Abstract
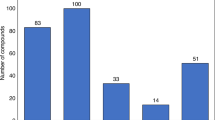
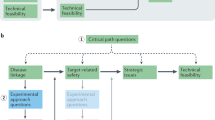
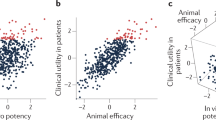
Maintaining research and development (R&D) productivity at a sustainable level is one of the main challenges currently facing the pharmaceutical industry. In this article, we discuss the results of a comprehensive longitudinal review of AstraZeneca's small-molecule drug projects from 2005 to 2010. The analysis allowed us to establish a framework based on the five most important technical determinants of project success and pipeline quality, which we describe as the five 'R's: the right target, the right patient, the right tissue, the right safety and the right commercial potential. A sixth factor — the right culture — is also crucial in encouraging effective decision-making based on these technical determinants. AstraZeneca is currently applying this framework to guide its R&D teams, and although it is too early to demonstrate whether this has improved the company's R&D productivity, we present our data and analysis here in the hope that it may assist the industry overall in addressing this key challenge.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Pammolli, F., Magazzini, L. & Riccaboni, M. The productivity crisis in pharmaceutical R&D. Nature Rev. Drug Discov. 10, 428–438 (2011).
Paul, S. M. et al. How to improve R&D productivity: the pharmaceutical industry's grand challenge. Nature Rev. Drug Discov. 9, 203–214 (2010).
Herper, M. The truly staggering cost of inventing new drugs. Forbes [online], (10 Feb 2012).
Munros, B. Lessons from 60 years of pharmaceutical innovation. Nature Rev. Drug Discov. 8, 959–968 (2009).
Scannell, J. W., Blanckley, A., Boldon, H. & Warrington, B. Diagnosing the decline in pharmaceutical R&D efficiency. Nature Rev. Drug Discov. 11, 191–200 (2012).
Handen, J. S. The industrialization of drug discovery. Drug Discov. Today 7, 83–85 (2002).
Linder, M. D. Clinical attrition due to biased preclinical assessments of potential efficacy. Pharmacol. Ther. 115, 148–175 (2007).
Ringel, M., Tollman, P., Hersch, G. & Schulze, U. Does size matter in R&D productivity? If not, what does? Nature Rev. Drug Discov. 12, 901–902 (2013).
Arrowsmith, J. & Miller, P. Trial Watch: Phase II and Phase III attrition rates 2011–2012. Nature Rev. Drug Discov. 12, 569 (2013).
Redfern, W. et al. Impact and frequency of different toxicities throughout the pharmaceutical life cycle. The Toxicologist 114, 231 (2010).
Kola, I. & Landis, J. Can the pharmaceutical industry reduce attrition rates? Nature Rev. Drug Discov. 3, 711–716 (2004).
Picker, S. M. In-vitro assessment of platelet function. Transfus. Apher. Sci. 44, 305–319 (2011).
Seok, J. et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl Acad. Sci. USA 110, 3507–3512 (2013).
de Jong, M. & Maina, T. Of mice and humans: are they the same? — Implications in cancer translational research. J. Nucl. Med. 51, 501–504 (2010).
Wendler, A. & Wehling, M. The translatability of animal models for clinical development: biomarkers and disease models. Curr. Opin. Pharmacol. 10, 601–606 (2010).
Jucker, M. The benefits and limitations of animal models for translational research in neurodegenerative diseases. Nature Med. 16, 1210–1214 (2010).
Pangalos, M. N., Schechter, L. E. & Hurko, O. Drug development for CNS disorders: strategies for balancing risk and reducing attrition. Nature Rev. Drug Discov. 6, 521–532 (2007).
Prahalad, S. Negative association between the chemokine receptor CCR5-Δ32 polymorphism and rheumatoid arthritis: a meta-analysis. Genes Immun. 7, 264–268 (2006).
Vierboom, M. P. et al. Inhibition of the development of collagen-induced arthritis in rhesus monkeys by a small molecular weight antagonist of CCR5. Arthritis Rheum. 52, 627–636 (2005).
Okamoto, H. & Kamatani, N. CCR-5 antagonist inhibits the development of adjuvant arthritis in rats. Rheumatology 45, 230–232 (2006).
Fleishaker, D. L. et al. Maraviroc, a chemokine receptor-5 antagonist, fails to demonstrate efficacy in the treatment of patients with rheumatoid arthritis in a randomized, double-blind placebo-controlled trial. Arthritis Res. Ther. 14, R11 (2012).
van Kuijk, A. W. et al. CCR5 blockade in rheumatoid arthritis: a randomised, double-blind, placebo-controlled clinical trial. Ann. Rheum. Dis. 69, 2013–2016 (2010).
Gerlag, D. M. et al. Preclinical and clinical investigation of a CCR5 antagonist, AZD5672, in patients with rheumatoid arthritis receiving methotrexate. Arthritis Rheum. 62, 3154–3160 (2010).
Wehling, M. Assessing the translatability of drug projects: what needs to be scored to predict success? Nature Rev. Drug Discov. 8, 541–546 (2009).
Morgan, P. et al. Can the flow of medicines be improved? Fundamental pharmacokinetic and pharmacological principles toward improving Phase II survival. Drug Discov. Today 17, 419–424 (2012).
Berman, R. M. et al. Antidepressant effects of ketamine in depressed patients. Biol. Psychiatry 47, 351–354 (2000).
aan het Rot, M. et al. Safety and efficacy of repeated-dose intravenous ketamine for treatment-resistant depression. Biol. Psychiatry 67, 139–145 (2010).
Quirk, M. et al. Abstract P-09-045. Effects of low-trapping NMDA channel blocker AZD6765 on gamma-band EEG and psychotomimetic liability: a comparison to ketamine in freely behaving rats. Int. J. Neuropsychopharmacol. 15 (Suppl. S1), 153 (2012).
Skolnick, P., Popik, P. & Trullas, R. Glutamate-based antidepressants: 20 years on. Trends Pharmacol. Sci. 30, 563–569 (2009).
Mealing, G., Lanthorn, T. H., Murray, C. L., Small, D. L. & Morley, P. Differences in degree of trapping of low-affinity uncompetitve N-methyl-d-aspartic acid receptor antagonists with similar kinetics of block. J. Pharmacol. Exp. Ther. 288, 204–210 (1999).
Zarate, C. A. et al. Replication of ketamine's antidepressant efficacy in bipolar depression: a randomized controlled add-on trial. Biol. Psychiatry 71, 939–946 (2012).
Lip, G. Y. et al. Oral direct thrombin inhibitor AZD0837 for the prevention of stroke and systemic embolism in patients with non-valvular atrial fibrillation: a randomised doseguiding, safety and tolerability study of four doses of AZD0837 versus vitamin K antagonists. Eur. Heart J. 30, 2897–2907 (2009).
Olsson, S. B. et al. Safety and tolerability of an immediate-release formulation of the oral direct thrombin inhibitor AZD0837 in the prevention of stroke and systemic embolism in patients with atrial fibrillation. Thromb. Haemost. 103, 604–612 (2010).
Pehrsson, S., Johansson K., Kjaer, M. & Elg, M. Evaluation of AR-H067637, the active metabolite of the new direct thrombin inhibitor AZD0837, in models of venous and arterial thrombosis and bleeding in anaesthetised rats. Thromb. Haemost. 104, 1242–1249 (2010).
Wolzt, M. et al. Effect on perfusion chamber thrombus size in patients with atrial fibrillation during anticoagulant treatment with oral direct thrombin inhibitors, AZD0837 or ximelagatran, or with vitamin K antagonists. Thromb. Res. 129, e83–e91 (2012).
Olsson, S. et al. Stroke preventions with the oral direct thrombin inhibitor ximelagatran compared with warfarin in patients with non-valvular atrial fibrillation (SPORTIF III): randomised clinical trial. Lancet 362, 1691–1698 (2003).
SPORTIF Executive Steering Committee for the SPORTIF V Investigators. Ximelagatran versus warfarin for stroke prevention in patients with nonvalvular atrial fibrillation: a randomized trial. JAMA 293, 690–698 (2005).
Uppoor, R. S. et al. The use of imaging in the early development of neuropharmacological drugs: a survey of approved NDAs. Clin. Pharmacol. Ther. 84, 69–74 (2008).
Freedman, N. M. et al. In vivo measurement of brain monoamine oxidase B occupancy by rasagiline, using 11C-L-Deprenyl and PET. J. Nucl. Med. 46, 1618–1624 (2005).
Thebault, J. J., Guillaume, M. & Levy, R. Tolerability, safety and pharmacodynamics and pharmacokinetics of rasagiline: a potent, selective and irreversible monoamine oxidase type B inhibitor. Pharmacotherapy 24, 1295–1305 (2004).
Ramsey, S. J., Attkins, N. J., Fish, R. & van der Graaf, P. H. Quantitative pharmacological analysis of antagonist binding kinetics at CRF1 receptors in vitro and in vivo. Br. J. Pharmacol. 164, 992–1007 (2011).
Qureshi, Z. P., Seoane-Vazquez, E., Rodriguez-Monguio, R., Stevenson, K. B. & Szeinbach, S. L. Market withdrawal of new molecular entities approved in the United States from 1980 to 2009. Pharmacoepidemiol. Drug Safety 20, 772–777 (2011).
King, A. Prevention: neuropsychiatric adverse effects signal the end of the line for rimonabant. Nature Rev. Cardiol. 7, 602 (2010).
Tralau, T. & Luch, A. Drug-mediated toxicity: illuminating the 'bad' in the test tube by means of cellular assays? Trends Pharmacol. Sci. 33, 353–364 (2012).
Farmer, H. et al. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature. 434, 917–921 (2005).
Fong, P. C. et al. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N. Engl. J. Med. 361, 123–134 (2009).
Ledermann, J. et al. Olaparib maintenance therapy in patients with platinum-sensitive relapsed serous ovarian cancer (SOC) and a BRCA mutation (BRCAm). J. Clin. Oncol. Abstr. 31, 5505 (2013).
Jack, C. R. et al. Evidence for ordering of Alzheimer disease biomarkers. JAMA Neurol. 68, 1526–1535 (2011).
Poirier, J. et al. Apolipoprotein E polymorphism and Alzheimer's disease. Lancet. 2, 697–699 (1993).
Holgate, S. Pathophysiology of asthma: what has our current understanding taught us about new therapeutic approaches? J. Allergy Clin. Immunol. 128, 495–505 (2011).
Izuhara, K. et al. [Clarification of the pathogenesis and development of clinical examination for allergic disease]. Rinsho Byori 55, 369–374 (in Japanese) (2007).
Izuhara, K. & Saito, H. Microarray-based identification of novel biomarkers in asthma. Allergol. Int. 55, 361–367 (2006).
Izuhara, K. et al. IL-13: a promising therapeutic target for bronchial asthma. Curr. Med. Chem. 13, 2291–2298 (2006).
Woodruff, P. G. et al. Genome-wide profiling identifies epithelial cell genes associated with asthma and with treatment response to corticosteroids. Proc. Natl Acad. Sci. USA. 104, 15858–15863 (2007).
Corren, J. et al. Lebrikizumab treatment in adults with asthma. N. Engl. J. Med. 365, 1088–1098 (2011).
Thomson, N. C., Patel, M. & Smith, A. D. Lebrikizumab in the personalized management of asthma. Biologics 6, 329–335 (2012).
Wenzel, S. E. et al. Evidence that severe asthma can be divided pathologically into two inflammatory subtypes with distinct physiologic and clinical characteristics. Am. J. Respir. Crit. Care Med. 160, 1001–1008 (1999).
Pavord, I. D. et al. Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet 380, 651–659 (2012).
Leckie, M. J. et al. Effects of an interleukin-5 blocking monoclonal antibody on eosinophils, airway hyper-responsiveness, and the late asthmatic response. Lancet 356, 2144–2148 (2000).
Visser, S. et al. Model-based drug discovery: implementation and impact. Drug Discov. Today 18, 746–775 (2013).
Lisman, J. E., Raghavachari, S. & Tsien, R. W. The sequence of events that underlie quantal transmission at central glutamatergic synapses. Nature Rev. Neurosci. 8, 597–609 (2007).
Moghaddam, B. & Javitt, D. From revolution to evolution: the glutamate hypothesis of schizophrenia and its implication for treatment. Neuropsychopharmacology 37, 4–15 (2012).
Patil, S. T. et al. Activation of mGlu2/3 receptors as a new approach to treat schizophrenia: a randomized Phase 2 clinical trial. Nature Med. 13, 1102–1107 (2007).
Geyer, M. A. & Moghaddam, B. in Neuropsychopharmacology: The Fifth Generation of Progress (eds Davies, K., Charney, D., Coyle, J. T. & Nemeroff, C.) 689–701 (Lippincott Williams & Wilkins, 2002).
Matsuoka, T. et al. Prostaglandin D2 as a mediator of allergic astma. Science 287, 2013–2017 (2000).
Arimura, A. et al. Prevention of allergic inflammation by a novel prostaglandin receptor antagonist, S-5751. J. Pharmacol. Exp. Ther. 298, 411–419 (2001).
Shichijo, M. et al. A prostaglandin D2 receptor antagonist modifies experiemental asthma in sheep. Clin. Exp. Allergy 39, 1404–1414 (2009).
Lukacs, N. W. et al. CRTH2 antagonism significantly ameliorates airway hyperreactivity and downregulates inflammation-induced genes in a mouse model of airway inflammation. Am. J. Physiol. Lung Cell. Mol. Physiol. 295, L767–L779 (2008).
Gervais, F. G. et al. Pharmacological characterization of MK-7246, a potent and selective CRTH2 (chemoattractant receptor-homologous molecule expressed on T-helper type 2 cells) antagonist. Mol. Pharmacol. 79, 69–76 (2011).
Barnes, N. et al. A randomized, double-blind, placebo-controlled strudy of the CRTH2 antagonist OC0000459 in moderate persistent asthma. Clin. Exp. Allergy. 42, 38–48 (2012).
Busse, W. W. et al. Safety and efficacy of the prostaglandin D2 receptor antagonist AMG 853 in asthmatic patients. J. Allergy Clin. Immunol. 131, 339–345 (2013).
Acknowledgements
The authors would like to thank S. Curran, J. Curwen, U. Eriksson, G. Johnston, R. Maciewicz, J. Munroe, M. Needham, P. Newham, W. Redfern, M. Snowden, F. Tierney, J.-P. Valentin and J. Waterton for their help in producing and reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All authors are employees and shareholders of AstraZeneca.
Related links
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Cook, D., Brown, D., Alexander, R. et al. Lessons learned from the fate of AstraZeneca's drug pipeline: a five-dimensional framework. Nat Rev Drug Discov 13, 419–431 (2014). https://doi.org/10.1038/nrd4309
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd4309
This article is cited by
-
Development of a human genetics-guided priority score for 19,365 genes and 399 drug indications
Nature Genetics (2024)
-
Pipeline progress and portfolio management of the top 30 pharma companies over the past two decades
Journal of Pharmaceutical Policy and Practice (2023)
-
ProfhEX: AI-based platform for small molecules liability profiling
Journal of Cheminformatics (2023)
-
Experimental strategies to improve drug-target identification in mass spectrometry-based thermal stability assays
Communications Chemistry (2023)
-
A cell-free strategy for host-specific profiling of intracellular antibiotic sensitivity and resistance
npj Antimicrobials and Resistance (2023)