Key Points
-
Over the past 18 months, recurrent activating mutations within the oestrogen receptor α (ER) LBD have been detected in 15–20% of patients with metastatic ER-positive endocrine-resistant breast cancer
-
The ER LBD mutations confer constitutive ligand-independent activity and are relatively resistant to tamoxifen and fulvestrant treatment
-
These mutations in the gene encoding the ER, ESR1, are detected mainly in metastatic tumours, implicating the clonal selection of these mutations as the mechanism of resistance to therapy
-
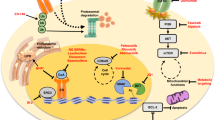
Structural studies have shown that the ER LBD mutations lead to ligand-independent stabilization of the LBD in the agonistic conformation
-
To study the clonal evolution and discern the full genetic complexity and clinical significance of the ESR1 mutations, new sensitive sequencing technologies will need to be applied
-
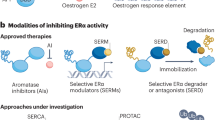
Potential strategies to overcome ER-related endocrine resistance include high-dose fulvestrant and tamoxifen, inhibitors of ER co-activator proteins, novel SERDs and SERMs, and other agents targeting the ER signalling axis
Abstract
Approximately 70% of breast cancers are oestrogen receptor α (ER) positive, and are, therefore, treated with endocrine therapies. However, about 25% of patients with primary disease and almost all patients with metastases will present with or eventually develop endocrine resistance. Despite the magnitude of this clinical challenge, the mechanisms underlying the development of resistance remain largely unknown. In the past 2 years, several studies unveiled gain-of-function mutations in ESR1, the gene encoding the ER, in approximately 20% of patients with metastatic ER-positive disease who received endocrine therapies, such as tamoxifen and aromatase inhibitors. These mutations are clustered in a 'hotspot' within the ligand-binding domain (LBD) of the ER and lead to ligand-independent ER activity that promotes tumour growth, partial resistance to endocrine therapy, and potentially enhanced metastatic capacity; thus, ER LBD mutations might account for a mechanism of acquired endocrine resistance in a substantial fraction of patients with metastatic disease. In general, the absence of detectable ESR1 mutations in patients with treatment-naive disease, and the correlation between the frequency of patients with tumours harbouring these mutations and the number of endocrine treatments received suggest that, under selective treatment pressure, clonal expansion of rare mutant clones occurs, leading to resistance. Preclinical and clinical development of rationale-based novel therapeutic strategies that inhibit these ER mutants has the potential to substantially improve treatment outcomes. We discuss the contribution of ESR1 mutations to the development of acquired resistance to endocrine therapy, and evaluate how mutated ER can be detected and targeted to overcome resistance and improve patient outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Parker, J. S. et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 27, 1160–1167 (2009).
Sorlie, T. et al. Gene expression patterns of breast carcinomas distinguish tumour subclasses with clinical implications. Proc. Natl Acad. Sci. USA 98, 10869–10874 (2001).
Perou, C. M. et al. Molecular portraits of human breast tumours. Nature 406, 747–752 (2000).
Creighton, C. J. The molecular profile of luminal B breast cancer. Biologics 6, 289–297 (2012).
Higa, G. M. & Fell, R. G. Sex hormone receptor repertoire in breast cancer. Int. J. Breast Cancer 2013, 284036 (2013).
Burns, K. A. & Korach, K. S. Oestrogen receptors and human disease: an update. Arch. Toxicol. 86, 1491–1504 (2012).
Schiff, R., Osborne, C. K. & Fuqua, S. A. in Diseases of the Breast 4th edn (eds Harris, J. R., Lippman, M. E., Morrow, M. & Osborne, C. K.) 408–430 (Wolters Kluwer/Lippincott Williams & Wilkins, 2009).
Huang, B., Warner, M. & Gustafsson, J. A. Oestrogen receptors in breast carcinogenesis and endocrine therapy. Mol. Cell. Endocrinol. http://dx.doi.org/10.1016/j.mce.2014.11.015 (2014).
Olefsky, J. M. Nuclear receptor minireview series. J. Biol. Chem. 276, 36863–36864 (2001).
Klinge, C. M. Oestrogen receptor interaction with oestrogen response elements. Nucleic Acids Res. 29, 2905–2919 (2001).
Kushner, P. J. et al. Oestrogen receptor pathways to AP-1. J. Steroid Biochem. Mol. Biol. 74, 311–317 (2000).
Schiff, R. et al. Advanced concepts in oestrogen receptor biology and breast cancer endocrine resistance: implicated role of growth factor signalling and oestrogen receptor coregulators. Cancer Chemother. Pharmacol. 56 (Suppl. 1), 10–20 (2005).
Lupien, M. et al. Growth factor stimulation induces a distinct ERα cistrome underlying breast cancer endocrine resistance. Genes Dev. 24, 2219–2227 (2010).
Levin, E. R. Elusive extranuclear oestrogen receptors in breast cancer. Clin. Cancer Res. 18, 6–8 (2012).
Johnston, S. R. & Yeo, B. The optimal duration of adjuvant endocrine therapy for early stage breast cancer—with what drugs and for how long? Curr. Oncol. Rep. 16, 358 (2014).
Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365, 1687–1717 (2005).
Gluck, S. Extending the clinical benefit of endocrine therapy for women with hormone receptor-positive metastatic breast cancer: differentiating mechanisms of action. Clin. Breast Cancer 14, 75–84 (2014).
Obiorah, I. & Jordan, V. C. Progress in endocrine approaches to the treatment and prevention of breast cancer. Maturitas 70, 315–321 (2011).
Williams, N. & Harris, L. N. The renaissance of endocrine therapy in breast cancer. Curr. Opin. Obstet. Gynecol. 26, 41–47 (2014).
Peng, J., Sengupta, S. & Jordan, V. C. Potential of selective oestrogen receptor modulators as treatments and preventives of breast cancer. Anticancer Agents Med. Chem. 9, 481–499 (2009).
Howell, A. Pure oestrogen antagonists for the treatment of advanced breast cancer. Endocr. Relat. Cancer 13, 689–706 (2006).
Cigler, T. & Goss, P. E. Breast cancer adjuvant endocrine therapy. Cancer J. 13, 148–155 (2007).
Francis, P. A. et al. Adjuvant ovarian suppression in premenopausal breast cancer. N. Engl. J. Med. 372, 436–446 (2015).
Musgrove, E. A. & Sutherland, R. L. Biological determinants of endocrine resistance in breast cancer. Nat. Rev. Cancer 9, 631–643 (2009).
Osborne, C. K. & Schiff, R. Mechanisms of endocrine resistance in breast cancer. Annu. Rev. Med. 62, 233–247 (2011).
Hoefnagel, L. D. et al. Prognostic value of oestrogen receptor alpha and progesterone receptor conversion in distant breast cancer metastases. Cancer 118, 4929–4935 (2012).
Drury, S. C. et al. Changes in breast cancer biomarkers in the IGF1R/PI3K pathway in recurrent breast cancer after tamoxifen treatment. Endocr. Relat. Cancer 18, 565–577 (2011).
Dodwell, D., Wardley, A. & Johnston, S. Postmenopausal advanced breast cancer: options for therapy after tamoxifen and aromatase inhibitors. Breast 15, 584–594 (2006).
Robertson, J. F. et al. Fulvestrant versus anastrozole for the treatment of advanced breast carcinoma in postmenopausal women: a prospective combined analysis of two multicentre trials. Cancer 98, 229–238 (2003).
Fowler, A. M. et al. Increases in oestrogen receptor-alpha concentration in breast cancer cells promote serine 118/104/106-independent AF-1 transactivation and growth in the absence of oestrogen. FASEB J. 18, 81–93 (2004).
Schiff, R., Massarweh, S., Shou, J. & Osborne, C. K. Breast cancer endocrine resistance: how growth factor signalling and oestrogen receptor coregulators modulate response. Clin. Cancer Res. 9, 447S–454S (2003).
Johnston, S. R. New strategies in oestrogen receptor-positive breast cancer. Clin. Cancer Res. 16, 1979–1987 (2010).
Spears, M. & Bartlett, J. The potential role of oestrogen receptors and the SRC family as targets for the treatment of breast cancer. Expert Opin. Ther. Targets 13, 665–674 (2009).
Cancer Genome Atlas, Comprehensive molecular portraits of human breast tumours. Nature 490, 61–70 (2012).
Memorial Sloan Kettering Cancer Centre. The cBioPortal for Cancer Genomics [online], (2015).
Karnik, P. S., Kulkarni, S., Liu, X. P., Budd, G. T. & Bukowski, R. M. Oestrogen receptor mutations in tamoxifen-resistant breast cancer. Cancer Res. 54, 349–353 (1994).
Roodi, N. et al. Oestrogen receptor gene analysis in oestrogen receptor-positive and receptor-negative primary breast cancer. J. Natl Cancer Inst. 87, 446–451 (1995).
Li, S. et al. Endocrine-therapy-resistant ESR1 variants revealed by genomic characterization of breast-cancer-derived xenografts. Cell Rep. 4, 1116–1130 (2013).
Toy, W. et al. ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat. Genet. 45, 1439–1445 (2013).
Robinson, D. R. et al. Activating ESR1 mutations in hormone-resistant metastatic breast cancer. Nat. Genet. 45, 1446–1451 (2013).
Merenbakh-Lamin, K. et al. D538G mutation in oestrogen receptor-alpha: a novel mechanism for acquired endocrine resistance in breast cancer. Cancer Res. 73, 856–864 (2013).
Jeselsohn, R. et al. Emergence of constitutively active oestrogen receptor-alpha mutations in pretreated advanced oestrogen receptor-positive breast cancer. Clin. Cancer Res. 20, 1757–1767 (2014).
Yu, H. A., Riely, G. J. & Lovly, C. M. Therapeutic strategies utilized in the setting of acquired resistance to EGFR tyrosine kinase inhibitors. Clin. Cancer Res 20, 5898–5907 (2014).
Rosenzweig, S. A. Acquired resistance to drugs targeting receptor tyrosine kinases. Biochem. Pharmacol. 83, 1041–1048 (2012).
Engelman, J. A. et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signalling. Science 316, 1039–1043 (2007).
Brady, S. W., Zhang, J., Seok, D., Wang, H. & Yu, D. Enhanced PI3K p110alpha signalling confers acquired lapatinib resistance that can be effectively reversed by a p110alpha-selective PI3K inhibitor. Mol. Cancer Ther. 13, 60–70 (2014).
Bertino, J. R. Cancer research: from folate antagonism to molecular targets. Best Pract. Res. Clin. Haematol. 22, 577–582 (2009).
Huw, L. Y. et al. Acquired PIK3CA amplification causes resistance to selective phosphoinositide 3-kinase inhibitors in breast cancer. Oncogenesis 2, e83 (2013).
Takezawa, K. et al. HER2 amplification: a potential mechanism of acquired resistance to EGFR inhibition in EGFR-mutant lung cancers that lack the second-site EGFRT790M mutation. Cancer Discov. 2, 922–933 (2012).
Visakorpi, T. et al. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat. Genet. 9, 401–406 (1995).
Holst, F. et al. Oestrogen receptor alpha (ESR1) gene amplification is frequent in breast cancer. Nat. Genet. 39, 655–660 (2007).
Tomita, S. et al. Oestrogen receptor alpha gene ESR1 amplification may predict endocrine therapy responsiveness in breast cancer patients. Cancer Sci. 100, 1012–1017 (2009).
Reis-Filho, J. S. et al. ESR1 gene amplification in breast cancer: a common phenomenon? Nat. Genet. 40, 809–810 (2008).
Adelaide, J. et al. Absence of ESR1 amplification in a series of breast cancers. Int. J. Cancer 123, 2970–2972 (2008).
Moelans, C. B. et al. ESR1 amplification is rare in breast cancer and is associated with high grade and high proliferation: a multiplex ligation-dependent probe amplification study. Anal. Cell. Pathol. (Amst.) 33, 13–18 (2010).
Albertson, D. G. ESR1 amplification in breast cancer: controversy resolved? J. Pathol. 227, 1–3 (2012).
Ooi, A. et al. Gene amplification of ESR1 in breast cancers—fact or fiction? A fluorescence in situ hybridization and multiplex ligation-dependent probe amplification study. J. Pathol. 227, 8–16 (2012).
Iggo, R. D. et al. Next generation sequencing shows clonal selection after treatment with anastrozole or fulvestrant in a randomized trial of postmenopausal patients with large operable or locally-advanced hormone-receptor-positive breast cancer [abstract]. 36th CTRC-AACR San Antonio Breast Cancer Symposium. PD3–03 (2013).
Piscuoglio, S. et al. Integrative genomic and transcriptomic characterization of papillary carcinomas of the breast. Mol. Oncol. 8, 1588–1602 (2014).
Veeraraghavan, J. et al. Recurrent ESR1–CCDC170 rearrangements in an aggressive subset of oestrogen receptor-positive breast cancers. Nat. Commun. 5, 4577 (2014).
Kandoth, C. et al. Mutational landscape and significance across 12 major cancer types. Nature 502, 333–339 (2013).
Fuqua, S. A. et al. A hypersensitive oestrogen receptor-alpha mutation in premalignant breast lesions. Cancer Res. 60, 4026–4029 (2000).
Fuqua, S. A., Gu, G. & Rechoum, Y. Oestrogen receptor (ER) alpha mutations in breast cancer: hidden in plain sight. Breast Cancer Res. Treat. 144, 11–19 (2014).
Giordano, C. et al. Growth factor-induced resistance to tamoxifen is associated with a mutation of oestrogen receptor alpha and its phosphorylation at serine 305. Breast Cancer Res. Treat. 119, 71–85 (2010).
Barone, I. et al. Expression of the K303R oestrogen receptor-alpha breast cancer mutation induces resistance to an aromatase inhibitor via addiction to the PI3K/Akt kinase pathway. Cancer Res. 69, 724–732 (2009).
Barone, I. et al. Phosphorylation of the mutant K303R oestrogen receptor alpha at serine 305 affects aromatase inhibitor sensitivity. Oncogene 29, 2404–2414 (2010).
Herynk, M. H. et al. Association between the oestrogen receptor alpha A908G mutation and outcomes in invasive breast cancer. Clin. Cancer Res. 13, 235–243 (2007).
Abbasi, S., Rasouli, M., Nouri, M. & Kalbasi, S. Association of oestrogen receptor-alpha A908G (K303R) mutation with breast cancer risk. Int. J. Clin. Exp. Med. 6, 39–49 (2013).
Conway, K. et al. The oestrogen receptor-alpha A908G (K303R) mutation occurs at a low frequency in invasive breast tumours: results from a population-based study. Breast Cancer Res. 7, R871–R880 (2005).
Ghimenti, C., Mello-Grand, M., Regolo, L., Zambelli, A. & Chiorino, G. Absence of the K303R oestrogen receptor alpha mutation in breast cancer patients exhibiting different responses to aromatase inhibitor anastrozole neoadjuvant treatment. Exp. Ther. Med. 1, 939–942 (2010).
Davies, M. P., O'Neill, P. A., Innes, H. & Sibson, D. R. Hypersensitive K303R oestrogen receptor-alpha variant not found in invasive carcinomas. Breast Cancer Res. 7, R113–R118 (2005).
Tokunaga, E., Kimura, Y. & Maehara, Y. No hypersensitive oestrogen receptor-alpha mutation (K303R) in Japanese breast carcinomas. Breast Cancer Res. Treat. 84, 289–292 (2004).
Baselga, J. et al. Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N. Engl. J. Med. 366, 520–529 (2012).
Zhang, Q. X., Borg, A., Wolf, D. M., Oesterreich, S. & Fuqua, S. A. An oestrogen receptor mutant with strong hormone-independent activity from a metastatic breast cancer. Cancer Res. 57, 1244–1249 (1997).
Carlson, K. E., Choi, I., Gee, A., Katzenellenbogen, B. S. & Katzenellenbogen, J. A. Altered ligand binding properties and enhanced stability of a constitutively active oestrogen receptor: evidence that an open pocket conformation is required for ligand interaction. Biochemistry 36, 897–905 (1997).
Lazennec, G., Ediger, T. R., Petz, L. N., Nardulli, A. M. & Katzenellenbogen, B. S. Mechanistic aspects of oestrogen receptor activation probed with constitutively active oestrogen receptors: correlations with DNA and coregulator interactions and receptor conformational changes. Mol. Endocrinol. 11, 1375–1386 (1997).
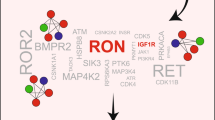
Dwyer, M. A. et al. WNT11 expression is induced by oestrogen-related receptor alpha and beta-catenin and acts in an autocrine manner to increase cancer cell migration. Cancer Res. 70, 298–308 (2010).
Masson, R. et al. In vivo evidence that the stromelysin-3 metalloproteinase contributes in a paracrine manner to epithelial cell malignancy. J. Cell. Biol. 140, 1535–1541 (1998).
Esseghir, S. et al. A role for glial cell derived neurotrophic factor induced expression by inflammatory cytokines and RET/GFR alpha 1 receptor upregulation in breast cancer. Cancer Res. 67, 11732–11741 (2007).
Gattelli, A. et al. Ret inhibition decreases growth and metastatic potential of oestrogen receptor positive breast cancer cells. EMBO Mol. Med. 5, 1335–1350 (2013).
Nettles, K. W. et al. NFkappaB selectivity of oestrogen receptor ligands revealed by comparative crystallographic analyses. Nat. Chem. Biol. 4, 241–247 (2008).
Brzozowski, A. M. et al. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature 389, 753–758 (1997).
Jeselsohn, R. M. et al. Emergence of constitutively active estrogen receptor mutations in advanced estrogen receptor positive breast cancer [abstract]. 36th CTRC-AACR San Antonio Breast Cancer Symposium. S3–06 (2013).
Wang, Y. et al. Clonal evolution in breast cancer revealed by single nucleus genome sequencing. Nature 512, 155–160 (2014).
Ng, C. K., Pemberton, H. N. & Reis-Filho, J. S. Breast cancer intratumour genetic heterogeneity: causes and implications. Expert Rev. Anticancer Ther. 12, 1021–1032 (2012).
Greaves, M. & Maley, C. C. Clonal evolution in cancer. Nature 481, 306–313 (2012).
Turner, N. C. & Reis-Filho, J. S. Genetic heterogeneity and cancer drug resistance. Lancet Oncol. 13, e178–e185 (2012).
Shendure, J. & Ji, H. Next-generation DNA sequencing. Nat. Biotechnol. 26, 1135–1145 (2008).
Gundry, M. & Vijg, J. Direct mutation analysis by high-throughput sequencing: from germline to low-abundant, somatic variants. Mutat. Res. 729, 1–15 (2012).
Meyerson, M., Gabriel, S. & Getz, G. Advances in understanding cancer genomes through second-generation sequencing. Nat. Rev. Genet. 11, 685–696 (2010).
Kinde, I., Wu, J., Papadopoulos, N., Kinzler, K. W. & Vogelstein, B. Detection and quantification of rare mutations with massively parallel sequencing. Proc. Natl Acad. Sci. USA 108, 9530–9535 (2011).
Schmitt, M. W. et al. Detection of ultra-rare mutations by next-generation sequencing. Proc. Natl Acad. Sci. USA 109, 14508–14513 (2012).
Vogelstein, B. & Kinzler, K. W. Digital PCR. Proc. Natl Acad. Sci. USA 96, 9236–9241 (1999).
Huggett, J. F., Cowen, S. & Foy, C. A. Considerations for digital PCR as an accurate molecular diagnostic tool. Clin. Chem. 61, 79–88 (2014).
Diaz, L. A. Jr & Bardelli, A. Liquid biopsies: genotyping circulating tumour DNA. J. Clin. Oncol. 32, 579–586 (2014).
Newman, A. M. et al. An ultrasensitive method for quantitating circulating tumour DNA with broad patient coverage. Nat. Med. 20, 548–554 (2014).
Di Leo, A. et al. Results of the CONFIRM phase III trial comparing fulvestrant 250 mg with fulvestrant 500 mg in postmenopausal women with oestrogen receptor-positive advanced breast cancer. J. Clin. Oncol. 28, 4594–4600 (2010).
Skapek, S. X. et al. Safety and efficacy of high-dose tamoxifen and sulindac for desmoid tumour in children: results of a Children's Oncology Group (COG) phase II study. Paediatr. Blood Cancer 60, 1108–1112 (2013).
McClay, E. F., McClay, M. T., Monroe, L., Jones, J. A. & Winski, P. J. A phase II study of high dose tamoxifen and weekly cisplatin in patients with metastatic melanoma. Melanoma Res. 11, 309–313 (2001).
Pinkerton, J. V. & Thomas, S. Use of SERMs for treatment in postmenopausal women. J. Steroid Biochem. Mol. Biol. 142, 142–154 (2014).
Mayer, I. A. et al. Phase I study of ARN-810, a novel selective estrogen receptor degrader, in postmenopausal women with locally advanced or metastatic estrogen receptor positive breast cancer [abstract]. CTRC-AACR San Antonio Breast Cancer Symposium. OT3-2-07 (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Wang, Y. et al. Bufalin is a potent small-molecule inhibitor of the steroid receptor co-activators SRC-3 and SRC-1. Cancer Res. 74, 1506–1517 (2014).
Yan, F. et al. Identification of verrucarin a as a potent and selective steroid receptor co-activator-3 small molecule inhibitor. PLoS ONE 9, e95243 (2014).
Finn, R. S. et al. PD 0332991, a selective cyclin D kinase 4/6 inhibitor, preferentially inhibits proliferation of luminal oestrogen receptor-positive human breast cancer cell lines in vitro. Breast Cancer Res. 11, R77 (2009).
Finn, R. S. et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 16, 25–35 (2015).
Abrams, S. I., Hand, P. H., Tsang, K. Y. & Schlom, J. Mutant ras epitopes as targets for cancer vaccines. Semin. Oncol. 23, 118–134 (1996).
Rahma, O. E. et al. The immunological and clinical effects of mutated ras peptide vaccine in combination with IL-2, GM-CSF, or both in patients with solid tumours. J. Transl. Med. 12, 55 (2014).
Buhrman, J. D. & Slansky, J. E. Improving T cell responses to modified peptides in tumour vaccines. Immunol. Res. 55, 34–47 (2013).
Yu, M. et al. Cancer therapy. Ex vivo culture of circulating breast tumour cells for individualized testing of drug susceptibility. Science 345, 216–220 (2014).
Acknowledgements
R.S. has been funded in part by NCI grants P50 CA58183 and P50 CA186784-01 (SPORE) and P30 CA125123, as well as by the Susan G. Komen for the Cure Promise Grant PG12221410, and the Breast Cancer Research Foundation. C.D.A. has been funded by the Cancer Prevention and Research Institute of Texas (CPRIT) programme RP140102 and the Baylor College of Medicine Comprehensive Cancer Training Program. R.J. has been funded in part by the Claudia Adams Barr Award and by the NCI grant P50 CA168508 (SPORE) Career Development Award.
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to the discussion, content, writing, and review/editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.B. has received a commercial research grant and is a consultant/advisory board member for Novartis Pharmaceuticals. R.S. has received research funding from AstraZeneca and GlaxoSmithKline. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Jeselsohn, R., Buchwalter, G., De Angelis, C. et al. ESR1 mutations—a mechanism for acquired endocrine resistance in breast cancer. Nat Rev Clin Oncol 12, 573–583 (2015). https://doi.org/10.1038/nrclinonc.2015.117
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2015.117
This article is cited by
-
Cyclin-dependent kinase 7 (CDK7) inhibitors as a novel therapeutic strategy for different molecular types of breast cancer
British Journal of Cancer (2024)
-
Efficacy of CDK4/6 inhibitors combined with endocrine therapy in HR+/HER2− breast cancer: an umbrella review
Journal of Cancer Research and Clinical Oncology (2024)
-
Modeling of Mouse Experiments Suggests that Optimal Anti-Hormonal Treatment for Breast Cancer is Diet-Dependent
Bulletin of Mathematical Biology (2024)
-
Upregulated PARP1 confers breast cancer resistance to CDK4/6 inhibitors via YB-1 phosphorylation
Experimental Hematology & Oncology (2023)
-
circRNA-SFMBT2 orchestrates ERα activation to drive tamoxifen resistance in breast cancer cells
Cell Death & Disease (2023)