Key Points
-
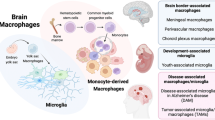
Microglia are a unique lineage of tissue macrophages that are distinct from any other type of myeloid cell inside and outside the brain. They originate exclusively from erythromyeloid precursors in the yolk sac. Microglia are long-lived and self-renew to ensure cell expansion.
-
Microglia can be distinguished from other myeloid cells in the brain by characteristic gene expression profiles. The molecules CSF1R (colony-stimulating factor 1 receptor), DAP12 (DNAX-activation protein 12), IRF8 (interferon regulatory factor 8) and transcription factor PU.1 are essential for the development and activity of microglia.
-
Microglia are vital for normal brain function and sensitive to degeneration. Microglial dysfunction can cause neuropsychiatric diseases that we have named microgliopathies.
-
Therapeutic benefit in neurological and psychiatric disorders can come from targeting bone marrow-derived myeloid cells to the CNS. Preclinical evidence is provided in animal models of Alzheimer's disease and Rett syndrome.
-
Innate immune cells in the CNS (microglia, monocytes, macrophages and dendritic cells) show complex interactions in response to pathogens, tissue damage and lymphocyte interactions, and reprogramme their function in an adaptive process termed polarization.
Abstract
Mononuclear phagocytic cells in the CNS used to be defined according to their anatomical location and surface marker expression. Recently, this concept has been challenged by the results of developmental and gene expression profiling studies that have used novel molecular biological tools to unravel the origin of microglia and to define their role as specialized tissue macrophages with long lifespans. Here, we describe how these results have redefined microglia and helped us to understand how different myeloid cell populations operate in the CNS based on their cell-specific gene expression signatures, distinct ontogeny and differential functions. Moreover, we describe the vulnerability of microglia to dysfunction and propose that myelomonocytic cells might be used in the treatment of neurological and psychiatric disorders that are characterized by primary or secondary 'microgliopathy'.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Prinz, M., Priller, J., Sisodia, S. S. & Ransohoff, R. M. Heterogeneity of CNS myeloid cells and their roles in neurodegeneration. Nature Neurosci. 14, 1227–1235 (2011).
Gomez, P. E., Schulz, C. & Geissmann, F. Development and homeostasis of “resident” myeloid cells: the case of the microglia. Glia 61, 112–120 (2013).
Geissmann, F. et al. Development of monocytes, macrophages, and dendritic cells. Science 327, 656–661 (2010).
Sieweke, M. H. & Allen, J. E. Beyond stem cells: self-renewal of differentiated macrophages. Science 342, 1242974 (2013).
Prinz, M. & Mildner, A. Microglia in the CNS: immigrants from another world. Glia 59, 177–187 (2011).
Gautier, E. L. et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nature Immunol. 13, 1118–1128 (2012).
Chow, A., Brown, B. D. & Merad, M. Studying the mononuclear phagocyte system in the molecular age. Nature Rev. Immunol. 11, 788–798 (2011).
Davies, L. C., Jenkins, S. J., Allen, J. E. & Taylor, P. R. Tissue-resident macrophages. Nature Immunol. 14, 986–995 (2013).
Butovsky, O. et al. Modulating inflammatory monocytes with a unique microRNA gene signature ameliorates murine ALS. J. Clin. Invest. 122, 3063–3087 (2012).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nature Neurosci. 16, 273–280 (2013).
Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science 336, 86–90 (2012).
Goldmann, T. et al. A new type of microglia gene targeting shows TAK1 to be pivotal in CNS autoimmune inflammation. Nature Neurosci. 16, 1618–1626 (2013). References 10–13 use fate-mapping and genetic tools to show that microglia are derived from the yolk sac.
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013). This study establishes a new line of transgenic mice in which Cre is expressed specifically in microglia.
Parkhurst, C. N. et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155, 1596–1609 (2013).
Prinz, M. et al. Distinct and nonredundant in vivo functions of IFNAR on myeloid cells limit autoimmunity in the central nervous system. Immunity 28, 675–686 (2008).
Heppner, F. L. et al. Experimental autoimmune encephalomyelitis repressed by microglial paralysis. Nature Med. 11, 146–152 (2005).
Pfrieger, F. W. & Slezak, M. Genetic approaches to study glial cells in the rodent brain. Glia 60, 681–701 (2012).
Ding, Z. et al. Antiviral drug ganciclovir is a potent inhibitor of microglial proliferation and neuroinflammation. J. Exp. Med. 211, 189–198 (2014).
Blank, T. & Prinz, M. Microglia as modulators of cognition and neuropsychiatric disorders. Glia 61, 62–70 (2013).
Priller, J. in Neuroglia 3rd edn (eds Kettenmann, H. & Ransom, B. R.) 906–916 (Oxford Univ. Press, 2013).
Hickey, W. F. & Kimura, H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science 239, 290–292 (1988).
Hickey, W. F., Vass, K. & Lassmann, H. Bone marrow-derived elements in the central nervous system: an immunohistochemical and ultrastructural survey of rat chimeras. J. Neuropathol. Exp. Neurol. 51, 246–256 (1992).
Bertrand, J. Y. et al. Three pathways to mature macrophages in the early mouse yolk sac. Blood 106, 3004–3011 (2005).
Cumano, A. & Godin, I. Ontogeny of the hematopoietic system. Annu. Rev. Immunol. 25, 745–785 (2007).
Alliot, F., Lecain, E., Grima, B. & Pessac, B. Microglial progenitors with a high proliferative potential in the embryonic and adult mouse brain. Proc. Natl Acad. Sci. USA 88, 1541–1545 (1991).
Alliot, F., Godin, I. & Pessac, B. Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res. Dev. Brain Res. 117, 145–152 (1999).
Ashwell, K. The distribution of microglia and cell death in the fetal rat forebrain. Brain Res. Dev. Brain Res. 58, 1–12 (1991).
Lawson, L. J., Perry, V. H. & Gordon, S. Turnover of resident microglia in the normal adult mouse brain. Neuroscience 48, 405–415 (1992).
Hutchins, K. D., Dickson, D. W., Rashbaum, W. K. & Lyman, W. D. Localization of morphologically distinct microglial populations in the developing human fetal brain: implications for ontogeny. Brain Res. Dev. Brain Res. 55, 95–102 (1990).
Rezaie, P. & Male, D. Colonisation of the developing human brain and spinal cord by microglia: a review. Microsc. Res. Tech. 45, 359–382 (1999).
Rezaie, P., Dean, A., Male, D. & Ulfig, N. Microglia in the cerebral wall of the human telencephalon at second trimester. Cereb. Cortex 15, 938–949 (2005).
Esiri, M. M., al Izzi, M. S. & Reading, M. C. Macrophages, microglial cells, and HLA-DR antigens in fetal and infant brain. J. Clin. Pathol. 44, 102–106 (1991).
Verney, C., Monier, A., Fallet-Bianco, C. & Gressens, P. Early microglial colonization of the human forebrain and possible involvement in periventricular white-matter injury of preterm infants. J. Anat. 217, 436–448 (2010).
Kierdorf, K. & Prinz, M. Factors regulating microglia activation. Front. Cell Neurosci. 7, 44 (2013).
Ashwell, K. Microglia and cell death in the developing mouse cerebellum. Brain Res. Dev. Brain Res. 55, 219–230 (1990).
Chan, W. Y., Kohsaka, S. & Rezaie, P. The origin and cell lineage of microglia: new concepts. Brain Res. Rev. 53, 344–354 (2007).
Lichanska, A. M. & Hume, D. A. Origins and functions of phagocytes in the embryo. Exp. Hematol. 28, 601–611 (2000).
Dai, X. M. et al. Targeted disruption of the mouse colony-stimulating factor 1 receptor gene results in osteopetrosis, mononuclear phagocyte deficiency, increased primitive progenitor cell frequencies, and reproductive defects. Blood 99, 111–120 (2002).
Erblich, B., Zhu, L., Etgen, A. M., Dobrenis, K. & Pollard, J. W. Absence of colony stimulation factor-1 receptor results in loss of microglia, disrupted brain development and olfactory deficits. PLoS ONE 6, e26317 (2011).
Wegiel, J. et al. Reduced number and altered morphology of microglial cells in colony stimulating factor-1-deficient osteopetrotic op/op mice. Brain Res. 804, 135–139 (1998).
Yoshida, H. et al. The murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor gene. Nature 345, 442–444 (1990).
Pixley, F. J. & Stanley, E. R. CSF-1 regulation of the wandering macrophage: complexity in action. Trends Cell Biol. 14, 628–638 (2004).
Metcalf, D. The granulocyte-macrophage colony stimulating factors. Cell 43, 5–6 (1985).
Lagasse, E. & Weissman, I. L. Enforced expression of Bcl-2 in monocytes rescues macrophages and partially reverses osteopetrosis in op/op mice. Cell 89, 1021–1031 (1997).
Nakahata, T., Gross, A. J. & Ogawa, M. A stochastic model of self-renewal and commitment to differentiation of the primitive hemopoietic stem cells in culture. J. Cell. Physiol. 113, 455–458 (1982).
Lin, H. et al. Discovery of a cytokine and its receptor by functional screening of the extracellular proteome. Science 320, 807–811 (2008).
Wei, S. et al. Functional overlap but differential expression of CSF-1 and IL-34 in their CSF-1 receptor-mediated regulation of myeloid cells. J. Leukoc. Biol. 88, 495–505 (2010).
Greter, M. et al. Stroma-derived interleukin-34 controls the development and maintenance of langerhans cells and the maintenance of microglia. Immunity 37, 1050–1060 (2012).
Wang, Y. et al. IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia. Nature Immunol. 13, 753–760 (2012).
Herbomel, P., Thisse, B. & Thisse, C. Zebrafish early macrophages colonize cephalic mesenchyme and developing brain, retina, and epidermis through a M-CSF receptor-dependent invasive process. Dev. Biol. 238, 274–288 (2001).
Otero, K. et al. Macrophage colony-stimulating factor induces the proliferation and survival of macrophages via a pathway involving DAP12 and β-catenin. Nature Immunol. 10, 734–743 (2009).
Rosenbauer, F. & Tenen, D. G. Transcription factors in myeloid development: balancing differentiation with transformation. Nature Rev. Immunol. 7, 105–117 (2007).
McKercher, S. R. et al. Targeted disruption of the PU.1 gene results in multiple hematopoietic abnormalities. EMBO J. 15, 5647–5658 (1996).
Beers, D. R. et al. Wild-type microglia extend survival in PU.1 knockout mice with familial amyotrophic lateral sclerosis. Proc. Natl Acad. Sci. USA 103, 16021–16026 (2006).
Zusso, M. et al. Regulation of postnatal forebrain amoeboid microglial cell proliferation and development by the transcription factor Runx1. J. Neurosci. 32, 11285–11298 (2012).
Jin, H. et al. Runx1 regulates embryonic myeloid fate choice in zebrafish through a negative feedback loop inhibiting Pu.1 expression. Blood 119, 5239–5249 (2012).
Holtschke, T. et al. Immunodeficiency and chronic myelogenous leukemia-like syndrome in mice with a targeted mutation of the ICSBP gene. Cell 87, 307–317 (1996).
Masuda, T. et al. IRF8 is a critical transcription factor for transforming microglia into a reactive phenotype. Cell Rep. 1, 334–340 (2012).
Horiuchi, M. et al. Interferon regulatory factor 8/interferon consensus sequence binding protein is a critical transcription factor for the physiological phenotype of microglia. J. Neuroinflammation 9, 227 (2012).
Minten, C., Terry, R., Deffrasnes, C., King, N. J. & Campbell, I. L. IFN regulatory factor 8 is a key constitutive determinant of the morphological and molecular properties of microglia in the CNS. PLoS ONE 7, e49851 (2012).
Hashimoto, D., Miller, J. & Merad, M. Dendritic cell and macrophage heterogeneity in vivo. Immunity 35, 323–335 (2011).
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nature Neurosci. 8, 752–758 (2005).
Sieger, D., Moritz, C., Ziegenhals, T., Prykhozhij, S. & Peri, F. Long-range Ca2+ waves transmit brain-damage signals to microglia. Dev. Cell 22, 1138–1148 (2012).
Hanisch, U. K. & Kettenmann, H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nature Neurosci. 10, 1387–1394 (2007).
Biswas, S. K. & Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nature Immunol. 11, 889–896 (2010).
Roca, H. et al. CCL2 and interleukin-6 promote survival of human CD11b+ peripheral blood mononuclear cells and induce M2-type macrophage polarization. J. Biol. Chem. 284, 34342–34354 (2009).
Hesse, M. et al. Differential regulation of nitric oxide synthase-2 and arginase-1 by type 1/type 2 cytokines in vivo: granulomatous pathology is shaped by the pattern of L-arginine metabolism. J. Immunol. 167, 6533–6544 (2001).
Wynn, T. A. Fibrotic disease and the TH1/TH2 paradigm. Nature Rev. Immunol. 4, 583–594 (2004).
David, S. & Kroner, A. Repertoire of microglial and macrophage responses after spinal cord injury. Nature Rev. Neurosci. 12, 388–399 (2011).
Kim, H. J. et al. Type 2 monocyte and microglia differentiation mediated by glatiramer acetate therapy in patients with multiple sclerosis. J. Immunol. 172, 7144–7153 (2004).
Durafourt, B. A. et al. Comparison of polarization properties of human adult microglia and blood-derived macrophages. Glia 60, 717–727 (2012).
Kigerl, K. A. et al. Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J. Neurosci. 29, 13435–13444 (2009).
Hu, X. et al. Microglia/macrophage polarization dynamics reveal novel mechanism of injury expansion after focal cerebral ischemia. Stroke 43, 3063–3070 (2012).
Wang, G. et al. Microglia/macrophage polarization dynamics in white matter after traumatic brain injury. J. Cereb. Blood Flow Metab. 33, 1864–1874 (2013).
Choi, S. H., Aid, S., Kim, H. W., Jackson, S. H. & Bosetti, F. Inhibition of NADPH oxidase promotes alternative and anti-inflammatory microglial activation during neuroinflammation. J. Neurochem. 120, 292–301 (2012).
Komohara, Y., Ohnishi, K., Kuratsu, J. & Takeya, M. Possible involvement of the M2 anti-inflammatory macrophage phenotype in growth of human gliomas. J. Pathol. 216, 15–24 (2008).
Pyonteck, S. M. et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nature Med. 19, 1264–1272 (2013).
Dal, B. A. et al. Multiple sclerosis and Alzheimer's disease. Ann. Neurol. 63, 174–183 (2008).
Vogel, D. Y. et al. Macrophages in inflammatory multiple sclerosis lesions have an intermediate activation status. J. Neuroinflammation 10, 35 (2013).
Takahashi, K., Prinz, M., Stagi, M., Chechneva, O. & Neumann, H. TREM2-transduced myeloid precursors mediate nervous tissue debris clearance and facilitate recovery in an animal model of multiple sclerosis. PLoS Med. 4, e124 (2007).
Guerreiro, R. et al. TREM2 variants in Alzheimer's disease. N. Engl. J. Med. 368, 117–127 (2013).
Heneka, M. T. et al. NLRP3 is activated in Alzheimer's disease and contributes to pathology in APP/PS1 mice. Nature 493, 674–678 (2012).
Moore, C. S. et al. miR-155 as a multiple sclerosis-relevant regulator of myeloid cell polarization. Ann. Neurol. 74, 709–720 (2013).
Wang, Y. et al. Transforming growth factor beta-activated kinase 1 (TAK1)-dependent checkpoint in the survival of dendritic cells promotes immune homeostasis and function. Proc. Natl Acad. Sci. USA 109, E343–E352 (2012).
Miron, V. E. et al. M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nature Neurosci. 16, 1211–1218 (2013).
Mikita, J. et al. Altered M1/M2 activation patterns of monocytes in severe relapsing experimental rat model of multiple sclerosis. Amelioration of clinical status by M2 activated monocyte administration. Mult. Scler. 17, 2–15 (2011).
Chiu, I. M. et al. A neurodegeneration-specific gene-expression signature of acutely isolated microglia from an amyotrophic lateral sclerosis mouse model. Cell Rep. 4, 385–401 (2013).
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nature Neurosci. 10, 1538–1543 (2007).
Ajami, B., Bennett, J. L., Krieger, C., McNagny, K. M. & Rossi, F. M. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nature Neurosci. 14, 1142–1149 (2011).
Giovanoli, S. et al. Stress in puberty unmasks latent neuropathological consequences of prenatal immune activation in mice. Science 339, 1095–1099 (2013).
Lazic, S. E. Comment on “Stress in puberty unmasks latent neuropathological consequences of prenatal immune activation in mice”. Science 340, 811 (2013).
Roumier, A. et al. Impaired synaptic function in the microglial KARAP/DAP12-deficient mouse. J. Neurosci. 24, 11421–11428 (2004).
Wake, H., Moorhouse, A. J., Jinno, S., Kohsaka, S. & Nabekura, J. Resting microglia directly monitor the functional state of synapses in vivo and determine the fate of ischemic terminals. J. Neurosci. 29, 3974–3980 (2009).
Paolicelli, R. C. et al. Synaptic pruning by microglia is necessary for normal brain development. Science 333, 1456–1458 (2011).
Tremblay, M. E., Lowery, R. L. & Majewska, A. K. Microglial interactions with synapses are modulated by visual experience. PLoS Biol. 8, e1000527 (2010).
Schafer, D. P. et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74, 691–705 (2012).
Schafer, D. P. & Stevens, B. Phagocytic glial cells: sculpting synaptic circuits in the developing nervous system. Curr. Opin. Neurobiol. 23, 1034–1040 (2013).
Hickman, S. E. et al. The microglial sensome revealed by direct RNA sequencing. Nature Neurosci. 16, 1896–1905 (2013).
Paloneva, J. et al. Mutations in two genes encoding different subunits of a receptor signaling complex result in an identical disease phenotype. Am. J. Hum. Genet. 71, 656–662 (2002).
Mosher, K. I. & Wyss-Coray, T. Microglial dysfunction in brain aging and Alzheimer's disease. Biochem. Pharmacol. http://dx.doi.org/10.1016/j.bcp.2014.01.008 (2014).
Hollingworth, P. et al. Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer's disease. Nature Genet. 43, 429–435 (2011).
Naj, A. C. et al. Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer's disease. Nature Genet. 43, 436–441 (2011).
Griciuc, A. et al. Alzheimer's disease risk gene CD33 inhibits microglial uptake of amyloid beta. Neuron 78, 631–643 (2013).
Guerreiro, R. J. et al. Using exome sequencing to reveal mutations in TREM2 presenting as a frontotemporal dementia-like syndrome without bone involvement. JAMA Neurol. 70, 78–84 (2013).
Rademakers, R. et al. Mutations in the colony stimulating factor 1 receptor (CSF1R) gene cause hereditary diffuse leukoencephalopathy with spheroids. Nature Genet. 44, 200–205 (2012). The first description of a primary 'microgliopathy' in humans.
De Jager, P. L. et al. Meta-analysis of genome scans and replication identify CD6, IRF8 and TNFRSF1A as new multiple sclerosis susceptibility loci. Nature Genet. 41, 776–782 (2009).
International Multiple Sclerosis Genetics Consortium. The genetic association of variants in CD6, TNFRSF1A and IRF8 to multiple sclerosis: a multicenter case-control study. PLoS ONE 6, e18813 (2011).
Geissmann, F., Jung, S. & Littman, D. R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 (2003).
Carlin, L. M. et al. Nr4a1-dependent Ly6Clow monocytes monitor endothelial cells and orchestrate their disposal. Cell 153, 362–375 (2013).
Michaud, J. P., Bellavance, M. A., Prefontaine, P. & Rivest, S. Real-time in vivo imaging reveals the ability of monocytes to clear vascular amyloid β. Cell Rep. 5, 646–653 (2013).
Mildner, A. et al. Distinct and non-redundant roles of microglia and myeloid subsets in mouse models of Alzheimer's disease. J. Neurosci. 31, 11159–11171 (2011).
Hawkes, C. A. & McLaurin, J. Selective targeting of perivascular macrophages for clearance of β-amyloid in cerebral amyloid angiopathy. Proc. Natl Acad. Sci. USA 106, 1261–1266 (2009).
Fiala, M. et al. Ineffective phagocytosis of amyloid-beta by macrophages of Alzheimer's disease patients. J. Alzheimers. Dis. 7, 221–232 (2005).
Fiala, M. et al. Innate immunity and transcription of MGAT-III and Toll-like receptors in Alzheimer's disease patients are improved by bisdemethoxycurcumin. Proc. Natl Acad. Sci. USA 104, 12849–12854 (2007).
Saunders, A. M. et al. Association of apolipoprotein E allele ɛ 4 with late-onset familial and sporadic Alzheimer's disease. Neurology 43, 1467–1472 (1993).
Jiang, Q. et al. ApoE promotes the proteolytic degradation of Aβ. Neuron 58, 681–693 (2008).
Lucin, K. M. et al. Microglial beclin 1 regulates retromer trafficking and phagocytosis and is impaired in Alzheimer's disease. Neuron 79, 873–886 (2013).
Krabbe, G. et al. Functional impairment of microglia coincides with β-amyloid deposition in mice with Alzheimer-like pathology. PLoS ONE 8, e60921 (2013).
Yan, S. D. et al. RAGE and amyloid-β peptide neurotoxicity in Alzheimer's disease. Nature 382, 685–691 (1996).
Xie, Z. et al. Peroxynitrite mediates neurotoxicity of amyloid β-peptide1–42- and lipopolysaccharide-activated microglia. J. Neurosci. 22, 3484–3492 (2002).
Grathwohl, S. A. et al. Formation and maintenance of Alzheimer's disease β-amyloid plaques in the absence of microglia. Nature Neurosci. 12, 1361–1363 (2009).
Doody, R. S. et al. Phase 3 trials of solanezumab for mild-to-moderate Alzheimer's disease. N. Engl. J. Med. 370, 311–321 (2014).
Salloway, S. et al. Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer's disease. N. Engl. J. Med. 370, 322–333 (2014).
Holmes, C. et al. Long-term effects of Aβ42 immunisation in Alzheimer's disease: follow-up of a randomised, placebo-controlled phase I trial. Lancet 372, 216–223 (2008).
Shechter, R. et al. Infiltrating blood-derived macrophages are vital cells playing an anti-inflammatory role in recovery from spinal cord injury in mice. PLoS Med. 6, e1000113 (2009).
Popovich, P. G. et al. Depletion of hematogenous macrophages promotes partial hindlimb recovery and neuroanatomical repair after experimental spinal cord injury. Exp. Neurol. 158, 351–365 (1999).
Shechter, R. et al. Recruitment of beneficial M2 macrophages to injured spinal cord is orchestrated by remote brain choroid plexus. Immunity 38, 555–569 (2013).
King, I. L., Dickendesher, T. L. & Segal, B. M. Circulating Ly-6C+ myeloid precursors migrate to the CNS and play a pathogenic role during autoimmune demyelinating disease. Blood 113, 3190–3197 (2009).
Mildner, A. et al. CCR2+Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain 132, 2487–2500 (2009).
Boche, D., Denham, N., Holmes, C. & Nicoll, J. A. Neuropathology after active Aβ42 immunotherapy: implications for Alzheimer's disease pathogenesis. Acta Neuropathol. 120, 369–384 (2010).
Priller, J. et al. Targeting gene-modified hematopoietic cells to the central nervous system: use of green fluorescent protein uncovers microglial engraftment. Nature Med. 7, 1356–1361 (2001).
Djukic, M. et al. Circulating monocytes engraft in the brain, differentiate into microglia and contribute to the pathology following meningitis in mice. Brain 129, 2394–2403 (2006).
Priller, J. et al. Early and rapid engraftment of bone marrow-derived microglia in scrapie. J. Neurosci. 26, 11753–11762 (2006).
Simard, A. R. & Rivest, S. Bone marrow stem cells have the ability to populate the entire central nervous system into fully differentiated parenchymal microglia. FASEB J. 18, 998–1000 (2004).
Nakano, K., Migita, M., Mochizuki, H. & Shimada, T. Differentiation of transplanted bone marrow cells in the adult mouse brain. Transplantation 71, 1735–1740 (2001).
Corti, S. et al. Neuroectodermal and microglial differentiation of bone marrow cells in the mouse spinal cord and sensory ganglia. J. Neurosci. Res. 70, 721–733 (2002).
Vallieres, L. & Sawchenko, P. E. Bone marrow-derived cells that populate the adult mouse brain preserve their hematopoietic identity. J. Neurosci. 23, 5197–5207 (2003).
Flugel, A., Bradl, M., Kreutzberg, G. W. & Graeber, M. B. Transformation of donor-derived bone marrow precursors into host microglia during autoimmune CNS inflammation and during the retrograde response to axotomy. J. Neurosci. Res. 66, 74–82 (2001).
Cogle, C. R. et al. Bone marrow transdifferentiation in brain after transplantation: a retrospective study. Lancet 363, 1432–1437 (2004).
Unger, E. R. et al. Male donor-derived cells in the brains of female sex-mismatched bone marrow transplant recipients: a Y-chromosome specific in situ hybridization study. J. Neuropathol. Exp. Neurol. 52, 460–470 (1993).
Kennedy, D. W. & Abkowitz, J. L. Kinetics of central nervous system microglial and macrophage engraftment: analysis using a transgenic bone marrow transplantation model. Blood 90, 986–993 (1997).
Lassmann, H. & Hickey, W. F. Radiation bone marrow chimeras as a tool to study microglia turnover in normal brain and inflammation. Clin. Neuropathol. 12, 284–285 (1993).
Krall, W. J., Challita, P. M., Perlmutter, L. S., Skelton, D. C. & Kohn, D. B. Cells expressing human glucocerebrosidase from a retroviral vector repopulate macrophages and central nervous system microglia after murine bone marrow transplantation. Blood 83, 2737–2748 (1994).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nature Neurosci. 10, 1544–1553 (2007). Together with reference 90, this study provides the first description of microglial self-renewal occurring independently of circulating myeloid cells.
Capotondo, A. et al. Brain conditioning is instrumental for successful microglia reconstitution following hematopoietic stem cell transplantation. Proc. Natl Acad. Sci. USA 109, 15018–15023 (2012).
Bottcher, C., Fernandez-Klett, F., Gladow, N., Rolfes, S. & Priller, J. Targeting myeloid cells to the brain using non-myeloablative conditioning. PLoS ONE 8, e80260 (2013).
Wilkinson, F. L. et al. Busulfan conditioning enhances engraftment of hematopoietic donor-derived cells in the brain compared with irradiation. Mol. Ther. 21, 868–876 (2013).
Kierdorf, K., Katzmarski, N., Haas, C. A. & Prinz, M. Bone marrow cell recruitment to the brain in the absence of irradiation or parabiosis bias. PLoS ONE 8, e58544 (2013).
Cartier, N. et al. Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science 326, 818–823 (2009).
Biffi, A. et al. Lentiviral hematopoietic stem cell gene therapy benefits metachromatic leukodystrophy. Science 341, 1233158 (2013).
Biffi, A., Aubourg, P. & Cartier, N. Gene therapy for leukodystrophies. Hum. Mol. Genet. 20, R42–R53 (2011).
Eichler, F. S. et al. Is microglial apoptosis an early pathogenic change in cerebral X-linked adrenoleukodystrophy? Ann. Neurol. 63, 729–742 (2008).
Boillee, S. et al. Onset and progression in inherited ALS determined by motor neurons and microglia. Science 312, 1389–1392 (2006).
Simard, A. R., Soulet, D., Gowing, G., Julien, J. P. & Rivest, S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer's disease. Neuron 49, 489–502 (2006).
El Khoury, J. et al. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nature Med. 13, 432–438 (2007).
Chen, S. K. et al. Hematopoietic origin of pathological grooming in Hoxb8 mutant mice. Cell 141, 775–785 (2010).
Castro, J., Mellios, N. & Sur, M. Mechanisms and therapeutic challenges in autism spectrum disorders: insights from Rett syndrome. Curr. Opin. Neurol. 26, 154–159 (2013).
Derecki, N. C. et al. Wild-type microglia arrest pathology in a mouse model of Rett syndrome. Nature 484, 105–109 (2012). Together with reference 158, this report suggests that dysfunction of microglia may induce behavioural disturbances.
Hoeffel, G. et al. Adult Langerhans cells derive predominantly from embryonic fetal liver monocytes with a minor contribution of yolk sac-derived macrophages. J. Exp. Med. 209, 1167–1181 (2012).
Chorro, L. et al. Langerhans cell (LC) proliferation mediates neonatal development, homeostasis, and inflammation-associated expansion of the epidermal LC network. J. Exp. Med. 206, 3089–3100 (2009).
Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38, 792–804 (2013).
Guilliams, M. et al. Alveolar macrophages develop from fetal monocytes that differentiate into long-lived cells in the first week of life via GM-CSF. J. Exp. Med. 210, 1977–1992 (2013).
Tamoutounour, S. et al. Origins and functional specialization of macrophages and of conventional and monocyte-derived dendritic cells in mouse skin. Immunity 39, 925–938 (2013).
Hsu, A. P. et al. GATA2 haploinsufficiency caused by mutations in a conserved intronic element leads to MonoMAC syndrome. Blood 121, 3830–3837 (2013).
Hambleton, S. et al. IRF8 mutations and human dendritic-cell immunodeficiency. N. Engl. J. Med. 365, 127–138 (2011).
Bigley, V. & Collin, M. Dendritic cell, monocyte, B and NK lymphoid deficiency defines the lost lineages of a new GATA-2 dependent myelodysplastic syndrome. Haematologica 96, 1081–1083 (2011).
Blevins, G. & Fedoroff, S. Microglia in colony-stimulating factor 1-deficient op/op mice. J. Neurosci. Res. 40, 535–544 (1995).
Acknowledgements
The authors apologize to all of those colleagues whose work was discussed without proper quotation owing space constraints. The authors thank S. M. Brendecke for critically reading the manuscript and M. Knust for help with the artwork. M.P. and J.P. are joint coordinators of the DFG-funded research unit (FOR) 1336. In addition, M.P. is supported by the BMBF-funded competence network of multiple sclerosis (KKNMS), the Gemeinnützige Hertie-Stiftung (GHST), the Fritz Thyssen Stiftung, the competence network of neurodegenerative disorders (KNDD) and the DFG (SFB 992). J.P. receives additional funding from the DFG (SFB/TRR43 and the Cluster of Excellence NeuroCure), the BMBF (Forschungsnetz zu psychischen Erkrankungen) and the Berlin Institute of Health.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Macrophages
-
Tissue-resident cells of the mononuclear phagocyte system that are characterized by their ability to phagocytose foreign particulate material, debris and colloidal material.
- Mononuclear phagocytes
-
A mononuclear cell type of the myeloid lineage (macrophages, monocytes or dendritic cells) that have the ability to phagocytose.
- Dendritic cells
-
Also known as an interdigitating reticular cells because of their branched morphology. Dendritic cells are the most potent stimulators of T cell responses.
- Monocyte
-
A type of mononuclear leukocyte that is derived from the bone marrow and circulates in the bloodstream. Monocytes typically migrate into tissues, where they can differentiate into various types of macrophages.
- Haematopoietic stem cells
-
(HSCs). Rare multipotent cells that give rise to all blood cells, including myeloid and lymphoid lineages.
- Leukocytes
-
White blood cells derived from multipotent haematopoietic stem cells in the bone marrow. Leukocytes are of myeloid or lymphoid lineage and are found in the blood and lymphatic system.
- Yolk sac
-
A membranous sac attached to the embryo that provides early nourishment in the form of yolk. It functions as the developmental circulatory system of the embryo before internal circulation begins.
- CD45
-
(Also known as leukocyte common antigen and PTPRC). A type I transmembrane protein present on all haematopoietic cells that assists in cell activation and the levels of which are reduced in mature parenchymal microglia.
- Neuroepithelium
-
The ectodermal epithelium in the embryo from which the CNS and its main cellular constituents (neurons, astrocytes, oligodendrocytes and ependymal cells) are derived.
- Natural killer cells
-
A type of cytotoxic lymphocyte that are crucial for the innate immune system.
- Myelopoiesis
-
The regulated formation of myeloid cells, including macrophages, monocytes, dendritic cells and granulocytes. Myelopoiesis takes place in the bone marrow or the yolk sac.
- Deep RNA sequencing
-
An approach enabled by next-generation sequencing technology that is particularly useful for identifying low-abundance RNAs or low-frequency mutations.
- Parabiotic mice
-
Mice in which shared blood circulation is created via surgical intervention. This procedure enables the fate of labelled donor cells to be followed in the parabiotic partner.
- Graft-versus-host disease
-
A complication following an allogeneic tissue transplant in which immune cells (white blood cells) in the tissue (the graft) recognize the recipient (the host) as 'foreign'.
- X-linked adrenoleukodystrophy
-
A rare X chromosome-linked disorder resulting from mutations in ABCD1 (ATP-binding cassette subfamily D member 1) that cause defects in peroxisomal β-oxidation and lead to the accumulation of very-long-chain fatty acids, particularly in the CNS and adrenal cortex.
Rights and permissions
About this article
Cite this article
Prinz, M., Priller, J. Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nat Rev Neurosci 15, 300–312 (2014). https://doi.org/10.1038/nrn3722
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn3722
This article is cited by
-
Taming microglia: the promise of engineered microglia in treating neurological diseases
Journal of Neuroinflammation (2024)
-
Lifelong absence of microglia alters hippocampal glutamatergic networks but not synapse and spine density
EMBO Reports (2024)
-
Minocycline protects against microgliopathy in a Csf1r haplo-insufficient mouse model of adult-onset leukoencephalopathy with axonal spheroids and pigmented glia (ALSP)
Journal of Neuroinflammation (2023)
-
Differential effects of two phosphodiesterase 4 inhibitors against lipopolysaccharide-induced neuroinflammation in mice
BMC Neuroscience (2023)
-
Low protein-induced intrauterine growth restriction as a risk factor for schizophrenia phenotype in a rat model: assessing the role of oxidative stress and neuroinflammation interaction
Translational Psychiatry (2023)