably to mTOR inhibitors.
Figure 5.
Excised tumors from athymic mice treated for 3 days with rapamycin, RAD001 or control diluent, were processed for histological evaluation. A, UDSCC2 tumors grow in control treated animals (upper left) as moderately differentiated squamous cell carcinomas, with solid sheets of cells occasionally forming keratinized clumps of cells. In animals treated with rapamycin or RAD001, edema and areas of initial tissue disruption were observed (upper, middle and right). The expression of both pS6 and pAktS473 was significantly decreased by treatment with both mTOR inhibitors, as seen in individual tissues by immunohistochemistry (A, middle and bottom row) and the quantification of multiple treated tumors (B). C, A very similar profile was observed with HeLa xenografts, which are poorly differentiated squamous cell carcinomas (left). The treated tumors showed features similar to those described for UDSCC2 (upper row, middle and right). A decrease in the expression of pS6 and pAktS473 is evident upon treatment with both mTOR inhibitors (C, middle and bottom row) as further confirmed by quantification of the immunoreactions (D). (** p<0.01; ***p<0.001).
Figure 6.
Athymic mice were transplanted with either UDSCC2 or HeLa cells. When the tumor volume reached approximately 200 mm3, the animals were randomized and treated daily ip with control diluent, or 5 mg/kg of rapamycin or RAD001. Tumor growth curves show significant size decrease upon RAD001 and rapamycin treatment with respect to control treated mice in both UDSCC2 and HeLa xenografts (n=20 xenografts in each treatment arm; *** p<0.001). A representative histological picture of each treatment group and cell lines is shown in the rights panels. The antitumor effect of rapamycin and RAD001 was further confirmed by evaluating tumor weights (bottom) at the end of the treatment period (n=20, ** p<0.01; ***p<0.001).
Discussion
The incidence rate of most cancer sites related to HPV infection has increased in the United States and in some European countries in the past two decades (7, 8). Anatomically, HPV-associated HNSCC appear to arise predominantly from the oropharynx, including base of base of tongue and tonsils (9, 30). In general, HPV+ oral cancers have a better prognosis, with increased survival and response to surgical treatment (11), radiotherapy (12, 13), and combined treatment approaches (14–16). However, HPV-positive HNSCC patients are significantly younger than those associated with traditional risk factors (8). This distinct demographic distribution may raise concerns about the potential increase in HNSCC recurrence given the longer life expectancy of HPV-associated HNSCC patients, and about the long-term consequences of exposing younger patients to radiation and aggressive cytotoxic treatments often used in HNSCC. Thus, there is an urgent need for the development and evaluation of novel molecular-targeted therapeutic approaches to treat HPV-related HNSCCs, hence reducing its morbidity and mortality, and increasing the overall patient’s quality of life. Similarly, surgery, chemoradiation with adjuvant or combination chemotherapy are the standard of care for cervical cancer patients (31, 32), highlighting the need for the development of less aggressive targeted therapeutic options. Indeed, we provide evidence that HPV-related cervical and HNSCC cases exhibit elevated mTOR function, and that clinically relevant mTOR inhibitors can decrease mTOR activity in the target tissues, thereby significantly reducing tumor burden in preclinical animal models for these HPV-associated squamous malignancies.
To begin evaluating the impact of HPV in HNSCC worldwide, we immunostained and quantified the expression of p16, which has been recognized as a reliable surrogate marker for high-risk HPV infection (20). This protein is encoded by the CDKN2A (MTS1, INK4A) tumor suppressor gene, and inactivates the function of cdk4- and cdk6-cyclin D complexes, thereby regulating cell proliferation by preventing the inactivation of the retinoblastoma tumor suppressor protein (pRb) by hyperphosphorylation (33). In tumors with biologically active HPV, E7 protein forms complexes with hypophosphorylated forms of the pRb, resulting in a decrease of the cellular pRb level and a release of E2F, a transcription factor involved in cell cycle progression (34). In these tumors, the inactivation of pRb by HPV E7 protein may lead to p16 overexpression because Rb normally represses p16 transcription (35). Alternatively, the high risk HPV E7-mediated increased expression of the histone demethylases KDM6A and KDM6B may result in de-repression of p16 gene transcription, thus resulting in high levels of p16 (36). Hence, p16 immunohistochemistry is considered comparable or even superior to in situ hybridization for the detection of high-risk HPV (18). However, negative p16 staining have been also described in HPV+ tumors, due to allelic loss and/or promoter hypermethylation (37), which might explain a single case of cervical SCC that was negative for p16. In HNSCC, ~18% of all cases from a large international collection of HNSCC lesions were p16+, suggesting that the impact of HPV-infection in oral cancer has reached a worldwide proportion. Among the tissues analyzed, the cases from South Africa and Thailand had a higher percentage of HPV-positive cases, with cases from China being the lowest. While these differences among countries did not reach statistical significance, likely due to the limited number of HPV+ cases for some of the geographical regions, these findings suggest the existence of distinct regional- and country-specific rates of HPV-related oral malignancies. This possibility may need to be investigated further, aiming at implementing appropriate preventing and treatment strategies.
The mTOR-regulated molecular network coordinates mitogenic signaling with nutrient-sensing pathways thereby controlling protein translation and synthesis, autophagy, cellular metabolism, and cell migration (27). This pathway is often initiated by growth factor- or oncogene-induced activation of PI3K, and the consequent activation of Akt upon its phosphorylation in threonine 308 by the PI3K-dependent stimulation of a kinase known as PDK1 (27). Active, pAktT308 then phosphorylates and inactivates a tumor-suppressor protein, tuberous sclerosis complex protein 2 (TSC2), which suppresses the function of the small GTPase Rheb1. This inactivation of TSC2 by Akt leads to the accumulation of the active Rheb1, which in turn promotes the activation of mTOR in its complex mTORC1. mTORC1 then phosphorylates p70-S6 kinase (p70S6K, S6K), which phosphorylates ribosomal protein S6, leading to the accumulation of pS6 (27). Amino acid and metabolic-regulated circuitries stimulate mTOR in its complex mTORC2, which phosphorylate multiple protein targets, including Akt in its serine 473, resulting in increased pAktS473 (27). In this context, the molecular mechanisms resulting in increased activation of mTOR by HPV are at the present not fully understood. High risk E6 causes the rapid degradation of TSC2, resulting in TORC1 activation (38). In stable HPV-infected cells, however, the levels of TSC2 are not greatly affected (39). In line with this, we did not observe consistent changes in TSC2 expression in HPV+ HNSCC cell lines (not shown). Of interest, HeLa cells are defective in the tumor suppressor protein LKB1 (40), also known as STK11, which inhibits mTOR by a pathway impinging on TSC2 stimulation (41). Thus, we can speculate that in addition of persistent expression of high-risk HPV E6 and E7 proteins, additional genetic and epigenetic events resulting in mTOR activation may be required for the development of HPV-related malignancies. In this regard, most HPV+ lesions do not express elevated levels of phosphorylated, active EGFR (see Figure 2), and while a fraction of HNSCC harbor activating mutations in PI3K, no mutations in PI3K were identified in the HPV+ HNSCC cell lines used in this study (not shown). Preliminary data, however, suggest that nearly 30% of the HPV+ cases may exhibit reduced expression of the tumor suppressor protein PTEN, which is a lipid phosphatase counteracting PIP3 accumulation by PI3K (42). Thus, while additional studies may be required to confirm the contributing role of PTEN and other candidate oncogenic and tumor suppressive molecules regulating mTOR in HPV-associated malignancies, we can conclude that activation of the mTOR pathway represents a common feature of nearly all HPV-associated cervical and oral SCCs, an observation that can now be exploited for therapeutic strategies.
mTORC1 is activated in most HPV-associated SCCs, including oral and cervical carcinomas, as judged by readily detection of accumulated of pS6. Regarding cervical SCCs, this observation is aligned with prior reports describing the presence of phosphorylated p70S6 kinase in cervical SCCs (43), although pS6 appears to be a more reliable marker to document TORC1 activation by IHC than its upstream kinase p70S6K (44). However, fewer HPV+ HNSCC cases were positive for pAktS473 than HPV- HNSCC lesions, 60% and 88% of the cases, respectively. This difference was even more dramatic in cervical SCCs, as only approximately (60%) were stained positive for pAktS473, even if most of these HPV-associated lesions exhibit elevated pS6. This information has significant implications, as it suggests that HPV+ SCC cases have more limited activation of mTORC2 than mTORC1, and hence may be more dependent for their growth of the mTORC1. Indeed, we observed that xenografts of HPV+ SCC cells are highly sensitive to allosteric inhibitors of mTORC1, rapamycin and RAD001, thus supporting their preclinical efficacy of targeting mTOR in these tumor lesions.
On the other hand, blockade of mTORC1 in vitro resulted in the fast increase of pAktS473 in cervical but not oral SCC-derived cells. The reason for this notable difference is unclear. In this regard, the disruption of negative feedback loops by rapalogs has been reported, which may result in Akt activation (45). However, we did not observe an increase in the level of pAktT308, which represents the active form of Akt (27). Thus, as we and others have previously reported, HNSCC cells do not exhibit an increase in Akt activity after mTORC1 inhibition with rapamycin, perhaps a distinct property that may contribute to its potential clinical efficacy (23, 46). The transient activation of the mTOR mTORC2 complex, which phosphorylates Akt in serine 473 (27), in HeLa cells may require further exploration. Nonetheless, both rapalogs, rapamycin and RAD001, caused a clear reduction in pAktS473 in vivo, suggesting that prolonged inhibition of mTORC1 may results in mTORC2 inhibition, as reported in HPV− HNSCC xenografts (23), in HNSCC patients treated with a rapamycin pro-drug, CCI-779 (47), and in certain cellular systems (48).
Of interest, our emerging results suggest that the inhibition of mTOR by rapamycin and RAD001 may result in potential therapeutic benefit in HPV− HNSCC as well as in HPV+ oropharyngeal and cervical SCCs. Indeed, both rapalogs had a remarkable antitumor activity when evaluated in HPV− xenograft models (23) as well as when HPV16+ oral cancer and HPV18+ cervical cancer cells were xenografted in mice. Overall, our present observations may provide a strong rationale for the clinical evaluation of rapalogs in management of HPV-associated malignancies. Specifically in the case of oral cancer, the clinical consequences of radiation and chemotherapy and their combinations are usually associated with significant side-effects that result in a poor quality of life and severe chronic functional problems that have proven difficult to address, including dysphagia, xerostomia, feeding-tube dependency from fibrosis and scarring of the pharyngeal muscles, chronic aspiration, and chronic fatigue. These side-effects are not known to be associated with mTOR inhibitors (49). Thus, we can envision that mTOR blocking agents may represent a promising therapeutic approach in HPV+ oral and cervical cancers, as a single agent in a neoadjuvant setting, to lower the dose of radiation/chemotherapy required for local control, or after conventional surgical excision of the tumor with or without chemoradiation, to prevent or delay tumor recurrence.
Supplementary Material
Clinical and Translational Relevance.
The emerging results may provide a rationale for the early evaluation of the clinical efficacy of mTOR inhibitors as a molecular targeted approach for the treatment of HPV-associated malignancies.
Acknowledgments
This article was supported by the Intramural Research Program of the National Institutes of Health, National Institute of Dental and Craniofacial Research, project Z01DE00558. Dr. Tanguy Seiwert was supported by a Flight Attendant Medical Research Institute Young Clinical Scientist Award (FAMRI YCSA). Dr. Nitin Gangane was supported by a Yamagiwa-Yoshida fellowship from the Union for International Cancer Control, International cancer technology transfer fellowships (ICRETT).
References
- 1.zur Hausen H. Papillomaviruses in the causation of human cancers - a brief historical account. Virology. 2009;384:260–5. doi: 10.1016/j.virol.2008.11.046. [DOI] [PubMed] [Google Scholar]
- 2.Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118:3030–44. doi: 10.1002/ijc.21731. [DOI] [PubMed] [Google Scholar]
- 3.Parkin DM, Whelan SL, Ferlay J, Teppo L, Thomas DB. Cancer Incidence in Five Continents. Lyon; 2005. [Google Scholar]
- 4.Warnakulasuriya S. Causes of oral cancer--an appraisal of controversies. Br Dent J. 2009;207:471–5. doi: 10.1038/sj.bdj.2009.1009. [DOI] [PubMed] [Google Scholar]
- 5.de Villiers EM, Weidauer H, Otto H, zur Hausen H. Papillomavirus DNA in human tongue carcinomas. Int J Cancer. 1985;36:575–8. doi: 10.1002/ijc.2910360510. [DOI] [PubMed] [Google Scholar]
- 6.Syrjanen SM, Syrjanen KJ, Lamberg MA. Detection of human papillomavirus DNA in oral mucosal lesions using in situ DNA-hybridization applied on paraffin sections. Oral Surg Oral Med Oral Pathol. 1986;62:660–7. doi: 10.1016/0030-4220(86)90262-8. [DOI] [PubMed] [Google Scholar]
- 7.Marur S, D’Souza G, Westra WH, Forastiere AA. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol. 2010;11:781–9. doi: 10.1016/S1470-2045(10)70017-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chaturvedi AK, Engels EA, Anderson WF, Gillison ML. Incidence Trends for Human Papillomavirus-Related and -Unrelated Oral Squamous Cell Carcinomas in the United States. J Clin Oncol. 2008;26:612–9. doi: 10.1200/JCO.2007.14.1713. [DOI] [PubMed] [Google Scholar]
- 9.D’Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, et al. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007;356:1944–56. doi: 10.1056/NEJMoa065497. [DOI] [PubMed] [Google Scholar]
- 10.Heck JE, Berthiller J, Vaccarella S, Winn DM, Smith EM, Shan’gina O, et al. Sexual behaviours and the risk of head and neck cancers: a pooled analysis in the International Head and Neck Cancer Epidemiology (INHANCE) consortium. Int J Epidemiol. 2010;39:166–81. doi: 10.1093/ije/dyp350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Licitra L, Perrone F, Bossi P, Suardi S, Mariani L, Artusi R, et al. High-risk human papillomavirus affects prognosis in patients with surgically treated oropharyngeal squamous cell carcinoma. J Clin Oncol. 2006;24:5630–6. doi: 10.1200/JCO.2005.04.6136. [DOI] [PubMed] [Google Scholar]
- 12.Lindquist D, Romanitan M, Hammarstedt L, Nasman A, Dahlstrand H, Lindholm J, et al. Human papillomavirus is a favourable prognostic factor in tonsillar cancer and its oncogenic role is supported by the expression of E6 and E7. Mol Oncol. 2007;1:350–5. doi: 10.1016/j.molonc.2007.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lassen P, Eriksen JG, Hamilton-Dutoit S, Tramm T, Alsner J, Overgaard J. Effect of HPV-associated p16INK4A expression on response to radiotherapy and survival in squamous cell carcinoma of the head and neck. J Clin Oncol. 2009;27:1992–8. doi: 10.1200/JCO.2008.20.2853. [DOI] [PubMed] [Google Scholar]
- 14.Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tan PF, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363:24–35. doi: 10.1056/NEJMoa0912217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fakhry C, Westra WH, Li S, Cmelak A, Ridge JA, Pinto H, et al. Improved Survival of Patients With Human Papillomavirus-Positive Head and Neck Squamous Cell Carcinoma in a Prospective Clinical Trial. J Natl Cancer Inst. 2008;100:261–9. doi: 10.1093/jnci/djn011. [DOI] [PubMed] [Google Scholar]
- 16.Settle K, Posner MR, Schumaker LM, Tan M, Suntharalingam M, Goloubeva O, et al. Racial survival disparity in head and neck cancer results from low prevalence of human papillomavirus infection in black oropharyngeal cancer patients. Cancer Prev Res (Phila) 2009;2:776–81. doi: 10.1158/1940-6207.CAPR-09-0149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Molinolo AA, Hewitt SM, Amornphimoltham P, Keelawat S, Rangdaeng S, Meneses Garcia A, et al. Dissecting the Akt/mammalian target of rapamycin signaling network: emerging results from the head and neck cancer tissue array initiative. Clin Cancer Res. 2007;13:4964–73. doi: 10.1158/1078-0432.CCR-07-1041. [DOI] [PubMed] [Google Scholar]
- 18.Kong CS, Balzer BL, Troxell ML, Patterson BK, Longacre TA. p16INK4A immunohistochemistry is superior to HPV in situ hybridization for the detection of high-risk HPV in atypical squamous metaplasia. Am J Surg Pathol. 2007;31:33–43. doi: 10.1097/01.pas.0000213347.65014.ee. [DOI] [PubMed] [Google Scholar]
- 19.Hafkamp HC, Speel EJM, Haesevoets A, Bot FJ, Dinjens WNM, Ramaekers FCS, et al. A subset of head and neck squamous cell carcinomas exhibits integration of HPV 16/18 DNA and overexpression of p16INK4A and p53 in the absence of mutations in p53 exons 5–8. Int J Cancer. 2003;107:394–400. doi: 10.1002/ijc.11389. [DOI] [PubMed] [Google Scholar]
- 20.Adelstein DJ, Ridge JA, Gillison ML, Chaturvedi AK, D’Souza G, Gravitt PE, et al. Head and neck squamous cell cancer and the human papillomavirus: summary of a National Cancer Institute State of the Science Meeting, November 9–10, 2008, Washington, D.C. Head Neck. 2009;31:1393–422. doi: 10.1002/hed.21269. [DOI] [PubMed] [Google Scholar]
- 21.Jeon GA, Lee JS, Patel V, Gutkind JS, Thorgeirsson SS, Kim EC, et al. Global gene expression profiles of human head and neck squamous carcinoma cell lines. Int J Cancer. 2004;112:249–58. doi: 10.1002/ijc.20399. [DOI] [PubMed] [Google Scholar]
- 22.Castilho R, Squarize CH, Leelahavanichkul K, Zheng Y, Bugge T, Gutkind JS. Rac1 is required for epithelial stem cell function during dermal and oral mucosal wound healing but not for tissue homeostasis in mice. PLos One. 2010;5:e10503. doi: 10.1371/journal.pone.0010503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Amornphimoltham P, Patel V, Sodhi A, Nikitakis NG, Sauk JJ, Sausville EA, et al. Mammalian target of rapamycin, a molecular target in squamous cell carcinomas of the head and neck. Cancer Res. 2005;65:9953–61. doi: 10.1158/0008-5472.CAN-05-0921. [DOI] [PubMed] [Google Scholar]
- 24.de Roda Husman AM, Walboomers JM, van den Brule AJ, Meijer CJ, Snijders PJ. The use of general primers GP5 and GP6 elongated at their 3′ ends with adjacent highly conserved sequences improves human papillomavirus detection by PCR. J Gen Virol. 1995;76(Pt 4):1057–62. doi: 10.1099/0022-1317-76-4-1057. [DOI] [PubMed] [Google Scholar]
- 25.Sotlar K, Diemer D, Dethleffs A, Hack Y, Stubner A, Vollmer N, et al. Detection and typing of human papillomavirus by e6 nested multiplex PCR. J Clin Microbiol. 2004;42:3176–84. doi: 10.1128/JCM.42.7.3176-3184.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kreimer AR, Clifford GM, Boyle P, Franceschi S. Human papillomavirus types in head and neck squamous cell carcinomas worldwide: a systematic review. Cancer Epidemiol Biomarkers Prev. 2005;14:467–75. doi: 10.1158/1055-9965.EPI-04-0551. [DOI] [PubMed] [Google Scholar]
- 27.Zoncu R, Efeyan A, Sabatini DM. mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol. 2011;12:21–35. doi: 10.1038/nrm3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lyons JG, Patel V, Roue NC, Fok SY, Soon LL, Halliday GM, et al. Snail up-regulates proinflammatory mediators and inhibits differentiation in oral keratinocytes. Cancer Res. 2008;68:4525–30. doi: 10.1158/1078-0432.CCR-07-6735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Amornphimoltham P, Patel V, Leelahavanichkul K, Abraham RT, Gutkind JS. A retroinhibition approach reveals a tumor cell-autonomous response to rapamycin in head and neck cancer. Cancer Res. 2008;68:1144–53. doi: 10.1158/0008-5472.CAN-07-1756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gillison ML. Current topics in the epidemiology of oral cavity and oropharyngeal cancers. Head Neck. 2007;29:779–92. doi: 10.1002/hed.20573. [DOI] [PubMed] [Google Scholar]
- 31.Downs L. Advances in cervical cancer treatment. Gynecol Oncol. 2011;121:431–3. doi: 10.1016/j.ygyno.2011.04.010. [DOI] [PubMed] [Google Scholar]
- 32.Landoni F, Maneo A, Colombo A, Placa F, Milani R, Perego P, et al. Randomised study of radical surgery versus radiotherapy for stage Ib-IIa cervical cancer. Lancet. 1997;350:535–40. doi: 10.1016/S0140-6736(97)02250-2. [DOI] [PubMed] [Google Scholar]
- 33.Serrano M, Hannon GJ, Beach D. A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature. 1993;366:704–7. doi: 10.1038/366704a0. [DOI] [PubMed] [Google Scholar]
- 34.Dyson N, Howley PM, Munger K, Harlow E. The human papilloma virus-16 E7 oncoprotein is able to bind to the retinoblastoma gene product. Science. 1989;243:934–7. doi: 10.1126/science.2537532. [DOI] [PubMed] [Google Scholar]
- 35.Lewis JS, Jr, Thorstad WL, Chernock RD, Haughey BH, Yip JH, Zhang Q, et al. p16 positive oropharyngeal squamous cell carcinoma:an entity with a favorable prognosis regardless of tumor HPV status. Am J Surg Pathol. 2010;34:1088–96. doi: 10.1097/PAS.0b013e3181e84652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.McLaughlin-Drubin ME, Crum CP, Munger K. Human papillomavirus E7 oncoprotein induces KDM6A and KDM6B histone demethylase expression and causes epigenetic reprogramming. Proc Natl Acad Sci U S A. 2011;108:2130–5. doi: 10.1073/pnas.1009933108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Poetsch M, Hemmerich M, Kakies C, Kleist B, Wolf E, Dorp F, et al. Alterations in the tumor suppressor gene p16 INK4A are associated with aggressive behavior of penile carcinomas. Virchows Archiv. 2010;458:221–9. doi: 10.1007/s00428-010-1007-4. [DOI] [PubMed] [Google Scholar]
- 38.Lu Z, Hu X, Li Y, Zheng L, Zhou Y, Jiang H, et al. Human papillomavirus 16 E6 oncoprotein interferences with insulin signaling pathway by binding to tuberin. J Biol Chem. 2004;279:35664–70. doi: 10.1074/jbc.M403385200. [DOI] [PubMed] [Google Scholar]
- 39.Spangle JM, Munger K. The human papillomavirus type 16 E6 oncoprotein activates mTORC1 signaling and increases protein synthesis. J Virol. 2010;84:9398–407. doi: 10.1128/JVI.00974-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Boudeau J, Baas AF, Deak M, Morrice NA, Kieloch A, Schutkowski M, et al. MO25alpha/beta interact with STRADalpha/beta enhancing their ability to bind, activate and localize LKB1 in the cytoplasm. EMBO J. 2003;22:5102–14. doi: 10.1093/emboj/cdg490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Corradetti MN, Inoki K, Bardeesy N, DePinho RA, Guan KL. Regulation of the TSC pathway by LKB1: evidence of a molecular link between tuberous sclerosis complex and Peutz-Jeghers syndrome. Genes Dev. 2004;18:1533–8. doi: 10.1101/gad.1199104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Molinolo AA, Amornphimoltham P, Squarize CH, Castilho RM, Patel V, Gutkind JS. Dysregulated molecular networks in head and neck carcinogenesis. Oral Oncol. 2009;45:324–34. doi: 10.1016/j.oraloncology.2008.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Feng W, Duan X, Liu J, Xiao J, Brown RE. Morphoproteomic evidence of constitutively activated and overexpressed mTOR pathway in cervical squamous carcinoma and high grade squamous intraepithelial lesions. Int J Clin Exp Pathol. 2009;2:249–60. [PMC free article] [PubMed] [Google Scholar]
- 44.Jimeno A, Rudek MA, Kulesza P, Ma WW, Wheelhouse J, Howard A, et al. Pharmacodynamic-guided modified continuous reassessment method-based, dose-finding study of rapamycin in adult patients with solid tumors. J Clin Oncol. 2008;26:4172–9. doi: 10.1200/JCO.2008.16.2347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cloughesy TF, Yoshimoto K, Nghiemphu P, Brown K, Dang J, Zhu S, et al. Antitumor activity of rapamycin in a Phase I trial for patients with recurrent PTEN-deficient glioblastoma. PLoS medicine. 2008;5:e8. doi: 10.1371/journal.pmed.0050008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ekshyyan O, Rong Y, Rong X, Pattani KM, Abreo F, Caldito G, et al. Comparison of radiosensitizing effects of the mammalian target of rapamycin inhibitor CCI-779 to cisplatin in experimental models of head and neck squamous cell carcinoma. Mol Cancer Ther. 2009;8:2255–65. doi: 10.1158/1535-7163.MCT-08-1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ekshyyan O, Mills GM, Lian T, Amirghahari N, Rong X, Lowery-Nordberg M, et al. Pharmacodynamic evaluation of temsirolimus in patients with newly diagnosed advanced-stage head and neck squamous cell carcinoma. Head Neck. 2010;32:1619–28. doi: 10.1002/hed.21374. [DOI] [PubMed] [Google Scholar]
- 48.Sarbassov DD, Ali SM, Sengupta S, Sheen JH, Hsu PP, Bagley AF, et al. Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol Cell. 2006;22:159–68. doi: 10.1016/j.molcel.2006.03.029. [DOI] [PubMed] [Google Scholar]
- 49.Soefje SA, Karnad A, Brenner AJ. Common toxicities of mammalian target of rapamycin inhibitors. Target Oncol. 2011;6:125–9. doi: 10.1007/s11523-011-0174-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.