Abstract
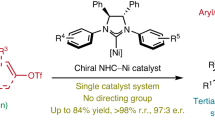
Asymmetric synthesis based on a metallate shift of tetracoordinate borons is an intriguing and challenging topic. Despite the construction of central chirality from tetracoordinate boron species via a 1,2-metallate shift, catalytic asymmetric synthesis of axially chiral compounds from such boron ‘ate’ complexes is an ongoing challenge. Axially chiral alkenes have received great attention due to their unique characteristics and intriguing molecular scaffolds. Here we report an enantioselective nickel-catalysed strategy for the construction of axially chiral alkenes via a 1,3-metallate shift of alkynyl tetracoordinate boron species. The chemoselectivity, regioselectivity and atroposelectivity can be regulated and well-controlled from readily accessible starting materials with a cheap transition-metal catalyst. Downstream transformations indicate the powerful conversion ability of such compounds in this protocol, and late-stage elaborations of bioactive compounds can also be achieved. Mechanistic experiments reveal that regioselective syn-addition of an aryl–Ni complex with a carbon–carbon triple bond and subsequent 1,3-phenyl migration are the two key steps for the synthesis of axially chiral alkenes.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the Article and its Supplementary Information. The X-ray crystallographic coordinates for structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition no. 2243354 (51). The data can be obtained free of charge from The Cambridge Crystallographic Data Centre.
References
Zweifel, G., Arzoumanian, H. & Whitney, C. C. A convenient stereoselective synthesis of substituted alkenes via hydroboration-iodination of alkynes. J. Am. Chem. Soc. 89, 3652–3653 (1967).
Ishikura, M. & Terashima, M. A new pathway into [b]-annelated indole derivatives through trialkyl(1-methyl-2-indolyl) borates. J. Chem. Soc. J. Chem. Soc. 1991, 1219–1221 (1991).
Kischkewitz, M., Okamoto, K., Mück-Lichtenfeld, C. & Studer, A. Radical-polar crossover reactions of vinylboron ate complexes. Science 355, 936–938 (2017).
Silvi, M., Sandford, C. & Aggarwal, V. K. Merging photoredox with 1,2-metallate rearrangements: the photochemical alkylation of vinyl boronate complexes. J. Am. Chem. Soc. 139, 5736–5739 (2017).
Yang, K. & Song, Q. Tetracoordinate boron intermediates enable unconventional transformations. Acc. Chem. Res. 54, 2298–2312 (2021).
Binger, P. & Koster, R. Synthesen von und mit Alkinylboranaten. Tetrahedron Lett. 6, 1901–1906 (1965).
Zu, B., Guo, Y. & He, C. Catalytic enantioselective construction of chiroptical boron stereogenic compounds. J. Am. Chem. Soc. 143, 16302–16310 (2021).
Zu, B., Guo, Y., Ren, L.-Q., Li, Y. & He, C. Catalytic enantioselective synthesis of boron-stereogenic BODIPYs. Nat. Synth. 2, 564–571 (2023).
Ma, X. et al. Modular assembly of versatile tetrasubstituted alkenyl monohalides from alkynyl tetracoordinate borons. Chem 9, 1164–1181 (2023).
Yang, Y. et al. An intramolecular coupling approach to alkyl bioisosteres for the synthesis of multisubstituted bicycloalkyl boronates. Nat. Chem. 13, 950–955 (2021).
Miyaura, N., Yamada, K. & Suzuki, A. A new stereospecific cross-coupling by the palladium-catalyzed reaction of 1-alkenylboranes with 1-alkenyl or 1-alkynyl halides. Tetrahedron Lett. 20, 3437–3440 (1979).
Miyaura, N. & Suzuki, A. Palladium-catalyzed cross-coupling reactions of organoboron compounds. Chem. Rev. 95, 2457–2483 (1995).
Han, J. et al. Pd/Xu-Phos-catalyzed asymmetric elimination of fully substituted enol triflates into axially chiral trisubstituted allenes. Sci. Adv. 9, eadg1002 (2023).
Cheng, J. K., Xiang, S.-H. & Tan, B. Organocatalytic enantioselective synthesis of axially chiral molecules: development of strategies and skeletons. Acc. Chem. Res. 55, 2920–2937 (2022).
Zhu, S., Mao, J.-H., Cheng, J. K., Xiang, S.-H. & Tan, B. Discovery and organocatalytic enantioselective construction of axially chiral cyclohexadienylidene skeletons. Chem 8, 2529–2541 (2022).
Zhou, M. et al. Asymmetric synthesis of vicinal tetrasubstituted diamines via reductive coupling of ketimines templated by chiral diborons. Angew. Chem. Int. Ed. 135, e202300334 (2023).
Zhu, Y., Dong, W. & Tang, W. Palladium-catalyzed cross-couplings in the synthesis of agrochemicals. Adv. Agrochem. 1, 125–138 (2022).
Sun, J., Yang, H. & Tang, W. Recent advances in total syntheses of complex dimeric natural products. Chem. Soc. Rev. 50, 2320–2336 (2021).
Matteson, D. S. Boronic esters in asymmetric synthesis. J. Org. Chem. 78, 10009–10023 (2013).
Matteson, D. S. & Ray, R. Directed chiral synthesis with pinanediol boronic esters. J. Am. Chem. Soc. 102, 7590–7591 (1980).
Stymiest, J. L., Dutheuil, G., Mahmood, A. & Aggarwal, V. K. Lithiated carbamates: chiral carbenoids for iterative homologation of boranes and boronic esters. Angew. Chem. Int. Ed. 46, 7491–7494 (2007).
Stymiest, J. L., Bagutski, V., French, R. & Aggarwal, V. K. Enantiodivergent conversion of chiral secondary alcohols into tertiary alcohols. Nature 456, 778–782 (2008).
Beckmann, E., Desai, V. & Hoppe, D. Stereospecific reaction of α-carbamoyloxy-2-alkenylboronates and α-carbamoyloxy-alkylboronates with Grignard reagents—synthesis of highly enantioenriched secondary alcohols. Synlett 13, 2275–2280 (2004).
Tao, Z., Robb, K. A., Panger, J. L. & Denmark, S. E. Enantioselective, Lewis base-catalyzed carbosulfenylation of alkenylboronates by 1,2-boronate migration. J. Am. Chem. Soc. 140, 15621–15625 (2018).
Fairchild, M. E., Noble, A. & Aggarwal, V. K. Diastereodivergent synthesis of cyclopentyl boronic esters bearing contiguous fully substituted stereocenters. Angew. Chem. Int. Ed. 61, e202205816 (2022).
Fasano, V., Mykura, R. C., Fordham, J. M. & Aggarwal, V. K. Automated stereocontrolled assembly-line synthesis of organic molecules. Nat. Synth. 1, 902–907 (2022).
Yeung, K., Mykura, R. C. & Aggarwal, V. K. Lithiation-borylation methodology in the total synthesis of natural products. Nat. Synth. 1, 117–126 (2022).
Bootwicha, T., Feilner, J. M., Myers, E. L. & Aggarwal, V. K. Iterative assembly line synthesis of polypropionates with full stereocontrol. Nat. Chem. 9, 896–902 (2017).
Brown, H. C. & Zweifel, G. Hydroboration. IX. The hydroboration of cyclic and bicyclic olefins-stereochemistry of the hydroboration reaction. J. Am. Chem. Soc. 83, 2544–2551 (1961).
Sandford, C. & Aggarwal, V. K. Stereospecific functionalizations and transformations of secondary and tertiary boronic esters. Chem. Commun. 53, 5481–5494 (2017).
Zhang, L. et al. Catalytic conjunctive cross-coupling enabled by metal-induced metallate rearrangement. Science 351, 70–74 (2016).
Chierchia, M., Law, C. & Morken, J. P. Nickel-catalyzed enantioselective conjunctive cross-coupling of 9-BBN borates. Angew. Chem. Int. Ed. 56, 11870–11874 (2017).
Chierchia, M., Xu, P., Lovinger, G. J. & Morken, J. P. Enantioselective radical addition/cross-coupling of organozinc reagents, alkyl iodides and alkenyl boron reagents. Angew. Chem. Int. Ed. 58, 14245–14249 (2019).
Namirembe, S. & Morken, J. P. Reactions of organoboron compounds enabled by catalyst-promoted metalate shifts. Chem. Soc. Rev. 48, 3464–3474 (2019).
Panda, S. & Ready, J. M. Palladium-catalyzed asymmetric three-component coupling of boronic esters, indoles and allylic acetates. J. Am. Chem. Soc. 139, 6038–6041 (2017).
Davis, C. R., Fu, Y., Liu, P. & Ready, J. M. Mechanistic basis for the iridium-catalyzed enantioselective allylation of alkenyl boronates. J. Am. Chem. Soc. 144, 16118–16130 (2022).
Davis, C. R., Luvaga, I. K. & Ready, J. M. Enantioselective allylation of alkenyl boronates promotes a 1,2-metalate rearrangement with 1,3-diastereocontrol. J. Am. Chem. Soc. 143, 4921–4927 (2021).
Sharma, H. A., Essman, J. Z. & Jacobsen, E. N. Enantioselective catalytic 1,2-boronate rearrangements. Science 374, 752–757 (2021).
Matteson, D. S. α-Halo boronic esters in asymmetric synthesis. Tetrahedron 54, 10555–10607 (1998).
Jonker, S. J. T. et al. Organocatalytic synthesis of α-trifluoromethyl allylboronic acids by enantioselective 1,2-borotropic migration. J. Am. Chem. Soc. 142, 21254–21259 (2020).
Wang, Q., Eriksson, L. & Szabó, K. J. Catalytic homologation-allylboration sequence for diastereo- and enantioselective synthesis of densely functionalized β-fluorohydrins with tertiary fluoride stereocenters. Angew. Chem. Int. Ed. 62, e202301481 (2023).
Wu, S. et al. Urea group-directed organocatalytic asymmetric versatile dihalogenation of alkenes and alkynes. Nat. Catal. 4, 692–702 (2021).
Miao, J.-H. et al. Organocatalyst-controlled site-selective arene C–H functionalization. Nat. Chem. 13, 982–991 (2021).
Cheng, J. K., Xiang, S.-H., Li, S., Ye, L. & Tan, B. Recent advances in catalytic asymmetric construction of atropisomers. Chem. Rev. 121, 4805–4902 (2021).
Wencel-Delord, J., Panossian, A., Lerouxb, F. R. & Colobert, F. Recent advances and new concepts for the synthesis of axially stereoenriched biaryls. Chem. Soc. Rev. 44, 3418–3430 (2015).
Wu, S., Xiang, S.-H., Cheng, J. K. & Tan, B. Axially chiral alkenes: atroposelective synthesis and applications. Tetrahedron Chem. 1, 100009 (2022).
Mori, K., Ohmori, K. & Suzuki, K. Hydrogen-bond control in axially chiral styrenes: selective synthesis of enantiomerically pure C2-symmetric paracyclophanes. Angew. Chem. Int. Ed. 48, 5638–5641 (2009).
Mori, K., Ohmori, K. & Suzuki, K. Stereochemical relay via axially chiral styrenes: asymmetric synthesis of the antibiotic TAN-1085. Angew. Chem. Int. Ed. 48, 5633–5637 (2009).
Song, H. et al. Synthesis of axially chiral styrenes through Pd-catalyzed asymmetric C-H olefination enabled by an amino amide transient directing group. Angew. Chem. Int. Ed. 59, 6576–6580 (2020).
Yang, C. et al. Facile synthesis of axially chiral styrene-type carboxylic acids via palladium-catalyzed asymmetric C-H activation. Chem. Sci. 12, 3726–3732 (2021).
Yang, C. et al. Development of axially chiral styrene-type carboxylic acid ligands via palladium-catalyzed asymmetric C-H alkynylation. Org. Lett. 23, 8132–8137 (2021).
Feng, X. & Du, H. Synthesis of chiral olefin ligands and their application in asymmetric catalysis. Asian J. Org. Chem. 1, 204–213 (2012).
Wang, Y.-B. et al. Rational design, enantioselective synthesis and catalytic applications of axially chiral EBINOLs. Nat. Catal. 2, 504–513 (2019).
Zheng, S.-C. et al. Organocatalytic atroposelective synthesis of axially chiral styrenes. Nat. Commun. 8, 15238 (2017).
Li, W. et al. Synthesis of axially chiral alkenylboronates through combined copper- and palladium-catalysed atroposelective arylboration of alkynes. Nat. Synth. 2, 140–151 (2023).
Jolliffe, J., Armstrong, R. & Smith, M. Catalytic enantioselective synthesis of atropisomeric biaryls by a cation-directed O-alkylation. Nat. Chem. 9, 558–562 (2017).
Jin, L. et al. Atroposelective synthesis of axially chiral styrenes via an asymmetric C-H functionalization strategy. Chem 6, 497–511 (2020).
Ishida, N., Miura, T. & Murakami, M. Stereoselective synthesis of trisubstituted alkenylboranes by palladium-catalysed reaction of alkynyltriarylborates with aryl halides. Chem. Commun. 2007, 4381–4383 (2007).
Kuang, Z. et al. Cu-catalyzed regio- and stereodivergent chemoselective sp2/sp3 1,3- and 1,4-diborylations of CF3-containing 1,3-enynes. Chem 6, 2347–2363 (2020).
Yang, K., Zhang, G. & Song, Q. Four-coordinate triarylborane synthesis via cascade B-Cl/C-B cross-metathesis and C-H bond borylation. Chem. Sci. 9, 7666–7672 (2018).
Jin, S., Liu, K., Wang, S. & Song, Q. Enantioselective cobalt-catalyzed cascade hydrosilylation and hydroboration of alkynes to access enantioenriched 1,1-silylboryl alkanes. J. Am. Chem. Soc. 143, 13124–13134 (2021).
Zhang, G. et al. Construction of boron-stereogenic compounds via enantioselective Cu-catalyzed desymmetric B–H bond insertion reaction. Nat. Commun. 13, 2624–2635 (2022).
Li, C. et al. Photo-induced trifunctionalization of bromostyrenes via remote radical migration reactions of tetracoordinate boron species. Nat. Commun. 13, 1748–1760 (2022).
Fan, Z. et al. Enantioselective copper-catalyzed sp2/sp3 diborylation of 1,1-chloro-trifluoromethyl-2-alkenes. ACS Cent. Sci. 8, 1134–1144 (2022).
Hong, D., Yang, Y. Y., Wang, Y. G. & Lin, X. F. A Yb(OTf)3/PEG-supported quaternary ammonium salt catalyst system for a three-component Mannich-type reaction in aqueous media. Synlett 7, 1107–1110 (2009).
Ariki, Z. T., Maekawa, Y., Nambo, M. & Crudden, C. M. Preparation of quaternary centers via nickel-catalyzed Suzuki-Miyaura cross-coupling of tertiary sulfones. J. Am. Chem. Soc. 140, 78–81 (2018).
Teng, F. et al. Palladium-catalyzed atroposelective coupling-cyclization of 2 isocyanobenzamides to construct axially chiral 2 aryl- and 2,3-diarylquinazolinones. J. Am. Chem. Soc. 143, 2722–2728 (2021).
Wagner, C. L., Herrera, G., Lin, Q., Hu, C. T. & Diao, T. Redox activity of pyridine-oxazoline ligands in the stabilization of low-valent organonickel radical complexes. J. Am. Chem. Soc. 143, 5295–5300 (2021).
Ye, M. et al. Arylation of terminal alkynes: transition-metal-free Sonogashira type coupling for the construction of C(sp)-C(sp2) bonds. Org. Lett. 25, 1787–1792 (2023).
Ishida, N., Narumi, M. & Murakami, M. Synthesis of azaaromatic-borane intramolecular complexes by palladium-catalyzed reaction of azaaromatic halides with alkynyltriarylborates. Helv. Chim. Acta 95, 2474–2480 (2012).
Hu, L. et al. Origin of ligand effects on stereoinversion in Pd-catalyzed synthesis of tetrasubstituted olefins. J. Org. Chem. 86, 18128–18138 (2021).
Acknowledgements
Financial support from the National Natural Science Foundation of China (grants 21931013 and 22271105 to Q.S.) and the Natural Science Foundation of Fujian Province (grant 2022J02009 to Q.S.) is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
Q.S. designed and directed the project. X.M. performed the experiments and developed the reactions. M.T., L.L., Z.Z., P.L. and J.L. helped with the collection of some experimental data. Q.S. and X.M. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Chuan He and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–16, Figs. 1–8 and starting material preparation, experimental procedures, synthetic transformations, mechanistic studies and product characterization.
Supplementary Data 1
Crystallographic data of compound 51, CCDC reference 2243354.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, X., Tan, M., Li, L. et al. Ni-catalysed assembly of axially chiral alkenes from alkynyl tetracoordinate borons via 1,3-metallate shift. Nat. Chem. 16, 42–53 (2024). https://doi.org/10.1038/s41557-023-01396-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01396-7
This article is cited by
-
Boron-enabled 1,3-metallate shift towards axially chiral alkenes
Nature Chemistry (2024)
-
Chemo-, regio- and stereoselective access to polysubstituted 1,3-dienes via Nickel-catalyzed four-component reactions
Nature Communications (2024)