Abstract
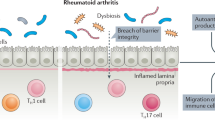
Individuals at high risk of developing seropositive rheumatoid arthritis (RA) can be identified for translational research and disease prevention studies through the presence of highly informative and predictive patterns of RA-related autoantibodies, especially anti-citrullinated protein antibodies (ACPAs), in the serum. In serologically positive individuals without arthritis, designated ACPA positive at risk, the presence of mucosal inflammatory processes associated with the presence of local ACPA production has been demonstrated. In other at-risk populations, local RA-related autoantibody production is present even in the absence of serum autoantibodies. Additionally, a proportion of at-risk individuals exhibit local mucosal ACPA production in the lung, as well as radiographic small-airway disease, sputum hypercellularity and increased neutrophil extracellular trap formation. Other mucosal sites in at-risk individuals also exhibit autoantibody production, inflammation and/or evidence of dysbiosis. As the proportion of individuals who exhibit such localized inflammation-associated ACPA production is substantially higher than the likelihood of an individual developing future RA, this finding raises the hypothesis that mucosal ACPAs have biologically relevant protective roles. Identifying the mechanisms that drive both the generation and loss of externally focused mucosal ACPA production and promote systemic autoantibody expression and ultimately arthritis development should provide insights into new therapeutic approaches to prevent RA.
Key points
-
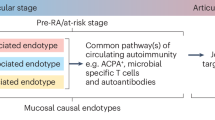
Patients who eventually develop seropositive rheumatoid arthritis (RA) pass through a period of RA-related autoantibody positivity that is associated with increased levels of cytokines and chemokines.
-
Various mucosal processes can influence the development of systemic immunity and autoimmunity.
-
Individuals who are at high risk of the future development of RA demonstrate evidence of chronic systemic and mucosal inflammation.
-
Rather than reflecting a loss of citrullinated antigen tolerance, immunoglobulin A (IgA) anti-citrullinated protein antibodies (ACPAs) are normally produced locally; a systemic IgG response likely results from loss of externally focused compartmentalization.
-
Ongoing studies are linking the development of mucosal dysbiosis, inflammation and autoantibody production to the next stages of development of systemic autoimmunity.
-
Novel autoimmune-promoting processes are likely to be identified that function primarily, or exclusively, in the preclinical period of RA and could be the targets for new prevention strategies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Catrina, A. I., Svensson, C. I., Malmström, V., Schett, G. & Klareskog, L. Mechanisms leading from systemic autoimmunity to joint-specific disease in rheumatoid arthritis. Nat. Rev. Rheumatol. 13, 79–86 (2017).
Catrina, A. I., Deane, K. D. & Scher, J. U. Gene, environment, microbiome and mucosal immune tolerance in rheumatoid arthritis. Rheumatology 55, 391–402 (2016).
Viatte, S., Plant, D. & Raychaudhuri, S. Genetics and epigenetics of rheumatoid arthritis. Nat. Rev. Rheumatol. 9, 141–153 (2013).
Firestein, G. S. & McInnes, I. B. Immunopathogenesis of rheumatoid arthritis. Immunity 46, 183–196 (2017).
Nielen, M. M. J. et al. Specific autoantibodies precede the symptoms of rheumatoid arthritis. Arthritis Rheum. 50, 380–386 (2004).
Rantapaa Dahlqvist, S. et al. Antibodies against citrullinated peptides (CCP) predict the development of rheumatoid arthritis. Arthritis Rheum. 48, 2701–2705 (2003).
Majka, D. S. et al. Duration of preclinical rheumatoid arthritis-related autoantibody positivity increases in subject with older age at time of disease diagnosis. Ann. Rheum. Dis. 67, 801–807 (2008).
Karlson, E. W. et al. Biomarkers of inflammation and development of rheumatoid arthritis in women from two prospective cohort studies. Arthritis Rheum. 60, 641–652 (2009).
Sokolove, J. et al. Autoantibody epitope spreading in the pre-clinical phase predicts progression to rheumatoid arthritis. PLOS One 7, e35296 (2012).
Deane, K. D. et al. The number of elevated cytokines and chemokines in preclinical seropositive rheumatoid arthritis predicts time to diagnosis in an age-dependent manner. Arthritis Rheum. 62, 3161–3172 (2010).
Gerlag, D. M. et al. EULAR recommendations for terminology and research in individuals at risk of rheumatoid arthritis: report from the Study Group for Risk Factors for Rheumatoid Arthritis. Ann. Rheum. Dis. 71, 638–641 (2012).
Scher, J. U. et al. The lung microbiota in early rheumatoid arthritis and autoimmunity. Microbiome 4, 60 (2016).
Scher, J. U. et al. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. eLife https://doi.org/10.7554/eLife.01202 (2013).
Scher, J. U. et al. Periodontal disease and the oral microbiota in new-onset rheumatoid arthritis. Arthritis Rheum. 64, 3083–3094 (2012).
Zhang, X. et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 21, 895–905 (2015).
Chen, J. et al. An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis. Genome Med. 8, 43 (2016).
Demoruelle, M. K. et al. Anti-citrullinated protein antibodies are associated with neutrophil extracellular traps in the sputum in relatives of rheumatoid arthritis patients. Arthritis Rheum. 69, 1165–1175 (2017).
Willis, V. C. et al. Sputa autoantibodies in patients with established rheumatoid arthritis and subjects at-risk for future clinically apparent disease. Arthritis Rheum. 65, 2545–2554 (2013).
Lee, D. M. & Weinblatt, M. E. Rheumatoid arthritis. Lancet 359, 903–911 (2001).
van Steenbergen, H. W. et al. EULAR definition of arthralgia suspicious for progression to rheumatoid arthritis. Ann. Rheum. Dis. 76, 491–496 (2017).
Fuchs, H. A. & Sergent, J. S. in Arthritis and Allied Conditions: A Textbook of Rheumatology (ed. Koopman, W. J.) 1041–1070 (Williams and Wilkins, 1997).
Schett, G. & Gravallese, E. M. Bone erosion in rheumatoid arthritis: Mechanisms, diagnosis and treatment. Nat. Rev. Rheumatol. 8, 656–664 (2012).
Silman, A. J. Epidemiology of rheumatoid arthritis. APMIS 102, 721–728 (1994).
Karlson, E. W. & Costenbader, K. H. Epidemiology: Interpreting studies of interactions between RA risk factors. Nat. Rev. Rheumatol. 6, 72–73 (2010).
Karlson, E. W. & Deane, K. D. Environmental and gene-environment interactions and risk of rheumatoid arthritis. Rheum. Dis. Clin. North Am. 38, 405–426 (2012).
Klareskog, L., Malmstrom, V., Lundberg, K., Padyukov, L. & Alfredsson, L. Smoking, citrullination and genetic variability in the immunopathogenesis of rheumatoid arthritis. Semin. Immunol. 23, 92–98 (2011).
Aletaha, D. et al. Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 62, 2569–2581 (2010).
Arnett, F. C. et al. The American Rheumatism Association1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 31, 314–324 (1998).
de Rooy, D. P. C., van der Linden, M. P. M., Knevel, R., Huizinga, T. W. & van der Helm-van Mil, A. H. M. Predicting arthritis outcomes-what can be learned from the Leiden Early Arthritis Clinic? Rheumatology 50, 93–100 (2011).
van der Helm-van Mil, A. H. M. et al. A prediction rule for disease outcome in patients with recent-onset undifferentiated arthritis: how to guide individual treatment decisions. Arthritis Rheum. 56, 433–440 (2007).
Padyukov, L., Silva, C., Stolt, P., Alfredsson, L. & Klareskog, L. A gene-environment interaction between smoking and shared epitope genes in HLA-DR provides a high risk of seropositive rheumatoid arthritis. Arthritis Rheum. 50, 3085–3092 (2004).
Schellekens, G. A., de Jong, B. A. W., van den Hoogen, F. H. J., van de Putte, L. B. A. & van Venrooij, W. J. Citrulline is an essential constituent of antigenic determinants recognized by rheumatoid arthritis-specific autoantibodies. J. Clin. Invest. 101, 273–281 (1998).
van Venrooij, W. J. & Pruijn, G. J. M. Citrullination: a small change for a protein with great consequences for rheumatoid arthritis. Arthritis Res. Ther. 2, 249–251 (2000).
Bas, S., Genevay, S., Meyer, O. & Gabay, C. Anti-cyclic citrullinated peptide antibodies, IgM and IgA rheumatoid factors in the diagnosis and prognosis of rheumatoid arthritis. Rheumatology 42, 677–680 (2003).
Trouw, L. A., Rispens, T. & Toes, R. E. M. Beyond citrullination: other post-translational protein modifications in rheumatoid arthritis. Nat. Rev. Rheumatol. 13, 331–339 (2017).
Shi, J. et al. Autoantibodies recognizing carbamylated proteins are present in sera of patients with rheumatoid arthritis and predict joint damage. Proc. Natl Acad. Sci. USA 108, 17372–17377 (2011).
Thiele, G. M. et al. Malondialdehyde-acetaldehyde adducts and anti-malondialdehyde-acetaldehyde antibodies in rheumatoid arthritis. Arthritis Rheum. 67, 645–655 (2015).
Pecani, A. et al. Prevalence, sensitivity and specificity of antibodies against carbamylated proteins in a monocentric cohort of patients with rheumatoid arthritis and other autoimmune rheumatic diseases. Arthritis Res. Ther. 18, 276 (2016).
Gan, R. W. et al. Anti-carbamylated protein antibodies are present prior to rheumatoid arthritis and are associated with its future diagnosis. J. Rheumatol. 42, 572–579 (2015).
Nepom, G. T. & Nepom, B. S. Prediction of susceptibility to rheumatoid arthritis by human leukocyte antigen phenotyping. Rheum. Dis. Clin. North Am. 18, 785–792 (1992).
Begovich, A. B. et al. A missense single-nucleotide polymorphism in a gene encoding a protein tyrosine phosphatase (PTPN22) is associated with rheumatoid arthritis. Am. J. Hum. Genet. 75, 330–337 (2004).
Viatte, S. & Barton, A. Genetics of rheumatoid arthritis susceptibility, severity, and treatment response. Semin. Immunopathol. 39, 395–408 (2017).
Hill, J. A. et al. The conversion of arginine to citrulline allows for a high-affinity peptide interaction with the rheumatoid arthritis-associated HLA-BRB1*0401 MHC Class II molecule. J. Immunol. 171, 538–541 (2003).
Law, S. C. et al. T-cell autoreactivity to citrullinated autoantigenic peptides in rheumatoid arthritis patients carrying HLA-DRB1 shared epitope alleles. Arthritis Res. Ther. 14, R118 (2012).
James, E. et al. Citrulline specific Th1 cells are increased in rheumatoid arthritis and their frequency is influenced by disease duration and therapy. Arthritis Rheum. 66, 1712–1722 (2014).
Klareskog, L. et al. A new model for an etiology of rheumatoid arthritis: smoking may trigger HLA-DR (shared epitope)-restricted immune reactions to autoantigens modified by citrullination. Arthritis Rheum. 54, 38–46 (2006).
Nakano, K., Whitaker, J. W., Boyle, D. L., Wang, W. & Firestein, G. S. DNA methylome signature in rheumatoid arthritis. Ann. Rheum. Dis. 72, 110–117 (2013).
Rhead, B. et al. Rheumatoid arthritis naive T cells share hypermethylation sites with synoviocytes. Arthritis Rheum. 69, 550–559 (2017).
Deane, K. D., Norris, J. M. & Holers, V. M. Pre-clinical rheumatoid arthritis: identification, evaluation and future directions for investigation. Rheum. Dis. Clin. North Am. 36, 213–241 (2010).
Brink, M. et al. Multiplex analyses of antibodies against citrullinated peptides in individuals prior to the development of rheumatoid arthritis. Arthritis Rheum. 65, 899–910 (2013).
Suwannalai, P. et al. Avidity maturation of anti-citrullinated protein antibodies in rheumatoid arthritis. Arthritis Rheum. 64, 1323–1328 (2012).
Trouw, L. A. et al. Anti-cyclic peptide antibodies from rheumatoid arthritis patients activate complement via both the classical and alternative pathways. Arthritis Rheum. 60, 1923–1931 (2009).
Rombouts, Y. et al. Anti-citrullinated protein antibodies acquire a pro-inflammatory Fc glycosylation phenotype prior to the onset of rheumatoid arthritis. Ann. Rheum. Dis. 74, 234–241 (2015).
Rombouts, Y. et al. Extensive glycosylation of ACPA-IgG variable domains modulates binding to citrullinated antigens in rheumatoid arthritis. Ann. Rheum. Dis. 75, 578–585 (2016).
Pfeifle, R. et al. Regulation of autoantibody activity by the IL-23-TH17 axis determines the onset of autoimmune disease. Nat. Immunol. 18, 104–113 (2017).
Harre, U. et al. Induction of osteoclastogenesis and bone loss by human autoantibodies against citrullinated vimentin. J. Clin. Invest. 122, 1791–1802 (2012).
Wigerblad, G. et al. Autoantibodies to citrullinated proteins induce joint pain independent of inflammation via a chemokine-dependent mechanism. Ann. Rheum. Dis. 75, 730–738 (2016).
Kokkonen, H. et al. Antibodies of IgG, IgA and IgM isotypes against cyclic citrullinated peptide precede the development of rheumatoid arthritis. Arthritis Res. Ther. 13, R13 (2011).
Jorgensen, C., Moynier, M., Bologna, C., Youinou, P. & Sany, J. Rheumatoid factor associated with a secretory component in rheumatoid arthritis. Br. Med. J. 34, 236–240 (1995).
Aho, K., Heliovaara, M., Maatela, J., Tuomi, T. & Palosuo, T. Rheumatoid factors antedating clinical rheumatoid arthritis. J. Rheumatol. 18, 1282–1284 (1991).
Kolfenbach, J. R. et al. A prospective approach to investigating the natural history of pre-clinical rheumatoid arthritis (RA) using first-degree relatives of probands with RA. Arthritis Care Res. 61, 1735–1741 (2009).
El-Gabalawy, H. S. et al. Familial clustering of the serum cytokine profile in the relatives of rheumatoid arthritis patients. Arthritis Rheum. 64, 1720–1729 (2012).
Deane, K. D. et al. Identification of undiagnosed inflammatory arthritis in a community health fair screen. Arthritis Care Res. 61, 1642–1649 (2009).
Lübbers, J. et al. The type I IFN signature as a biomarker of preclinical rheumatoid arthritis. Ann. Rheum. Dis. 72, 776–780 (2013).
Young, K. A. et al. Relatives without rheumatoid arthritis show reactivity to anti-citrullinated protein/peptide antibodies that are associated with arthritis-related traits: studies of the etiology of rheumatoid arthritis. Arthritis Rheum. 65, 1995–2004 (2013).
Hughes-Austin, J. et al. Multiple cytokines and chemokines are associated with rheumatoid arthritis-related autoimmunity in first-degree relatives without rheumatoid arthritis: Studies of the Aetiology of Rheumatoid Arthritis (SERA). Ann. Rheum. Dis. 72, 901–907 (2012).
Aslam, A. et al. Emergence of proinflammatory autoreactive T-cell responses in preclinical rheumatoid arthritis. Lancet 383, S22 (2014).
Hunt, L. et al. T cell subsets: an immunological biomarker to predict progression to clinical arthritis in ACPApositive individuals. Ann. Rheum. Dis. 75, 1884–1889 (2016).
Nielen, M. M. J. et al. Increased levels of C-Reactive Protein in serum from blood donors before the onset of rheumatoid arthritis. Arthritis Rheum. 50, 2423–2427 (2004).
van de Stadt, L. A. et al. The extent of the anti-citrullinated protein antibody repertoire is associated with arthritis development in patients with seropositive arthralgia. Ann. Rheum. Dis. 70, 128–133 (2011).
Arkema, E. V. et al. Anti-citrullinated peptide autoantibodies, human leukocyte antigen shared epitope and risk of future rheumatoid arthritis: a nested case–control study. Arthritis Res. Ther. 15, R159 (2013).
Bhatia, S. S. et al. Rheumatoid factor seropositivity is inversely associated with oral contraceptive use in women without arthritis. Ann. Rheum. Dis. 66, 267–269 (2007).
Hensvold, A. H. et al. Environmental and genetic factors in the development of anticitrullinated protein antibodies (ACPAs) and ACPA-positive rheumatoid arthritis: an epidemiological investigation in twins. Ann. Rheum. Dis. 74, 375–380 (2015).
van Zanten, A. et al. Presence of anticitrullinated protein antibodies in a large population-based cohort from the Netherlands. Ann. Rheum. Dis. 76, 1184–1190 (2017).
Hedström, A. K., Stawiarz, L., Klareskog, L. & Alfredsson, L. Smoking and susceptibility to rheumatoid arthritis in a Swedish population-based case-control study. Eur. J. Epidemiol. 33, 415–423 (2018).
van Wesemael, T. J. et al. Smoking is associated with the concurrent presence of multiple autoantibodies in rheumatoid arthritis rather than with anti-citrullinated protein antibodies per se: a multicenter cohort study. Arthritis Res. Ther. 18, 285 (2016).
Terao, C. et al. Effects of smoking and shared epitope on the production of anti-citrullinated peptide antibody in a Japanese adult population. Arthritis Care Res. 66, 1818–1827 (2014).
Gan, R. W. et al. Relationship between air pollution and positivity of RA-related autoantibodies in individuals without established RA: a report on SERA. Ann. Rheum. Dis. 72, 2002–2005 (2013).
Hart, J. E., Laden, F., Puett, R. C., Costenbader, K. H. & Karlson, E. W. Exposure to traffic pollution and increased risk of rheumatoid arthritis. Environ. Health Perspect. 117, 1065–1069 (2009).
Tedeschi, S. K. et al. Elevated BMI and antibodies to citrullinated proteins interact to increase rheumatoid arthritis risk and shorten time to diagnosis: a nested case-control study of women in the Nurses’ Health Studies. Semin. Arthritis Rheum. 46, 692–698 (2017).
Stolt, P. et al. Silica exposure is associated with increased risk of developing rheumatoid arthritis: results from the Swedish EIRA study. Ann. Rheum. Dis. 64, 582–586 (2005).
Dawczynski, C. et al. Docosahexaenoic acid in the treatment of rheumatoid arthritis: A double-blind, placebo-controlled, randomized cross-over study with microalgae vs. sunflower oil. Clin. Nutr. 37, 494–504 (2017).
Souza, P. R. & Norling, L. V. Implications for eicosapentaenoic acid- and docosahexaenoic acid-derived resolvins as therapeutics for arthritis. Eur. J. Pharmacol. 785, 165–173 (2016).
Di Giuseppe, D., Crippa, A., Orsini, N. & Wolk, A. Fish consumption and risk of rheumatoid arthritis: a doseresponse meta-analysis. Arthritis Res. Ther. 16, 446 (2014).
Gan, R. W. et al. Lower omega-3 fatty acids are associated with the presence of anti-cyclic citrullinated peptide autoantibodies in a population at risk for future rheumatoid arthritis: a nested case-control study. Rheumatology 55, 367–376 (2016).
Gan, R. W. et al. Omega-3 fatty acids are associated with a lower prevalence of autoantibodies in shared epitope positive subjects at risk for rheumatoid arthritis. Ann. Rheum. Dis. 76, 147–152 (2017).
Gan, R. W. et al. The association between omega-3 fatty acid biomarkers and inflammatory arthritis in an anticitrullinated protein antibody positive population. Rheumatology 56, 2229–2236 (2017).
Li, S., Yu, Y., Yue, Y., Zhang, Z. & Su, K. Microbial infection and rheumatoid arthritis. J. Clin. Cell. Immunol. 4, 174 (2013).
Lundberg, K., Wegner, N., Yucel-Lindberg, T. & Venables, P. J. Periodontitis in RA-the citrullinated enolase connection. Nat. Rev. Rheumatol. 6, 727–730 (2010).
Longman, R. S. & Littman, D. R. The functional impact of the intestinal microbiome on mucosal immunity and systemic autoimmunity. Curr. Opin. Rheum. 27, 381–387 (2015).
Scher, J. U., Littman, D. R. & Abramson, S. B. Review: Microbiome in inflammatory arthritis and human rheumatic diseases. Arthritis Rheum. 68, 35–45 (2015).
Belkaid, Y. & Hand, T. W. Role of the microbiota in immunity and inflammation. Cell 157, 121–141 (2014).
Cash, H. L., Whitham, C. V., Behrendt, C. L. & Hooper, L. V. Symbiotic bacteria direct expression of an intestinal bactericidal lectin. Science 313, 1126–1130 (2006).
Szabady, R. L. & McCormick, B. A. Control of neutrophil inflammation at mucosal surfaces by secreted epithelial products. Front. Immunol. https://doi.org/10.3389/fimmu.2013.00220 (2013).
Howson, L. J., Salio, M. & Cerundolo, V. MR1-restricted mucosal-associated invariant T cells and their activation during infectious diseases. Front. Immunol. https://doi.org/10.3389/fimmu.2015.00303 (2015).
Sonnenberg, G. F., Monticelli, L. A., Elloso, M. M., Fouser, L. A. & Artis, D. CD4(+) lymphoid tissue-inducer cells promote innate immunity in the gut. Immunity 34, 122–134 (2011).
Goto, Y. et al. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science 345, 1254009 (2014).
von Andrian, U. & Mackay, C. R. T-cell function and migration. Two sides of the same coin. New Engl. J. Med. 343, 1020–1034 (2000).
Kurmaeva, E. et al. T cell-associated a4b7 but not a4b1 integrin is required for the induction and perpetuation of chronic coliti. Mucosal Immunol. 7, 1354–1365 (2014).
Macpherson, A. J., McCoy, K. D., Johansen, F. E. & Brandtzaeg, P. The immune geography of IgA induction and function. Mucosal Immunol. 11, 11–22 (2008).
Fagarasan, S., Kawamoto, S., Kanagawa, O. & Suzuki, K. Adaptive immune regulation in the gut: T celldependent and T cell-independent IgA synthesis. Annu. Rev. Immunol. 28, 243–273 (2010).
Corthésy, B. Multi-faceted functions of secretory IgA at mucosal surfaces. Front. Immunol. https://doi.org/10.3389/fimmu.2013.00185 (2013).
Aleyd, E., Al, M., Tuk, C. W., van der Laken, C. J. & van Egmond, M. IgA complexes in plasma and synovial fluid of patients with rheumatoid arthritis induce neutrophil extracellular traps via FcαRI. J. Immunol. 197, 4552–4559 (2016).
Russell, M. W., Sibley, D. A., Nikolova, E. B., Tomana, M. & Mestecky, J. IgA antibody as a non-inflammatory regulator of immunity. Biochem. Soc. Trans. 25, 466–470 (1997).
Baker, K., Lencer, W. I. & Blumberg, R. S. Beyond IgA: the mucosal immunoglobulin alphabet. Mucosal Immunol. 3, 324–325 (2010).
Bunker, J. J. et al. Natural polyreactive IgA antibodies coat the intestinal microbiota. Science 358, eaan6619 (2017).
Vaishnava, S. et al. The antibacterial lectin RegIIIgamma promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011).
Virgin, H. W. The virome in mammalian physiology and disease. Cell 157, 142–150 (2014).
Smith, P. M. et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341, 569–573 (2013).
Kuhn, K. A. et al. Antibodies to citrullinated proteins enhance tissue injury in experimental arthritis. J. Clin. Invest. 116, 961–973 (2006).
Liu, X. et al. Role of the gut microbiome in modulating arthritis progression in mice. Sci. Rep. 6, 30594 (2016).
Jubair, W. K. et al. Modulation of inflammatory arthritis by gut microbiota through mucosal inflammation and autoantibody generation. Arthritis Rheum. 70, 1220–1233 (2018).
Jacques, P. & Elewaut, D. Joint expedition: linking gut inflammation to arthritis. Mucosal Immunol. 1, 364–371 (2008).
Ji, H. et al. Arthritis critically dependent on innate immune system players. Immunity 16, 157–168 (2002).
Block, K. E., Zheng, Z., Dent, A. L., Kee, B. L. & Huang, H. Gut microbiota regulates K/BxN autoimmune arthritis through follicular helper T but not Th17 cells. J. Immunol. 196, 1550–1557 (2016).
Wu, H. J. et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32, 815–827 (2010).
Marietta, E. V. et al. Suppression of inflammatory arthritis by human gut-derived Prevotella histicola in humanized mice. Arthritis Rheum. 68, 2878–2888 (2016).
Tan, Y. C. et al. High-throughput sequencing of natively paired antibody chains provides evidence for original antigenic sin shaping the antibody response to influenza vaccination. Clin. Immunol. Immunopathol. 151, 55–66 (2014).
Kinslow, J. D. et al. IgA plasmablasts are elevated in subjects at risk for future rheumatoid arthritis. Arthritis Rheum. 68, 2372–2383 (2016).
Tan, Y. C. et al. Barcode-enabled sequencing of plasmablast antibody repertoires in rheumatoid arthritis. Arthritis Rheum. 66, 2706–2715 (2014).
Chang, H. H. et al. A molecular timeline of preclinical rheumatoid arthritis defined by dysregulated PTPN22. JCI Insight https://doi.org/10.1172/jci.insight.90045 (2016).
Chang, H. H., Dwivedi, N., Nicholas, A. P. & Ho, I. C. The W620 polymorphism in PTPN22 disrupts its interaction with peptidylarginine deiminase type 4 and enhances citrullination and NETosis. Arthritis Rheum. 67, 2323–2334 (2015).
Yang, Z., Fujii, H., Mohan, S. V., Goronzy, J. J. & Weyand, C. M. Phosphofructokinase deficiency impairs ATP generation, autophagy, and redox balance in rheumatoid arthritis T cells. J. Exp. Med. 210, 2119–2134 (2013).
Yang, Z. et al. Restoring oxidant signaling suppresses proarthritogenic T cell effector functions in rheumatoid arthritis. Sci. Transl Med. 8, 331ra38 (2016).
Pianta, A. et al. Evidence of the immune relevance of Prevotella copri, a gut microbe, in patients With rheumatoid arthritis. Arthritis Rheum. 69, 964–975 (2017).
Rangel-Moreno, J. et al. Inducible bronchus-associated lymphoid tissue (iBALT) in patients with pulmonary complications of rheumatoid arthritis. J. Clin. Invest. 116, 3183–3194 (2006).
Reynisdottir, G. et al. Signs of immune activation and local inflammation are present in the bronchial tissue of patients with untreated early rheumatoid arthritis. Ann. Rheum. Dis. 75, 1722–1727 (2016).
Ytterberg, A. J. et al. Identification of shared citrullinated immunological targets in the lungs and joints of patients with rheumatoid arthritis. Ann. Rheum. Dis. 74, 1772–1777 (2012).
Demoruelle, M. K. et al. Airways abnormalities and rheumatoid arthritis-related autoantibodies in subjects without arthritis: Early injury or initiating site of autoimmunity? Arthritis Rheum. 64, 1756–1761 (2012).
Demoruelle, M. K. et al. Antibody responses to citrullinated and non-citrullinated antigens in the sputum of subjects with and at-risk for rheumatoid arthritis. Arthritis Rheum. 70, 516–527 (2018).
Fischer, A. et al. Lung disease with anti-CCP antibodies but not rheumatoid arthritis or connective tissue disease. Respir. Med. 106, 1040–1047 (2012).
Bingham, C. O. & Moni, M. Periodontal disease and rheumatoid arthritis: the evidence accumulates for complex pathobiologic interactions. Curr. Opin. Rheumatol. 25, 345–353 (2013).
Eriksson, K. et al. Prevalence of periodontitis in patients with established rheumatoid arthritis: a Swedish population based case-control study. PLOS One https://doi.org/10.1371/journal.pone.0155956 (2016).
Konig, M. F. et al. Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci. Transl Med. 8, 369ra176 (2017).
Mikuls, T. R. et al. Porphyromonas gingivalis and disease-related autoantibodies in individuals at increased risk of rheumatoid arthritis. Arthritis Rheum. 64, 3522–3530 (2012).
Cheng, Z. et al. The subgingival microbiomes in periodontitis and health of individuals with rheumatoid arthritis and at risk of developing rheumatoid arthritis. J. Oral Microbiol. 9, Abstr. 1325216 (2017).
Harvey, G. P. et al. Expression of peptidylarginine deiminase-2 and -4, citrullinated proteins and anticitrullinated protein antibodies in human gingiva. J. Periodontal Res. 48, 252–261 (2013).
Svärd, A., Kastbom, A., Sommarin, Y. & Skogh, T. Salivary IgA antibodies to cyclic citrullinated peptides (CCP) in rheumatoid arthritis. Immunobiology 218, 232–237 (2013).
Otten, H. G. et al. IgA rheumatoid factor in mucosal fluids and serum of patients with rheumatoid arthritis: immunological aspects and clinical significance. Clin. Exp. Immunol. 90, 256–259 (1992).
Ebringer, A. & Rashid, T. Rheumatoid arthritis is caused by a Proteus urinary tract infection. APMIS 122, 363–368 (2013).
Whiteside, S. A., Razvi, H., Dave, S., Reid, G. & Burton, J. P. The microbiome of the urinary tract — a role beyond infection. Nat. Rev. Urol. 12, 81–90 (2015).
Gosmann, C. et al. Lactobacillus-deficient cervicovaginal bacterial communities are associated with increased HIV acquisition in young south African women. Immunity 46, 29–37 (2017).
Tudor, D. et al. HIV-1 gp41-specific monoclonal mucosal IgAs derived from highly exposed but IgGseronegative individuals block HIV-1 epithelial transcytosis and neutralize CD4(+) cell infection: an IgA gene and functional analysis. Mucosal Immunol. 2, 412–426 (2009).
Khatter, S. et al. Anti-CCP antibody levels are elevated in cervicovaginal fluid in association with local inflammation in premenopausal women without RA [abstract]. Arthritis Rheum. (2017).
Hogeboom, C. Peptide motif analysis predicts alphaviruses as triggers for rheumatoid arthritis. Mol. Immunol. 68, 465–475 (2015).
van Heemst, J. et al. Crossreactivity to vinculin and microbes provides a molecular basis for HLA-based protection against rheumatoid arthritis. Nat. Commun. 6, 6681 (2015).
Cole, D. K. et al. Hotspot autoimmune T cell receptor binding underlies pathogen and insulin peptide crossreactivit. J. Clin. Invest. 126, 2191–2204 (2016).
Li, S. et al. Autoantibodies from single circulating plasmablasts react with citrullinated antigens and Porphyromonas gingivalis in rheumatoid arthritis. Arthritis Rheum. 68, 614–626 (2016).
Svärd, A. et al. Associations with smoking and shared epitope differ between IgA- and IgG-class antibodies to cyclic citrullinated peptides in early rheumatoid arthritis. Arthritis Rheum. 67, 2032–2037 (2015).
Makrygiannakis, D. et al. Smoking increases peptidylarginine deiminase 2 enzyme expression in human lungs and increases citrullination in BAL cells. Ann. Rheum. Dis. 67, 1488–1492 (2008).
Serhan, C. N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
Campbell, E. L. et al. Resolvin E1 promotes mucosal surface clearance of neutrophils: a new paradigm for inflammatory resolution. FASEB J. 21, 3162–3170 (2007).
Uddin, M. & Levy, B. D. Resolvins: natural agonists for resolution of pulmonary inflammation. Prog. Lipid Res. 50, 75–88 (2011).
Norling, L. V. et al. Proresolving and cartilage-protective actions of resolvin D1 in inflammatory arthritis. JCI Insight 1, e85922 (2016).
Donadio, J. V. & Grande, J. P. The role of fish oil/omega-3 fatty acids in the treatment of IgA nephropathy. Semin. Nephrol. 24, 225–243 (2004).
Dennis, G. J. et al. Synovial phenotypes in rheumatoid arthritis correlate with response to biologic therapeutics. Arthritis Res. Ther. 16, R90 (2014).
Sokolove, J. et al. Rheumatoid factor as a potentiator of anti-citrullinated protein antibody mediated inflammation in rheumatoid arthritis. Arthritis Rheum. 66, 813–821 (2015).
Kunwar, S., Dahal, K. & Sharma, S. Anti-IL-17 therapy in treatment of rheumatoid arthritis: a systematic literature review and meta-analysis of randomized controlled trials. Rheumatol. Intl 36, 1065–1075 (2016).
Yen, D. et al. IL-23 is essential for T cell–mediated colitis and promotes inflammation via IL-17 and IL-6. J. Clin. Invest. 116, 1310–1316 (2006).
Azizi, G., Jadidi-Niaragh, F. & Mirshafiey, A. Th17 Cells in Immunopathogenesis and treatment of rheumatoid arthritis. Intl. J. Rheum. Dis. 16, 243–253 (2013).
van der Vlist, M., Kuball, J., Radstake, T. R. & Meyaard, L. Immune checkpoints and rheumatic diseases: what can cancer immunotherapy teach us? Nat. Rev. Rheumatol. 12, 593–604 (2016).
O’Neill, L. A., Kishton, R. J. & Rathmell, J. A guide to immunometabolism for immunologists. Nat. Rev. Immunol 16, 553–565 (2016).
Acknowledgements
The work of the authors is supported by U01 AI101981 (V.M.H.), T32 AR07534 (V.M.H.), UH2 AR067681 (V.M.H.), UM1 AI110503 (V.M.H.), the Rheumatology Research Foundation (V.M.H. and K.D.), R01 AR051394 (V.M.H.), U19 AI50864 (V.M.H.), K08 DK107905 and Pfizer ASPIRE (K.A.K.). The authors appreciate the early input by R. Thomas to the conception of this Review.
Author information
Authors and Affiliations
Contributions
V.M.H. researched data, contributed to discussion of content, wrote, reviewed and edited the manuscript before submission. M.K.D., K.A.K., Y.O., J.M.N. and K.D.D. contributed to discussion of content, and reviewed and edited the manuscript before submission. J.H.B. and W.H.R. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Dysbiosis
-
A general term used to indicate a change in the normal bacterial ecology, usually with a potential for association with a disease state.
- Shared epitope
-
A set of HLA antigens that are preferentially found in patients with RA and that are characterized by conserved amino acids within the peptide-binding groove.
- Epitope spreading
-
A process in which the epitopes (target antigen shapes or sequences recognized by B and T cell receptors) that are distinct from and non-cross-reactive with the initial target epitope become major targets of an ongoing immune response.
- Avidity maturation
-
A process in which the accumulated strength of multiple affinities of individual non-covalent binding interactions between antigens and antibodies increases over time.
- NETosis
-
A unique form of neutrophil extracellular trap (NET)-associated cell death characterized by nuclear condensation and extrusion of chromatic and granular contents in a manner especially conducive to capturing pathogens.
- Molecular mimicry
-
The development of autoreactivity commonly considered to be through the recognition of a microbial or other foreign pathogen peptide that is structurally similar to self-antigen.
Rights and permissions
About this article
Cite this article
Holers, V.M., Demoruelle, M.K., Kuhn, K.A. et al. Rheumatoid arthritis and the mucosal origins hypothesis: protection turns to destruction. Nat Rev Rheumatol 14, 542–557 (2018). https://doi.org/10.1038/s41584-018-0070-0
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-018-0070-0