Abstract
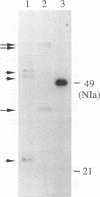
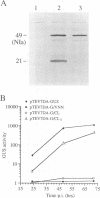
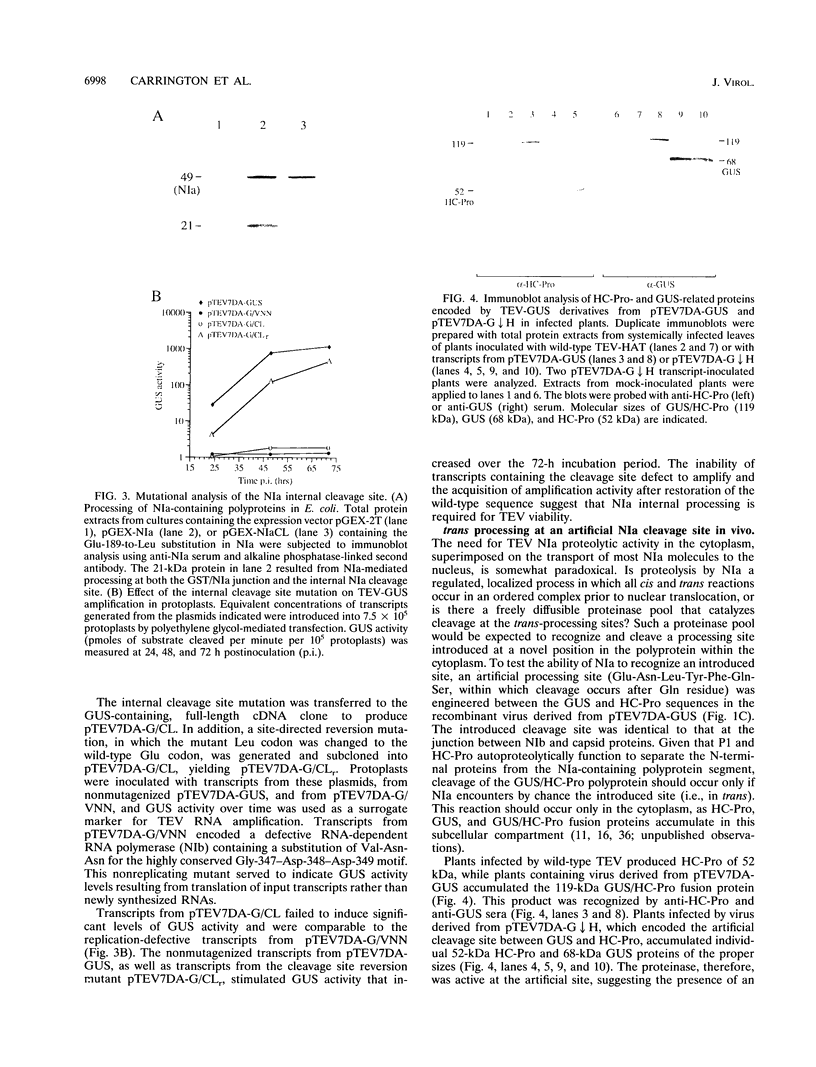
The NIa protein of plant potyviruses is a bifunctional protein containing an N-terminal VPg domain and a C-terminal proteinase region. The majority of tobacco etch potyvirus (TEV) NIa molecules are localized to the nucleus of infected cells, although a proportion of NIa is attached covalently as VPg to viral RNA in the cytoplasm. A suboptimal cleavage site that is recognized by the NIa proteinase is located between the two domains. This site was found to be utilized in the VPg-associated, but not the nuclear, pool of NIa. A mutation converting Glu-189 to Leu at the P1 position of the processing site inhibited internal cleavage. Introduction of this mutation into TEV-GUS, an engineered variant of TEV that expresses a reporter protein (beta-glucuronidase [GUS]) fused to the N terminus of the helper component-proteinase (HC-Pro), rendered the virus replication defective in tobacco protoplasts. Site-specific reversion of the mutant internal processing site to the wild-type sequence restored virus viability. In addition, the trans-processing activity of NIa proteinase was tested in vivo after introduction of an artificial cleavage site between the GUS and HC-Pro sequences in the cytoplasmic GUS/HC-Pro polyprotein encoded by TEV-GUS. The novel site was recognized and processed in plants infected by the engineered virus, indicating the presence of excess NIa processing capacity in the cytoplasm. The potential roles of internal NIa processing in TEV-infected cells are discussed.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andino R., Rieckhof G. E., Baltimore D. A functional ribonucleoprotein complex forms around the 5' end of poliovirus RNA. Cell. 1990 Oct 19;63(2):369–380. doi: 10.1016/0092-8674(90)90170-j. [DOI] [PubMed] [Google Scholar]
- Andino R., Rieckhof G. E., Trono D., Baltimore D. Substitutions in the protease (3Cpro) gene of poliovirus can suppress a mutation in the 5' noncoding region. J Virol. 1990 Feb;64(2):607–612. doi: 10.1128/jvi.64.2.607-612.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bazan J. F., Fletterick R. J. Viral cysteine proteases are homologous to the trypsin-like family of serine proteases: structural and functional implications. Proc Natl Acad Sci U S A. 1988 Nov;85(21):7872–7876. doi: 10.1073/pnas.85.21.7872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Cary S. M., Dougherty W. G. Mutational analysis of tobacco etch virus polyprotein processing: cis and trans proteolytic activities of polyproteins containing the 49-kilodalton proteinase. J Virol. 1988 Jul;62(7):2313–2320. doi: 10.1128/jvi.62.7.2313-2320.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Cary S. M., Parks T. D., Dougherty W. G. A second proteinase encoded by a plant potyvirus genome. EMBO J. 1989 Feb;8(2):365–370. doi: 10.1002/j.1460-2075.1989.tb03386.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Dougherty W. G. A viral cleavage site cassette: identification of amino acid sequences required for tobacco etch virus polyprotein processing. Proc Natl Acad Sci U S A. 1988 May;85(10):3391–3395. doi: 10.1073/pnas.85.10.3391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Dougherty W. G. Small nuclear inclusion protein encoded by a plant potyvirus genome is a protease. J Virol. 1987 Aug;61(8):2540–2548. doi: 10.1128/jvi.61.8.2540-2548.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D. Cap-independent enhancement of translation by a plant potyvirus 5' nontranslated region. J Virol. 1990 Apr;64(4):1590–1597. doi: 10.1128/jvi.64.4.1590-1597.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D., Leinicke A. J. Bipartite signal sequence mediates nuclear translocation of the plant potyviral NIa protein. Plant Cell. 1991 Sep;3(9):953–962. doi: 10.1105/tpc.3.9.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D., Oh C. S. Expression of potyviral polyproteins in transgenic plants reveals three proteolytic activities required for complete processing. EMBO J. 1990 May;9(5):1347–1353. doi: 10.1002/j.1460-2075.1990.tb08249.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D., Sanders T. C. Autocatalytic processing of the potyvirus helper component proteinase in Escherichia coli and in vitro. J Virol. 1989 Oct;63(10):4459–4463. doi: 10.1128/jvi.63.10.4459-4463.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson W. O. Synthesis of TMV RNA at restrictive high temperatures. Virology. 1976 Sep;73(2):319–326. doi: 10.1016/0042-6822(76)90393-7. [DOI] [PubMed] [Google Scholar]
- Dolja V. V., Herndon K. L., Pirone T. P., Carrington J. C. Spontaneous mutagenesis of a plant potyvirus genome after insertion of a foreign gene. J Virol. 1993 Oct;67(10):5968–5975. doi: 10.1128/jvi.67.10.5968-5975.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolja V. V., McBride H. J., Carrington J. C. Tagging of plant potyvirus replication and movement by insertion of beta-glucuronidase into the viral polyprotein. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10208–10212. doi: 10.1073/pnas.89.21.10208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dougherty W. G., Carrington J. C., Cary S. M., Parks T. D. Biochemical and mutational analysis of a plant virus polyprotein cleavage site. EMBO J. 1988 May;7(5):1281–1287. doi: 10.1002/j.1460-2075.1988.tb02942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dougherty W. G., Cary S. M., Parks T. D. Molecular genetic analysis of a plant virus polyprotein cleavage site: a model. Virology. 1989 Aug;171(2):356–364. doi: 10.1016/0042-6822(89)90603-x. [DOI] [PubMed] [Google Scholar]
- Dougherty W. G., Parks T. D., Cary S. M., Bazan J. F., Fletterick R. J. Characterization of the catalytic residues of the tobacco etch virus 49-kDa proteinase. Virology. 1989 Sep;172(1):302–310. doi: 10.1016/0042-6822(89)90132-3. [DOI] [PubMed] [Google Scholar]
- Dougherty W. G., Parks T. D. Molecular genetic and biochemical evidence for the involvement of the heptapeptide cleavage sequence in determining the reaction profile at two tobacco etch virus cleavage sites in cell-free assays. Virology. 1989 Sep;172(1):145–155. doi: 10.1016/0042-6822(89)90116-5. [DOI] [PubMed] [Google Scholar]
- Dougherty W. G., Parks T. D. Post-translational processing of the tobacco etch virus 49-kDa small nuclear inclusion polyprotein: identification of an internal cleavage site and delimitation of VPg and proteinase domains. Virology. 1991 Aug;183(2):449–456. doi: 10.1016/0042-6822(91)90974-g. [DOI] [PubMed] [Google Scholar]
- García J. A., Laín S., Cervera M. T., Riechmann J. L., Martín M. T. Mutational analysis of plum pox potyvirus polyprotein processing by the NIa protease in Escherichia coli. J Gen Virol. 1990 Dec;71(Pt 12):2773–2779. doi: 10.1099/0022-1317-71-12-2773. [DOI] [PubMed] [Google Scholar]
- Gorbalenya A. E., Donchenko A. P., Blinov V. M., Koonin E. V. Cysteine proteases of positive strand RNA viruses and chymotrypsin-like serine proteases. A distinct protein superfamily with a common structural fold. FEBS Lett. 1989 Jan 30;243(2):103–114. doi: 10.1016/0014-5793(89)80109-7. [DOI] [PubMed] [Google Scholar]
- Hellmann G. M., Shaw J. G., Rhoads R. E. In vitro analysis of tobacco vein mottling virus NIa cistron: evidence for a virus-encoded protease. Virology. 1988 Apr;163(2):554–562. doi: 10.1016/0042-6822(88)90296-6. [DOI] [PubMed] [Google Scholar]
- Kunkel T. A., Roberts J. D., Zakour R. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Methods Enzymol. 1987;154:367–382. doi: 10.1016/0076-6879(87)54085-x. [DOI] [PubMed] [Google Scholar]
- Murphy J. F., Rhoads R. E., Hunt A. G., Shaw J. G. The VPg of tobacco etch virus RNA is the 49-kDa proteinase or the N-terminal 24-kDa part of the proteinase. Virology. 1990 Sep;178(1):285–288. doi: 10.1016/0042-6822(90)90405-g. [DOI] [PubMed] [Google Scholar]
- Murphy J. F., Rychlik W., Rhoads R. E., Hunt A. G., Shaw J. G. A tyrosine residue in the small nuclear inclusion protein of tobacco vein mottling virus links the VPg to the viral RNA. J Virol. 1991 Jan;65(1):511–513. doi: 10.1128/jvi.65.1.511-513.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Restrepo M. A., Freed D. D., Carrington J. C. Nuclear transport of plant potyviral proteins. Plant Cell. 1990 Oct;2(10):987–998. doi: 10.1105/tpc.2.10.987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riechmann J. L., Laín S., García J. A. Highlights and prospects of potyvirus molecular biology. J Gen Virol. 1992 Jan;73(Pt 1):1–16. doi: 10.1099/0022-1317-73-1-1. [DOI] [PubMed] [Google Scholar]
- Riechmann J. L., Laín S., García J. A. The genome-linked protein and 5' end RNA sequence of plum pox potyvirus. J Gen Virol. 1989 Oct;70(Pt 10):2785–2789. doi: 10.1099/0022-1317-70-10-2785. [DOI] [PubMed] [Google Scholar]
- Rorrer K., Parks T. D., Scheffler B., Bevan M., Dougherty W. G. Autocatalytic activity of the tobacco etch virus NIa proteinase in viral and foreign protein sequences. J Gen Virol. 1992 Apr;73(Pt 4):775–783. doi: 10.1099/0022-1317-73-4-775. [DOI] [PubMed] [Google Scholar]
- Shahabuddin M., Shaw J. G., Rhoads R. E. Mapping of the tobacco vein mottling virus VPg cistron. Virology. 1988 Apr;163(2):635–637. doi: 10.1016/0042-6822(88)90307-8. [DOI] [PubMed] [Google Scholar]
- Verchot J., Koonin E. V., Carrington J. C. The 35-kDa protein from the N-terminus of the potyviral polyprotein functions as a third virus-encoded proteinase. Virology. 1991 Dec;185(2):527–535. doi: 10.1016/0042-6822(91)90522-d. [DOI] [PubMed] [Google Scholar]