Abstract
A loss of neurons is observed in the hippocampus of many patients with epilepsies of temporal lobe origin. It has been hypothesized that damage limitation or repair, for example using neurotrophic factors (NTFs), may prevent the transformation of a normal tissue into epileptic (epileptogenesis). Here, we used viral vectors to locally supplement two NTFs, fibroblast growth factor–2 (FGF-2) and brain-derived neurotrophic factor (BDNF), when epileptogenic damage was already in place. These vectors were first characterized in vitro, where they increased proliferation of neural progenitors and favored their differentiation into neurons, and they were then tested in a model of status epilepticus-induced neurodegeneration and epileptogenesis. When injected in a lesioned hippocampus, FGF-2/BDNF expressing vectors increased neuronogenesis, embanked neuronal damage, and reduced epileptogenesis. It is concluded that reduction of damage reduces epileptogenesis and that supplementing specific NTFs in lesion areas represents a new approach to the therapy of neuronal damage and of its consequences.
Keywords: epilepsy, gene therapy, neurotrophic factors
Epilepsies originating in the temporal lobe of the brain are the most common in adults. In patients with these diseases, focal pathological abnormalities can be observed, the most prominent of which is a loss of neurons in the hippocampus termed hippocampal sclerosis. These abnormalities develop in a previously healthy tissue, often after an initial “epileptogenic” event that can produce damage (for example, an episode of prolonged, uncontrolled seizures know as status epilepticus [SE]). After a latent period of weeks to years, epileptogenic events my be followed by the occurrence of spontaneous recurrent seizures (SRS), i.e., epilepsy (1). Unfortunately, currently available antiseizure drugs do not prevent this process.
It can be hypothesized that embanking or repairing damage may lead to antiepileptic effects (1). To date, however, the treatment of diseases associated with neuronal death has been restricted to attempts to prevent or limit the damage. More recently, the discovery of neural stem cells disclosed two new approaches: the transplantation of stem cells and the recruitment of endogenous stem cells for generating new neurons by means of extracellular proliferation/differentiation factors (2). In view of all approaches, key extracellular regulators of neuron survival and of stem cell proliferation and differentiation into neurons are the neurotrophic factors (NTFs) (3).
Endogenous NTFs are actually produced after an epileptogenic insult, but they remain insufficient or inadequate for neuroprotection and/or for providing the endogenous stem cells with the proper cues to proliferate, differentiate into neurons, and restore function (4, 5). In fact, damage ensues and a reactive neurogenesis occurs but fails to repair it (1). The aim of this study was to attempt suppressing neuronal damage and preventing the development of epilepsy by means of a local supplementation of specific NTFs within the lesioned, epileptogenic tissue.
To pursue this aim, we generated a viral vector expressing a combination of fibroblast growth factor–2 (FGF-2) and brain-derived neurotrophic factor (BDNF). We chose this combination because: (i) both these NTFs have neuroprotective properties; (ii) FGF-2 potently induces proliferation of hippocampal progenitors (6, 7); (iii) BDNF is required for neuronogenesis in the hippocampus (8–10). We used replication-defective herpes simplex virus–1 (HSV-1) vectors, for multiple reasons (11): they efficiently infect nonreplicating cells such as neurons; they can accommodate large inserts; they do not integrate their genome into the host DNA (that is, there is no mutagenesis risk); they can be transported retrogradely in neurons (therefore, transgene expression can occur in remote areas through nerve terminals afferent to the injection area); and finally, they produce a transient transgene expression: It is desirable to obtain transient expression of NTFs, because NTFs can trigger plastic changes that remain detectable when they are no longer expressed, whereas their long-term expression may be detrimental for brain function (12).
Results and Discussion
Vectors.
A schematic representation of the vectors we used is shown in supporting information (SI) Fig. S1. For FGF-2, we used a previously characterized vector, TH-FGF2 (13), in which the transgene was under the control of the human cytomegalovirus (HCMV) immediate-early (IE) promoter. For BDNF, we used a vector, T0-BDNF, in which the transgene was under the control of the ICP0 promoter. The double mutant (TH-FGF2/0-BDNF) was obtained by crossing over TH-FGF2 and T0-BDNF. Promoters were chosen to promote a synergy between the NTFs, making use first of the FGF-2 property of potently inducing proliferation of hippocampal progenitors and then of the BDNF property of inducing their neuronal differentiation: the HCMV IE promoter driving FGF-2 is known to ensure robust, but transient, transgene expression, whereas the ICP0 promoter driving BDNF provides a longer-lasting expression (14).
The vector-induced transgene expression and the biological activity of the NTF proteins produced by infected cells were first studied in vitro. The data we obtained (SI Text “In vitro Characterization” and Fig. S2) indicate that: (i) vectors induce production and release of FGF-2 and BDNF at biologically active levels; and (ii) FGF-2 and BDNF supplied together, not individually, potently favor survival, proliferation, and neuronal differentiation of neural progenitors. Therefore, we elected to test the vector expressing FGF-2 and BDNF together in vivo.
The efficiency of infection and the time-course of transgene expression in vivo was examined after a single stereotaxical inoculation of the vector in the hippocampus of adult rats (SI Text “Transgene Expression in Vivo” and Fig. S3). The data show that inoculation of TH-FGF2/0-BDNF provides a bilateral, short-term (≈1 week) increase in FGF-2 expression accompanied and followed by a bilateral, slightly longer-lasting (at least 11 days) increase in BDNF expression, and that these events are not associated with significant toxicity.
Effects on SE-Induced Cell Death and Neurogenesis.
We tested TH-FGF2/0-BDNF on the pilocarpine SE–induced neurodegeneration and epileptogenesis. In this epilepsy model, an episode of SE produces intense neuronal damage and, after a latent period of 2–3 weeks, SRSs, i.e., epilepsy (15).
In the present experimental series, pilocarpine (300 mg/kg i.p.) rapidly induced a robust convulsive SE (latency, 15 ± 2 minutes) that was interrupted after 2 hours through administration of the anticonvulsant diazepam (10 mg/kg i.p.). Based on behavioral observation and EEG recordings, the severity of SE in the different animals was indistinguishable. This procedure caused damage in several brain areas: in particular (16, 17), hippocampal damage closely resembled human hippocampal sclerosis and was invariably remarkable. Typically, 3 days after pilocarpine administration, Fluoro-Jade C (FJC) staining allowed identification of several degenerating cells in CA1, CA3, and the hilus of the dentate gyrus. A pronounced edema with cell loss was also evident. A three-dimensional structural reconstruction demonstrated a 27.8 ± 3.1% reduction of hippocampal volume 3 days after pilocarpine administration, as compared with the response on control animals (P < 0.001, Mann-Whitney U test for unpaired data).
Three days after SE, these lesioned animals were randomly assigned to three groups: one group was injected in one hippocampus with the vector expressing FGF-2 and BDNF; the second group was injected with a control vector; and the third group was not treated at all (no difference was observed between these latter two groups in any of the parameters examined, and therefore they have been pooled together for statistical analysis, and collectively termed “control”). To explore the mitogenic effect of the vectors, animals were treated with bromo-deoxyuridine (BrdU, four 50-mg/kg i.p. injections, one every 2 hours) 6 days after SE (i.e., 3 days after vector inoculation, Fig. S4). This time point was chosen because it is both within the peak of SE-induced proliferation of neural stem cells (18) and of vector-induced overexpression of FGF-2 (see above). A subset of animals in each group was killed 14 days after SE (i.e., 11 days after vector inoculation) to examine possible effects on cell survival and neurogenesis, and another subset was killed 28 days after SE (25 days after vector inoculation) to examine tissue morphology. These latter animals also underwent 20 days behavioral and EEG monitoring, to examine the outcome of the treatment (Fig. S4).
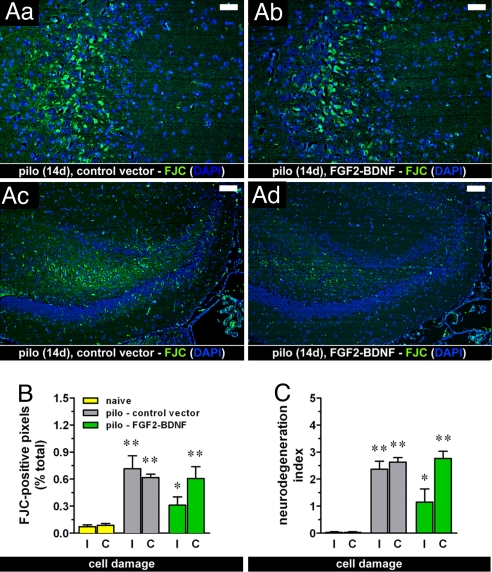
Although the bulk of SE-induced damage is in the first few days and, thus, was already in place when we injected the vector, several degenerating cells were still observed 14 days after pilocarpine administration in different hippocampal subfields (CA1, CA3, hilus of the dentate gyrus; Fig. 1Aa and Ac). Administration of the vector expressing FGF-2 and BDNF did not alter this ongoing cell loss, except for a slight, nonsignificant attenuation in the injected hippocampus (Fig. 1), indicating that, in vivo, the neuroprotective effect of this vector is limited or may require more prolonged or higher-level transgene expression.
Fig. 1.
(A) Effect of the treatment with the vector expressing FGF-2 and BDNF together (TH-FGF2/0-BDNF) on SE-induced ongoing damage. Degenerating cells are marked with FJC (green); nuclei marked by DAPI (blue). Fourteen days after pilocarpine (pilo)–induced SE (14d), ongoing hippocampal damage was still detectable in animals injected with the control vector (control), especially in the CA3 area (Aa) and in the dentate (Ac). This pattern was identical to the pattern observed in untreated animals (not shown). In animals treated with TH-FGF2/0-BDNF (FGF2-BDNF), a reduction, but not the abolishment of damage was observed (Ab and Ad). Horizontal bars, 25 μm in (Aa) and (Ab) and 50 μm in (Ac) and (Ad). (B, C) Quantification of neurodegeneration. Shown is the percentage of FJC-positive pixels in the hippocampus (B) and the degeneration index (C), calculated as described in Materials and Methods, in naïve rats (yellow bars) and pilocarpine-treated rats, injected with either the control vector (gray bars) or TH-FGF2/0-BDNF (green bars). I, injected (ipsilateral) hippocampus; C, noninjected (contralateral) hippocampus. Data are means ± SE for seven to eight animals per group. *P < 0.05, **P < 0.01 vs. naïve; Kruskal-Wallis test. No significant difference was observed between pilo – control vector and pilo – FGF2-BDNF.
In contrast to the limited effect on cell death, the effect of the vector expressing FGF-2 and BDNF on neurogenesis was quite remarkable. In the adult hippocampus, neural progenitors are found both in the subgranular zone (SGZ) and in the caudal subventricular zone (SVZ). SE has been reported to increase proliferation of hippocampal progenitors in both areas, but SGZ progenitors differentiate into neurons that, in large part, migrate ectopically to the hilus and SVZ progenitors differentiate into glial, not neuronal, cells: that is, SE-induced neurogenesis is aberrant and may contribute to epileptogenesis (19, 20).
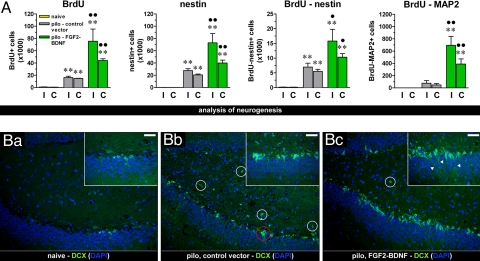
We used pulse-chase BrdU labeling and immunohistochemistry for markers of progenitors to determine the mitogenic and differentiative activity of the NTFs supplemented by the vectors. To explore the mitogenic effect, we counted BrdU-positive cells in the dorsal hippocampus 14 days after pilocarpine. As expected, SE per se induced a significant increase in the number of mitotically active cells (Fig. 2 and Fig. S5). In keeping with previous reports (6, 18, 20), this increase was observed in the dentate gyrus and in the pyramidal gyrus (especially CA1 strata oriens and lacunosum-molecularis, near the SVZ). Inoculation of the control vector did not alter this pattern. In contrast, inoculation of the vector expressing FGF-2 and BDNF produced a further, bilateral, highly significant increase in the number of BrdU-positive cells (Fig. 2A), indicating proliferation of progenitors. Notably, an increased number of positive cells was also observed in the ventral hippocampus (Fig. S6).
Fig. 2.
Effect of the vector expressing FGF-2 and BDNF together on cell proliferation. (A) Average number of BrdU-positive, nestin-positive, double-labeled BrdU-positive and nestin-positive, double-labeled BrdU-positive and MAP2abc-positive cells in the dorsal hippocampus of naïve rats (yellow bars) and of pilocarpine-treated rats 11 days after inoculation of control vector (pilo – control vector, gray bars) or of the vector expressing FGF-2 and BDNF (pilo – FGF2-BDNF, green bars) in the right, ipsilateral to inoculation (I) and in the left, contralateral (C) hippocampus. Data are the means ± SE of four to five animals per group. ** P < 0.01 vs. naïve; ● P < 0.05, ●● P < 0.01 vs. pilo – control vector; analysis of variance (ANOVA) and post hoc Newman-Keuls test. (B) The vector expressing FGF-2 and BDNF together induced proliferation of DCX-positive cells in vivo. Immunofluorescence (dentate gyrus region) for DCX (green) in control (naïve) rats (Ba), in pilocarpine-treated rats given the control vector (Bb) and in pilocarpine-treated rats given the vector expressing FGF-2 and BDNF (Bc). Nuclei are marked by DAPI (blue). It should be noted that DCX-positive cells in the naïve animal dentate gyrus area are located in the subgranular zone and present detectable elongations projecting across the granular layer (Ba Inset); pilocarpine-induced SE (both in untreated animals and in animals administered the control vector) caused an increase in DCX-positive cells (Bb), which tended to produce relatively fewer elongations (Bb, inset), to group into clusters (example in red circle), and to localize ectopically (white circles). Treatment with the double mutant (Bc) further increased the number of DCX-positive cells while reducing their aberrant features, i.e., lower numbers of ectopic cells and presence of numerous elongations across the granular layer (arrowheads, Bc, inset). Horizontal bar, 25 μm.
In both the SGZ and SVZ, a series of differentiation steps have been described: from early, glia-like progenitors (termed type-1 for SGZ and type-B for SVZ), to transiently amplifying cells (type-2 and type-C), to late, migrating cells (type-3 and type-A) (21, 22). Type-1 and type-B cells are positive to GFAP, nestin, and Sry-related HMG box transcription factor (Sox2); type-2 and type-B cells to nestin and Sox2, but not to GFAP; type-3 and type-A cells to doublecortin (DCX) (22, 23). Pilocarpine SE per se significantly increased the number of nestin-positive cells in the hippocampus as compared with naïve controls, but treatment with TH-FGF2/0-BDNF dramatically potentiated this effect, more markedly in the injected hippocampus (Fig. 2A). Many (≈20%) of these nestin-positive cells were also BrdU-positive, indicating that part of them were actively proliferating at the time of BrdU administration (Fig. 2A). Furthermore, a subset of these cells was also GFAP-positive (Fig. S5B), an indication of the presence of early progenitors. Finally, we observed an increased number of Sox2-positive cells in pilocarpine animals treated with TH-FGF2/0-BDNF, 7 days after SE (Fig. S5D). These data indicate that SE per se causes proliferation of hippocampal early to intermediate progenitors and that vector-mediated supplementation of FGF-2 and BDNF greatly increases this phenomenon.
In the subsequent steps of neurogenesis, progenitors begin to migrate and become DCX-positive. As stated above, this migration is partly aberrant after SE, because many SGZ progenitors do not head toward the granular layer but toward the hilus (19). Therefore, we also analyzed DCX-positive cells. Under control conditions (naïve animals), a limited number of DCX cells, with elongations projecting across the granular layer, was found in the SGZ (Fig. 2Ba) and even less so in the SVZ (essentially, just a few, weakly labeled DCX-positive cells in the CA1 stratum oriens). SE per se caused a robust increase in the number of DCX-positive cells in the dentate gyrus (Fig. 2Bb). However, these cells did not often produce elongations, tended to aggregate into clusters, and were often ectopically located (Fig. 2Bb). These observations are consistent with the aberrant aspects of SE-induced neurogenesis (19, 24). A slightly increased number of DCX-positive cells was also observed in the pyramidal gyrus (CA3 and CA1). This pattern was not modified by the administration of the control vector. In contrast, administration of TH-FGF2/0-BDNF in pilocarpine-treated animals caused a greater increase in the number of DCX-positive cells compared with pilocarpine alone or pilocarpine and control vector (Fig. 2B). Moreover, it increased the length and number of elongations, reduced cluster formation, and reduced the number of ectopic cells (Fig. 2Bc). An increased number of DCX-positive cells was also observed in the pyramidal gyrus, indicating commitment to neuronal differentiation (Fig. S5E).
Finally, we checked whether, consistent with our in vitro data, a subset of these new cells were differentiating into neurons and migrating into the appropriate location to possibly restore function. Thus, we performed double-label immunofluorescence with BrdU and MAP2abc, a marker of immature and mature neurons. SE per se did not significantly increase the number of BrdU-MAP2–positive cells; in contrast, we found several double-labeled cells in the hippocampus of pilocarpine animals treated with the double mutant (Fig. 2A).
These data suggest that injection of the vector expressing FGF-2 and BDNF in a SE-damaged hippocampus favors proliferation of early progenitors according to a pattern that, at variance with what was observed in untreated animals, appears to follow the physiological pattern more faithfully, leading to the production of cells that enter the neuronal lineage of differentiation and reducing the aberrant aspects of SE-induced neurogenesis.
Outcome: Pathology and Behavior.
To evaluate the outcome of the treatment, we compared the different experimental groups for the morphology of the hippocampus. First: compared with naïve controls, the dorsal hippocampus of pilocarpine animals remained smaller 4 weeks after SE (approximately −30%, like 3 days after SE: see above). Treatment with the double mutant produced a significant increase in hippocampal volume, with a recovery of approximately a third of the loss (Fig. S7A).
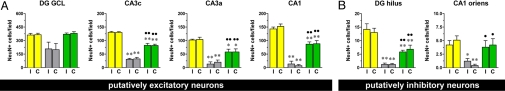
Importantly, the SE-damaged hippocampus presents a clearly reduced density of neuronal cells, most notably hilar and pyramidal cells, that was not observed in animals treated with the double mutant (Fig. S7). We measured the density of astrocytes and neurons in the hippocampus using GFAP and NeuN immunofluorescence. The density of GFAP-positive cells in the entire hippocampus was significantly increased in pilocarpine-treated animals compared with naïve controls, an indication of reactive astrocytosis (25). In the pilocarpine group of animals treated with the vector expressing FGF-2 and BDNF, the density of GFAP-positive cells decreased slightly in comparison with untreated animals, but was still greater than in naïve animals (Fig. S7B). Neuronal density was measured using NeuN. Putatively excitatory dentate gyrus granule cells were not significantly decreased by SE and treatment with the double mutant did not alter the situation (Fig. 3A). In contrast, putatively excitatory pyramidal neurons were dramatically decreased in animals that experienced SE, and the double mutant significantly increased their number (Fig. 3A). Putatively inhibitory neurons in the dentate hilus and in the stratum oriens of CA1 were even more dramatically decreased in animals that experienced SE; and, again, TH-FGF2/0-BDNF significantly improved the situation (Fig. 3B).
Fig. 3.
Neuropathological outcome of the treatment with the vector expressing FGF-2 and BDNF (TH-FGF2/0-BDNF), 28 days after pilocarpine-induced status epilepticus. (A) Measure of the density of NeuN-positive, presumably excitatory neurons in selected subareas of the dorsal hippocampus: dentate granule layer (DG GCL), CA3c pyramidal layer (CA3c), CA3a pyramidal layer (CA3a), and CA1 pyramidal layer (CA1). I, injected (ipsilateral) hippocampus; C, noninjected (contralateral) hippocampus. (B) Measure of the density of NeuN-positive, presumably inhibitory neurons in selected subareas of the dorsal hippocampus: hilus of the dentate gyrus (DG hilus) and stratum oriens of CA1 (CA1 oriens). Data are the means ± SE of 10–12 animals per group. *P < 0.05, **P < 0.01 vs. naïve; ● P < 0.05; ●● P < 0.01 vs. pilo – control vector; ANOVA and post hoc Newman-Keuls test.
These data suggest that treatment with the vector expressing FGF-2 and BDNF partially heals the damage induced by SE, maintaining a good ratio between excitation and inhibition in the circuit. However, some inhibitory cells are present in the pyramidal layer and excitatory cells are present in the hilus (for example, the glutamateric mossy cells). Thus, we deepened the investigation analyzing some of the main populations of GABAergic interneurons, namely parvalbumin, somatostatin, and calbindin. Parvalbumin-containing interneurons exert perisomatic, whereas somatostatin and calbindin exert dendritic inhibition; calbindin also labels a subpopulation of principal, glutamatergic cells, both in the pyramidal and in the granular layer (26). In keeping with previous reports (27–29), we found that the number of parvalbumin-positive cells in epileptic animals was similar to that in controls (Fig. S8A), while that of somatostatin-positive cells was halved (Fig. S8B). Also, calbindin-positive principal neurons in the pyramidal and granular layer, but not calbindin-positive interneurons in the strata oriens and radiatum and in the dentate gyrus hilus, were significantly decreased in the pilocarpine group (Fig. S8C) (30, 31). Treatment with TH-FGF2/0-BDNF effectively corrected these cell losses (Fig. S8).
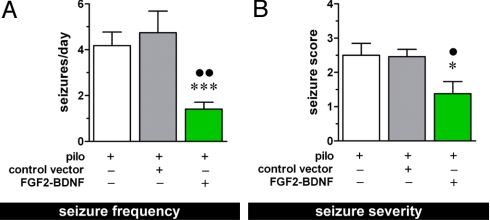
Avoiding the loss of somatostatin cells may imply maintenance of dendritic inhibition and, therefore, decreased susceptibility to seizures. This leads to the key question: do these beneficial effects ameliorate the outcome in terms of SRSs? We performed behavioral and EEG analyses: animals were video-EEG monitored for 20 days and the occurrence, severity, and duration of SRSs were recorded. As expected, all non vector-injected pilocarpine-treated rats exhibited SRSs beginning ≈2 weeks after SE. On average, we observed in this group four seizures per day (Fig. 4A) lasting about 1 minute each and with a mean severity score (32) of 2.5 (Fig. 4B). Administration of the control vector did not modify this pattern. In contrast, rats treated with the double mutant displayed a highly significant improvement: (i) a subset of animals (two of 11) never developed SRSs during the time frame of observation; (ii) the average number of seizures per day in this group was highly significantly reduced compared with the other two groups (Fig. 4A); (iii) the seizure severity was also significantly decreased (Fig. 4B). Thus, the FGF-2 and BDNF-expressing vector, injected in the hippocampus after the establishment of hippocampal sclerosis, provided both significant recovery from damage and significant reduction of SRSs. These results are striking, considering that this epilepsy model is accompanied by extensive damage also in extra-hippocampal regions (33), such that only a subset of SRSs may actually originate in the hippocampus (34), i.e., where we documented favorable effects by the vector. In other words, SRSs originating in the hippocampus may be essentially abolished by vector treatment.
Fig. 4.
EEG and behavioral analysis. Average frequency (A) and severity (B) of spontaneous seizures in the chronic period (14–28 days after pilocarpine-induced SE). Data are the means ± SE for 10–12 animals per group. *P < 0.05 and ***P < 0.001 vs. pilo; ● P < 0.05 and ●● P < 0.01 vs. pilo – control vector; ANOVA and post hoc Newman-Keuls test for (A), Kruskal-Wallis test for (B).
Finally, to test whether the vector has a truly antiepileptogenic effect and is not merely dampening SRSs (i.e., blocking ictogenesis), we investigated whether the vector expressing FGF-2 and BDNF was effective in controlling seizures when injected in animals that were already experiencing SRSs. To address this question, a group of pilocarpine-treated rats was video-EEG monitored beginning 2 weeks after SE. Three weeks after the first spontaneous seizure, these animals were injected either with the control vector or with TH-FGF2/0-BDNF, and monitoring was continued for 4 more weeks. The frequency, duration, and severity of SRSs were then comparatively analyzed in the 2 weeks preceding vector inoculation and in the third and fourth weeks after inoculation. Inoculation of the FGF-2/BDNF expressing vector did not affect any of the parameters examined (Fig. S9), indicating that supplementation of FGF-2 and BDNF can interfere with epileptogenesis but not with ictogenesis.
In conclusion, the main finding of this study is that supplementation of FGF-2 and BDNF in the hippocampus lesioned by prolonged seizures effectively limits or repairs damage, maintaining a good ratio between excitation and inhibition, and significantly improves the natural history of the disease.
Understanding in depth the mechanistic basis of these effects will require further studies, but the present data provide some initial insights. FGF-2 and BDNF may provide protection against damage or gain of function of partially damaged neurons. Even if the present study did not provide support to this idea, subtle effects on specific cell populations that could not be detected with the techniques that we used cannot be excluded. Nonetheless, another hypothesis, namely, that FGF-2 and BDNF exert effects on neural stem cells and progenitors to generate new neurons, seems more plausible. The phenotype(s) of these new neurons remain uncertain, but we identified one interesting candidate in a subtype of interneurons (somatostatin). GABA-somatostatin interneurons play an important role in dendritic inhibition, and their loss may favor the development of epilepsy (28); thus, the replacement of these neurons may explain the favorable effects of FGF-2 and BDNF supplementation.
Some aspects of the present findings are worthy of note. First, virus injection was performed under conditions compatible with the clinical settings of patient observation (after the epileptogenic insult), conditions that reproduce those that may allow therapeutic intervention. Second, the disease-modifying effect is striking and can be interpreted as anti-epileptogenic, an effect that is not achieved by the currently available therapy. Further studies will be required in both respects, to establish how long the effect lasts and to challenge the hypothesis that it is truly anti-epileptogenic. Last but not least, these findings may have a heuristic value for the many other neurological diseases associated with neuronal damage.
Materials and Methods
A detailed description of all of the methods used in this study is provided in the SI Text “Materials and Methods.”
Animals.
Male Sprague-Dawley rats (240–260 g; Harlan Italy) were used for experiments. All procedures were carried out in accordance with guidelines by the European Community and national laws and policies.
Hippocampal Volumetry.
Paraffin-embedded brains were cut in successive 10 μm sections across the entire hippocampus and one of every five sections was stained with hematoxylin and eosin. Sections were scanned and digitalized using a Nikon Coolscope. Segmentation and orientation were obtained by Amira TM 4.0 (Mercury Computer Systems), by two researchers blind to the animal groups. Three-dimensional reconstructions were then calculated and averaged using the same software.
HSV Infusion.
Under ketamine and xylazine anesthesia, a glass needle connected to a perfusion pump was implanted in the dorsal hippocampus. A total of 1.6 × 106 pfu of vector were injected in a volume of 2 μl at a flow rate of 0.1 μl/min.
FJC Staining.
FJC staining was performed on 6-μm paraffin-embedded sections as previously described (35).
Immunohistochemistry and Immunofluorescence.
For BrdU immunostaining, cellular DNA was denatured with HCl. The primary antibodies used for immunohistochemistry were as follows: BrdU mouse monoclonal (1:100, Roche Molecular Biochemicals); nestin mouse monoclonal (1:100; Chemicon); MAP2abc mouse monoclonal (1:100; Chemicon); GFAP rabbit polyclonal (1:100; Sigma); parvalbumin mouse monoclonal (1:50; Swant); and calbindin rabbit polyclonal (1:50; Swant). Detection was obtained using the biotin-streptavidin system (Ultra Vision Detection System, Lab Vision Corporation). The reaction product was detected using a 3,3-diamino-benzidinetetrahydrochloride (DAB) substrate kit for peroxidase (Vector Laboratories).
For immunofluorescence, the primary antibodies were: BrdU (rat monoclonal, AbD Serotec) and nestin (mouse monoclonal, Chemicon) 1:10 and 1:10 respectively; BrdU (as above) and MAP2abc (mouse monoclonal, Immunological Sciences) 1:10 and 1:10; nestin (mouse monoclonal, Chemicon) and GFAP (rabbit polyclonal, Sigma) 1:25 and 1:100; NeuN (mouse monoclonal, Chemicon) and GFAP (as above) 1:25 and 1:100; MAP2abc (mouse monoclonal, Immunological Sciences) and GFAP (as above) 1:25 and 1:100; Sox2 (rabbit polyclonal, Immunological Sciences) and GFAP (mouse monoclonal, Immunological Sciences) 1:25 and 1:100; DCX (rabbit polyclonal, Cell Signaling) and nestin (as above) 1:20 and 1:20; DCX (goat polyclonal, Santa Cruz Biotechnology) 1:25; somatostatin-14 (rabbit polyclonal, Peninsula Laboratories) 1:25.
Telemetry.
Seizure onset, severity, and duration were assessed by video-EEG monitoring of the animals, performed by means of Phenotyper cages (Noldus Information Technology), using telemetric technology (Dataquest A.R.T. Data Acquisition, Data Sciences International).
Quantification and Statistical Analysis.
All experiments were performed strictly under double-blind conditions. The volume of the dorsal hippocampus was calculated using a stereological approach based on the principle of Cavalieri (36). The degree of cell damage was quantified using two approaches: the “neurodegeneration score” (37) and a thresholding of digital images (38). Counting of cells positive for the different markers was conducted on one of every 26 sections across the entire dorsal hippocampus (i.e., nine regularly spaced sections per animal). Quantification of GFAP-positive and NeuN-positive cells was performed in frames taken from the various hippocampal subareas. Behavioral alterations and seizure scoring were performed by means of the Observer (Noldus). Seizure detection (frequency, duration and severity), as well as single ictal events (<2 seconds) were scored for each individual rat for the whole analysis period (24 h/day for 20 consecutive days).
Supplementary Material
Acknowledgments.
The authors are grateful to O. Lindvall (University of Lund, Sweden) and L. Beani (University of Ferrara, Italy) for their thoughtful comments on the manuscript. We also thank A. Sbarbati for the support and scientific advice on hippocampal volumetry, telemetry and video-EEG, and M. Barbieri, D. Nardo, and F. Mora for technical support. M.M. was supported by a fellowship of the foundation “Cassa di Risparmio di Ferrara.” This work was supported by grants from the University of Ferrara (STAMINA, to M.S.), the Italian Ministry for the University and Scientific Research (Prin 2005 and 2007 to M.S.; Prin 2005 to R.M.; Prin 2006 to A.M.), the Italian National Institute of Health (Program Stem Cells, CS 126.1, to PM; ISS-2006–533F/C/3 to R.M.), and the European Community (EU Research Grants LSHB-CT-2005–018649 [THOVLEN] to R.M. and LSH-CT-2006–037315 [EPICURE], thematic priority LIFESCIHEALTH, to M.S. and P.F.F.).
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/cgi/content/full/0810710106/DCSupplemental.
References
- 1.Pitkanen A, Sutula TP. Is epilepsy a progressive disorder? Prospects for new therapeutic approaches in temporal-lobe epilepsy. Lancet Neurol. 2002;1:173–181. doi: 10.1016/s1474-4422(02)00073-x. [DOI] [PubMed] [Google Scholar]
- 2.Lindvall O, Kokaia Z. Stem cells for the treatment of neurological disorders. Nature. 2006;441:1094–1096. doi: 10.1038/nature04960. [DOI] [PubMed] [Google Scholar]
- 3.Hagg T. Molecular regulation of adult CNS neurogenesis: An integrated view. Trends Neurosci. 2005;28:589–595. doi: 10.1016/j.tins.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 4.Simonato M, Tongiorgi E, Kokaia M. Angels and demons: Neurotrophic factors and epilepsy. Trends Pharmacol Sci. 2006;27:631–638. doi: 10.1016/j.tips.2006.10.002. [DOI] [PubMed] [Google Scholar]
- 5.Kempermann G. They are not too excited: The possible role of adult-born neurons in epilepsy. Neuron. 2006;52:935–937. doi: 10.1016/j.neuron.2006.12.004. [DOI] [PubMed] [Google Scholar]
- 6.Becq H, Jorquera I, Ben-Ari Y, Weiss S, Represa A. Differential properties of dentate gyrus and CA1 neural precursors. J Neurobiol. 2005;62:243–261. doi: 10.1002/neu.20089. [DOI] [PubMed] [Google Scholar]
- 7.Nakatomi H, et al. Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous neural progenitors. Cell. 2002;110:429–441. doi: 10.1016/s0092-8674(02)00862-0. [DOI] [PubMed] [Google Scholar]
- 8.Lee J, Duan W, Mattson MP. Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J Neurochem. 2002;82:1367–1375. doi: 10.1046/j.1471-4159.2002.01085.x. [DOI] [PubMed] [Google Scholar]
- 9.Scharfman H, et al. Increased neurogenesis and the ectopic granule cells after intrahippocampal BDNF infusion in adult rats. Exp Neurol. 2005;192:348–356. doi: 10.1016/j.expneurol.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 10.Young KM, Merson TD, Sotthibundhu A, Coulson EJ, Bartlett PF. p75 neurotrophin receptor expression defines a population of BDNF-responsive neurogenic precursor cells. J Neurosci. 2007;27:5146–5155. doi: 10.1523/JNEUROSCI.0654-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Simonato M, Manservigi R, Marconi P, Glorioso J. Gene transfer into neurones for the molecular analysis of behaviour: Focus on herpes simplex vectors. Trends Neurosci. 2000;23:183–190. doi: 10.1016/s0166-2236(99)01539-8. [DOI] [PubMed] [Google Scholar]
- 12.Thoenen H, Sendtner M. Neurotrophins: From enthusiastic expectations through sobering experiences to rational therapeutic approaches. Nat Neurosci. 2002;5(Suppl):1046–1050. doi: 10.1038/nn938. [DOI] [PubMed] [Google Scholar]
- 13.Marconi P, et al. Replication-defective herpes simplex virus vectors for neurotrophic factor gene transfer in vitro and in vivo. Gene Ther. 1999;6:904–912. doi: 10.1038/sj.gt.3300882. [DOI] [PubMed] [Google Scholar]
- 14.Marconi P, et al. Effects of defective herpes simplex vectors expressing neurotrophic factors on the proliferation and differentiation of nervous cells in vivo. Gene Ther. 2005;12:559–569. doi: 10.1038/sj.gt.3302438. [DOI] [PubMed] [Google Scholar]
- 15.Pitkanen A, Schwartzkroin PA, Moshè SL. Models of seizures and epilepsy. Burlington, MA: Elsevier Academic; 2006. [Google Scholar]
- 16.Lehmann TN, et al. Alterations of neuronal connectivity in area CA1 of hippocampal slices from temporal lobe epilepsy patients and from pilocarpine-treated epileptic rats. Epilepsia. 2000;41(Suppl 6):S190–S194. doi: 10.1111/j.1528-1157.2000.tb01580.x. [DOI] [PubMed] [Google Scholar]
- 17.Poirier JL, Capek R, De Koninck Y. Differential progression of Dark Neuron and Fluoro-Jade labelling in the rat hippocampus following pilocarpine-induced status epilepticus. Neuroscience. 2000;97:59–68. doi: 10.1016/s0306-4522(00)00026-9. [DOI] [PubMed] [Google Scholar]
- 18.Parent JM, et al. Dentate granule cell neurogenesis is increased by seizures and contributes to aberrant network reorganization in the adult rat hippocampus. J Neurosci. 1997;17:3727–3738. doi: 10.1523/JNEUROSCI.17-10-03727.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Parent JM, Elliott RC, Pleasure SJ, Barbaro NM, Lowenstein DH. Aberrant seizure-induced neurogenesis in experimental temporal lobe epilepsy. Ann Neurol. 2006;59:81–91. doi: 10.1002/ana.20699. [DOI] [PubMed] [Google Scholar]
- 20.Parent JM, von dem Bussche N, Lowenstein DH. Prolonged seizures recruit caudal subventricular zone glial progenitors into the injured hippocampus. Hippocampus. 2006;16:321–328. doi: 10.1002/hipo.20166. [DOI] [PubMed] [Google Scholar]
- 21.Kempermann G, Jessberger S, Steiner B, Kronenberg G. Milestones of neuronal development in the adult hippocampus. Trends Neurosci. 2004;27:447–452. doi: 10.1016/j.tins.2004.05.013. [DOI] [PubMed] [Google Scholar]
- 22.Zhao C, Deng W, Gage FH. Mechanisms and functional implications of adult neurogenesis. Cell. 2008;132:645–660. doi: 10.1016/j.cell.2008.01.033. [DOI] [PubMed] [Google Scholar]
- 23.Steiner B, et al. Type-2 cells as link between glial and neuronal lineage in adult hippocampal neurogenesis. Glia. 2006;54:805–814. doi: 10.1002/glia.20407. [DOI] [PubMed] [Google Scholar]
- 24.McCloskey DP, Hintz TM, Pierce JP, Scharfman HE. Stereological methods reveal the robust size and stability of ectopic hilar granule cells after pilocarpine-induced status epilepticus in the adult rat. Eur J Neurosci. 2006;24:2203–2210. doi: 10.1111/j.1460-9568.2006.05101.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Garzillo CL, Mello LE. Characterization of reactive astrocytes in the chronic phase of the pilocarpine model of epilepsy. Epilepsia. 2002;43(Suppl 5):107–109. doi: 10.1046/j.1528-1157.43.s.5.40.x. [DOI] [PubMed] [Google Scholar]
- 26.Freund TF, Buzsaki G. Interneurons of the hippocampus. Hippocampus. 1996;6:347–470. doi: 10.1002/(SICI)1098-1063(1996)6:4<347::AID-HIPO1>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 27.Cossart R, et al. Dendritic but not somatic GABAergic inhibition is decreased in experimental epilepsy. Nat Neurosci. 2001;4:52–62. doi: 10.1038/82900. [DOI] [PubMed] [Google Scholar]
- 28.Binaschi A, Bregola G, Simonato M. On the role of somatostatin in seizure control: Clues from the hippocampus. Rev Neurosci. 2003;14:285–301. doi: 10.1515/revneuro.2003.14.3.285. [DOI] [PubMed] [Google Scholar]
- 29.Sun C, Mtchedlishvili Z, Bertram EH, Erisir A, Kapur J. Selective loss of dentate hilar interneurons contributes to reduced synaptic inhibition of granule cells in an electrical stimulation-based animal model of temporal lobe epilepsy. J Comp Neurol. 2007;500:876–893. doi: 10.1002/cne.21207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Magloczky Z, Halasz P, Vajda J, Czirjak S, Freund TF. Loss of Calbindin-D28K immunoreactivity from dentate granule cells in human temporal lobe epilepsy. Neuroscience. 1997;76:377–385. doi: 10.1016/s0306-4522(96)00440-x. [DOI] [PubMed] [Google Scholar]
- 31.Scharfman HE, Sollas AL, Goodman JH. Spontaneous recurrent seizures after pilocarpine-induced status epilepticus activate calbindin-immunoreactive hilar cells of the rat dentate gyrus. Neuroscience. 2002;111:71–81. doi: 10.1016/s0306-4522(01)00599-1. [DOI] [PubMed] [Google Scholar]
- 32.Racine RJ. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol. 1972;32:281–294. doi: 10.1016/0013-4694(72)90177-0. [DOI] [PubMed] [Google Scholar]
- 33.Fabene PF, Marzola P, Sbarbati A, Bentivoglio M. Magnetic resonance imaging of changes elicited by status epilepticus in the rat brain: Diffusion-weighted and T2-weighted images, regional blood volume maps, and direct correlation with tissue and cell damage. Neuroimage. 2003;18:375–389. doi: 10.1016/s1053-8119(02)00025-3. [DOI] [PubMed] [Google Scholar]
- 34.Sloviter RS. The neurobiology of temporal lobe epilepsy: Too much information, not enough knowledge. C R Biol. 2005;328:143–153. doi: 10.1016/j.crvi.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 35.Schmued LC, Stowers CC, Scallet AC, Xu L. Fluoro-Jade C results in ultra high resolution and contrast labeling of degenerating neurons. Brain Res. 2005;1035:24–31. doi: 10.1016/j.brainres.2004.11.054. [DOI] [PubMed] [Google Scholar]
- 36.von Bohlen und Halbach O, Unsicker K. Morphological alterations in the amygdala and hippocampus of mice during ageing. Eur J Neurosci. 2002;16:2434–2440. doi: 10.1046/j.1460-9568.2002.02405.x. [DOI] [PubMed] [Google Scholar]
- 37.Zucchini S, et al. Neuroprotective activity of CHF3381, a putative N-methyl-D-aspartate receptor antagonist. Neuroreport. 2002;13:2071–2074. doi: 10.1097/00001756-200211150-00016. [DOI] [PubMed] [Google Scholar]
- 38.Zucchini S, et al. FGF-2 overexpression increases excitability and seizure susceptibility but decreases seizure-induced cell loss. J Neurosci. 2008;28:13112–13124. doi: 10.1523/JNEUROSCI.1472-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.