Abstract
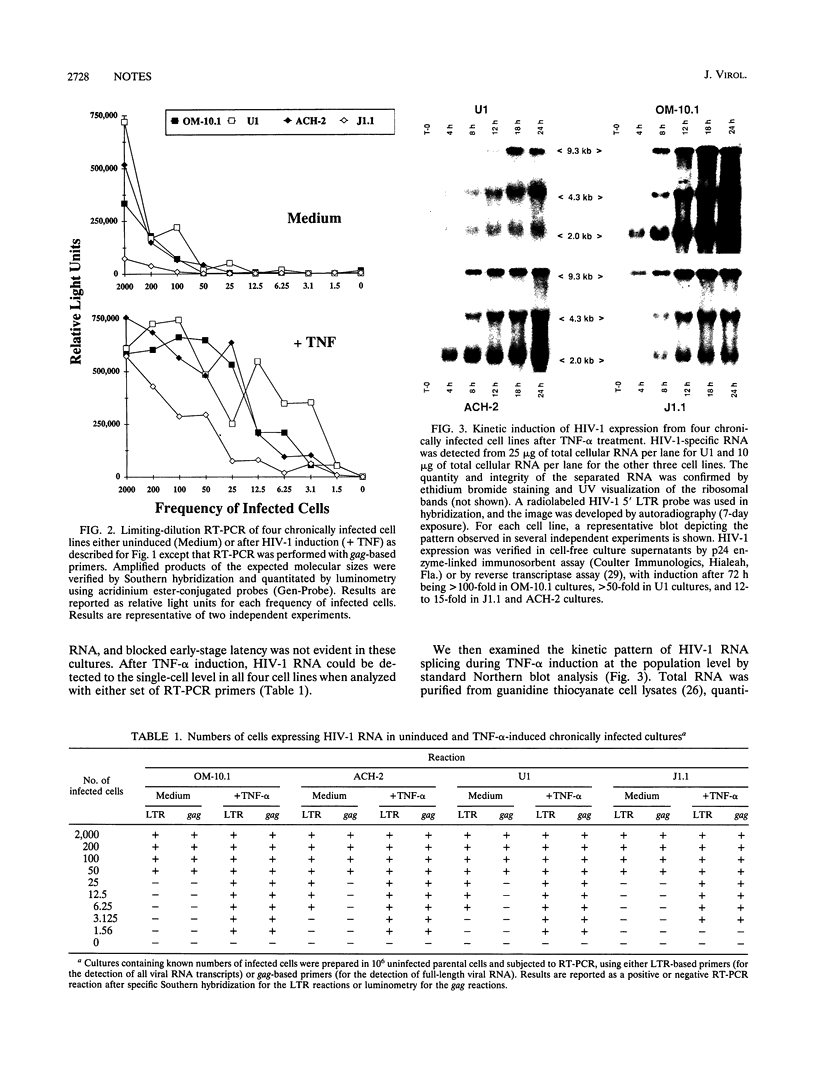
Recent information has suggested that posttranscriptional mechanisms, whereby human immunodeficiency virus type 1 (HIV-1) RNA exists as multiply spliced transcripts without promoting an accumulation of the larger messages, are responsible for maintaining a stable state of nonproductive viral expression or viral latency. To test the universality of these observations, we compared the patterns of viral RNA splicing and the frequencies of cells actually harboring HIV-1 RNA in four chronically HIV-1-infected cell lines (U1 [promonocytic], ACH-2 [T lymphocytic], OM-10.1 [promyelocytic], and J1.1 [T lymphocytic]). In uninduced U1 and ACH-2 cultures, a high frequency of cells (approximately one in six) contained HIV-1 RNA but mainly as multiply spliced transcripts, again supporting a posttranscriptional mechanism maintaining viral latency. In sharp contrast, only 1 in 50 cells in uninduced OM-10.1 and J1.1 cultures contained HIV-1 RNA, indicating a primary transcriptional mechanism controlling viral expression in these cells. Furthermore, those OM-10.1 and J1.1 cells that did contain viral RNA were in a state of productive HIV-1 expression marked by the presence of both spliced and unspliced transcripts. Even though the total absence of viral RNA in the majority of OM-10.1 and J1.1 cells indicated a state of absolute latency, treatment with tumor necrosis factor alpha induced transcription of HIV-1 RNA in nearly 100% of the cells in all four of the chronically infected cultures. Tumor necrosis factor alpha induction of U1, ACH-2, and OM-10.1 cultures resulted in an initial accumulation of multiply spliced HIV-1 RNA followed by a transition to the larger unspliced viral RNA transcripts. This RNA splice transition was less apparent in the J1.1 cell line. These results demonstrate that host cell-specific transcriptional and posttranscriptional mechanisms are important factors in the control of HIV-1 latency.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bednarik D. P., Folks T. M. Mechanisms of HIV-1 latency. AIDS. 1992 Jan;6(1):3–16. doi: 10.1097/00002030-199201000-00001. [DOI] [PubMed] [Google Scholar]
- Butera S. T., Folks T. M. Application of latent HIV-1 infected cellular models to therapeutic intervention. AIDS Res Hum Retroviruses. 1992 Jun;8(6):991–995. doi: 10.1089/aid.1992.8.991. [DOI] [PubMed] [Google Scholar]
- Butera S. T., Perez V. L., Wu B. Y., Nabel G. J., Folks T. M. Oscillation of the human immunodeficiency virus surface receptor is regulated by the state of viral activation in a CD4+ cell model of chronic infection. J Virol. 1991 Sep;65(9):4645–4653. doi: 10.1128/jvi.65.9.4645-4653.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butera S. T., Roberts B. D., Folks T. M. Regulation of HIV-1 expression by cytokine networks in a CD4+ model of chronic infection. J Immunol. 1993 Jan 15;150(2):625–634. [PubMed] [Google Scholar]
- Clouse K. A., Powell D., Washington I., Poli G., Strebel K., Farrar W., Barstad P., Kovacs J., Fauci A. S., Folks T. M. Monokine regulation of human immunodeficiency virus-1 expression in a chronically infected human T cell clone. J Immunol. 1989 Jan 15;142(2):431–438. [PubMed] [Google Scholar]
- Embretson J., Zupancic M., Ribas J. L., Burke A., Racz P., Tenner-Racz K., Haase A. T. Massive covert infection of helper T lymphocytes and macrophages by HIV during the incubation period of AIDS. Nature. 1993 Mar 25;362(6418):359–362. doi: 10.1038/362359a0. [DOI] [PubMed] [Google Scholar]
- Fauci A. S., Schnittman S. M., Poli G., Koenig S., Pantaleo G. NIH conference. Immunopathogenic mechanisms in human immunodeficiency virus (HIV) infection. Ann Intern Med. 1991 Apr 15;114(8):678–693. doi: 10.7326/0003-4819-114-8-678. [DOI] [PubMed] [Google Scholar]
- Feinberg M. B., Jarrett R. F., Aldovini A., Gallo R. C., Wong-Staal F. HTLV-III expression and production involve complex regulation at the levels of splicing and translation of viral RNA. Cell. 1986 Sep 12;46(6):807–817. doi: 10.1016/0092-8674(86)90062-0. [DOI] [PubMed] [Google Scholar]
- Felber B. K., Hadzopoulou-Cladaras M., Cladaras C., Copeland T., Pavlakis G. N. rev protein of human immunodeficiency virus type 1 affects the stability and transport of the viral mRNA. Proc Natl Acad Sci U S A. 1989 Mar;86(5):1495–1499. doi: 10.1073/pnas.86.5.1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Folks T. M., Justement J., Kinter A., Dinarello C. A., Fauci A. S. Cytokine-induced expression of HIV-1 in a chronically infected promonocyte cell line. Science. 1987 Nov 6;238(4828):800–802. doi: 10.1126/science.3313729. [DOI] [PubMed] [Google Scholar]
- Kim S. Y., Byrn R., Groopman J., Baltimore D. Temporal aspects of DNA and RNA synthesis during human immunodeficiency virus infection: evidence for differential gene expression. J Virol. 1989 Sep;63(9):3708–3713. doi: 10.1128/jvi.63.9.3708-3713.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malim M. H., Cullen B. R. HIV-1 structural gene expression requires the binding of multiple Rev monomers to the viral RRE: implications for HIV-1 latency. Cell. 1991 Apr 19;65(2):241–248. doi: 10.1016/0092-8674(91)90158-u. [DOI] [PubMed] [Google Scholar]
- Malim M. H., Hauber J., Le S. Y., Maizel J. V., Cullen B. R. The HIV-1 rev trans-activator acts through a structured target sequence to activate nuclear export of unspliced viral mRNA. Nature. 1989 Mar 16;338(6212):254–257. doi: 10.1038/338254a0. [DOI] [PubMed] [Google Scholar]
- Michael N. L., Morrow P., Mosca J., Vahey M., Burke D. S., Redfield R. R. Induction of human immunodeficiency virus type 1 expression in chronically infected cells is associated primarily with a shift in RNA splicing patterns. J Virol. 1991 Mar;65(3):1291–1303. doi: 10.1128/jvi.65.3.1291-1303.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muesing M. A., Smith D. H., Cabradilla C. D., Benton C. V., Lasky L. A., Capon D. J. Nucleic acid structure and expression of the human AIDS/lymphadenopathy retrovirus. Nature. 1985 Feb 7;313(6002):450–458. doi: 10.1038/313450a0. [DOI] [PubMed] [Google Scholar]
- Ou C. Y., Kwok S., Mitchell S. W., Mack D. H., Sninsky J. J., Krebs J. W., Feorino P., Warfield D., Schochetman G. DNA amplification for direct detection of HIV-1 in DNA of peripheral blood mononuclear cells. Science. 1988 Jan 15;239(4837):295–297. doi: 10.1126/science.3336784. [DOI] [PubMed] [Google Scholar]
- Pantaleo G., Graziosi C., Demarest J. F., Butini L., Montroni M., Fox C. H., Orenstein J. M., Kotler D. P., Fauci A. S. HIV infection is active and progressive in lymphoid tissue during the clinically latent stage of disease. Nature. 1993 Mar 25;362(6418):355–358. doi: 10.1038/362355a0. [DOI] [PubMed] [Google Scholar]
- Pantaleo G., Graziosi C., Fauci A. S. New concepts in the immunopathogenesis of human immunodeficiency virus infection. N Engl J Med. 1993 Feb 4;328(5):327–335. doi: 10.1056/NEJM199302043280508. [DOI] [PubMed] [Google Scholar]
- Perez V. L., Rowe T., Justement J. S., Butera S. T., June C. H., Folks T. M. An HIV-1-infected T cell clone defective in IL-2 production and Ca2+ mobilization after CD3 stimulation. J Immunol. 1991 Nov 1;147(9):3145–3148. [PubMed] [Google Scholar]
- Pomerantz R. J., Bagasra O., Baltimore D. Cellular latency of human immunodeficiency virus type 1. Curr Opin Immunol. 1992 Aug;4(4):475–480. doi: 10.1016/s0952-7915(06)80042-7. [DOI] [PubMed] [Google Scholar]
- Pomerantz R. J., Seshamma T., Trono D. Efficient replication of human immunodeficiency virus type 1 requires a threshold level of Rev: potential implications for latency. J Virol. 1992 Mar;66(3):1809–1813. doi: 10.1128/jvi.66.3.1809-1813.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pomerantz R. J., Trono D., Feinberg M. B., Baltimore D. Cells nonproductively infected with HIV-1 exhibit an aberrant pattern of viral RNA expression: a molecular model for latency. Cell. 1990 Jun 29;61(7):1271–1276. doi: 10.1016/0092-8674(90)90691-7. [DOI] [PubMed] [Google Scholar]
- Rogers M. F., Ou C. Y., Rayfield M., Thomas P. A., Schoenbaum E. E., Abrams E., Krasinski K., Selwyn P. A., Moore J., Kaul A. Use of the polymerase chain reaction for early detection of the proviral sequences of human immunodeficiency virus in infants born to seropositive mothers. New York City Collaborative Study of Maternal HIV Transmission and Montefiore Medical Center HIV Perinatal Transmission Study Group. N Engl J Med. 1989 Jun 22;320(25):1649–1654. doi: 10.1056/NEJM198906223202503. [DOI] [PubMed] [Google Scholar]
- Seshamma T., Bagasra O., Trono D., Baltimore D., Pomerantz R. J. Blocked early-stage latency in the peripheral blood cells of certain individuals infected with human immunodeficiency virus type 1. Proc Natl Acad Sci U S A. 1992 Nov 15;89(22):10663–10667. doi: 10.1073/pnas.89.22.10663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw G. M., Hahn B. H., Arya S. K., Groopman J. E., Gallo R. C., Wong-Staal F. Molecular characterization of human T-cell leukemia (lymphotropic) virus type III in the acquired immune deficiency syndrome. Science. 1984 Dec 7;226(4679):1165–1171. doi: 10.1126/science.6095449. [DOI] [PubMed] [Google Scholar]
- Strohman R. C., Moss P. S., Micou-Eastwood J., Spector D., Przybyla A., Paterson B. Messenger RNA for myosin polypeptides: isolation from single myogenic cell cultures. Cell. 1977 Feb;10(2):265–273. doi: 10.1016/0092-8674(77)90220-3. [DOI] [PubMed] [Google Scholar]
- Trono D., Baltimore D. A human cell factor is essential for HIV-1 Rev action. EMBO J. 1990 Dec;9(12):4155–4160. doi: 10.1002/j.1460-2075.1990.tb07638.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaishnav Y. N., Vaishnav M., Wong-Staal F. Identification and characterization of a nuclear factor that specifically binds to the Rev response element (RRE) of human immunodeficiency virus type 1 (HIV-1). New Biol. 1991 Feb;3(2):142–150. [PubMed] [Google Scholar]
- Willey R. L., Smith D. H., Lasky L. A., Theodore T. S., Earl P. L., Moss B., Capon D. J., Martin M. A. In vitro mutagenesis identifies a region within the envelope gene of the human immunodeficiency virus that is critical for infectivity. J Virol. 1988 Jan;62(1):139–147. doi: 10.1128/jvi.62.1.139-147.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]