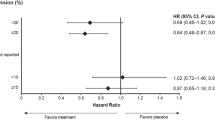
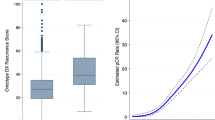
Abstract
Recommendation of systemic adjuvant therapy and choice of optimal agents for early-stage breast cancer remains a challenge. Adjuvant therapy is indicated on the assumption of residual micrometastatic disease. Adjuvant assessment tools for prognosis and prediction of treatment benefit, including Adjuvant! Online, the St Gallen Consensus, Oncotype DX® and MammaPrint®, aid clinical decision making. However, all of these tools have limitations that must be considered in their judicious application. Clinicopathological based tools are critically dependent on accurate, standardized measurement of parameters. Multigene tools are appealing for their objectivity and reproducibility, particularly regarding analysis of proliferation, but these approaches still overlook the biological heterogeneity within tumors evidenced by distinct cell subpopulations with different genomic patterns and function. The greatest treatment challenge remains for patients assessed as intermediate risk of relapse, a problem not overcome by multigene tools. Remarkable diversity in breast cancer dictates that adjuvant management must be biologically driven. Future identification of predictive biomarkers for specific chemotherapy sensitivity may allow targeted use of available agents, including anthracyclines, taxanes and DNA damaging agents. The presence of drug targets and targetable signaling pathways, rather than molecularly defined subgroups, may ultimately drive treatment decisions.
Key Points
-
Early breast cancer assessment tools for prognosis and prediction of treatment benefit may aid clinical decision making; however, their limitations must be considered
-
Adjuvant! Online and the St Gallen Consensus incorporate discriminating clinicopathological factors; critical to these guidelines is accurate, standardized measurement of pathological parameters
-
Multigene signatures, Oncotype DX® and MammaPrint®, offer objective, reproducible analysis, particularly of proliferation but it remains to be seen whether they are more beneficial than accurate, standardized clinicopathological assessment
-
The greatest treatment challenge remains for patients assessed as intermediate risk of relapse
-
Breast cancers show remarkable diversity in biology and treatment efficacy; optimal management must be biologically driven and predictive biomarkers may guide use of systemic therapy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Perou, C. M. et al. Molecular portraits of human breast tumours. Nature 406, 747–752 (2000).
Cuzick, J. et al. Prognostic value of a combined, ER, PgR, Ki67, HER2 immunohistochemical (IHC4) score and comparison with the GHI recurrence score – results from TransATAC [abstract]. Cancer Res. 69 (24 Suppl.), a74 (2009).
Carter, C. L., Allen, C. & Henson, D. E. Relation of tumor size, lymph node status, and survival in 24,740 breast cancer cases. Cancer 63, 181–187 (1989).
Galea, M. H., Blamey, R. W., Elston, C. E. & Ellis, I. O. The Nottingham Prognostic Index in primary breast cancer. Breast Cancer Res. Treat. 22, 207–219 (1992).
Press, M. F. et al. HER-2/neu gene amplification characterized by fluorescence in situ hybridization: poor prognosis in node-negative breast carcinomas. J. Clin. Oncol. 15, 2894–2904 (1997).
Bonadonna, G. et al. 30 years' follow up of randomised studies of adjuvant CMF in operable breast cancer: cohort study. BMJ 330, 217 (2005).
Ravdin, P. M. et al. Computer program to assist in making decisions about adjuvant therapy for women with early breast cancer. J. Clin. Oncol. 19, 980–991 (2001).
Goldhirsch, A. et al. Thresholds for therapies: highlights of the St. Gallen International Expert Consensus on the primary therapy of early breast cancer 2009. Ann. Oncol. 20, 1319–1329 (2009).
Rhodes, A., Jasani, B., Barnes, D. M., Bobrow, L. G. & Miller, K. D. Reliability of immunohistochemical demonstration of oestrogen receptors in routine practice: interlaboratory variance in the sensitivity of detection and evaluation of scoring systems. J. Clin. Pathol. 53, 125–130 (2000).
Francis, G. D., Dimech, M., Giles, L. & Hopkins, A. Frequency and reliability of oestrogen receptor, progesterone receptor and HER2 in breast carcinoma determined by immunohistochemistry in Australasia: results of the RCPA Quality Assurance Program. J. Clin. Pathol. 60, 1277–1283 (2007).
Wolff, A. C. et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J. Clin. Oncol. 25, 118–145 (2007).
Bueno-de-Mesquita, J. M. et al. Use of 70-gene signature to predict prognosis of patients with node-negative breast cancer: a prospective community-based feasibility study (RASTER). Lancet Oncol. 8, 1079–1087 (2007).
Hammond, M. E. et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Clin. Oncol. 28, 2784–2795 (2010).
Mook, S. et al. Calibration and discriminatory accuracy of prognosis calculation for breast cancer with the online Adjuvant! program: a hospital-based retrospective cohort study. Lancet Oncol. 10, 1070–1076 (2009).
Olivotto, I. A. et al. Population-based validation of the prognostic model ADJUVANT! for early breast cancer. J. Clin. Oncol. 23, 2716–2725 (2005).
Goldhirsch, A. et al. Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. Ann. Oncol. 18, 1133–1144 (2007).
Hanna, W., Nofech-Mozes, S. & Kahn, H. J. Intratumoral heterogeneity of HER2/neu in breast cancer--a rare event. Breast J. 13, 122–129 (2007).
Miller, D. V. et al. Focal HER2/neu amplified clones partially account for discordance between immunohistochemistry and fluorescence in-situ hybridization results: data from NCCTG N9831 Intergroup Adjuvant Trial [abstract]. J. Clin. Oncol. 2004 ASCO Annual Meeting Proceedings (Post-Meeting Edition) 22 (July 15 Suppl.), a568 (2004).
Ross, J. S., Symmans, W. F., Pusztai, L. & Hortobagyi, G. N. Standardizing slide-based assays in breast cancer: hormone receptors, HER2, and sentinel lymph nodes. Clin. Cancer Res. 13, 2831–2835 (2007).
Stuart-Harris, R., Caldas, C., Pinder, S. E. & Pharoah, P. Proliferation markers and survival in early breast cancer: a systematic review and meta-analysis of 85 studies in 32,825 patients. Breast 17, 323–334 (2008).
Cheang, M. C. et al. Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J. Natl Cancer Inst. 101, 736–750 (2009).
Mengel, M. et al. Inter-laboratory and inter-observer reproducibility of immunohistochemical assessment of the Ki-67 labelling index in a large multi-centre trial. J. Pathol. 198, 292–299 (2002).
Rhodes, A., Jasani, B., Balaton, A. J. & Miller, K. D. Immunohistochemical demonstration of oestrogen and progesterone receptors: correlation of standards achieved on in house tumours with that achieved on external quality assessment material in over 150 laboratories from 26 countries. J. Clin. Pathol. 53, 292–301 (2000).
Ibrahim, M. et al. Potential for false-positive staining with a rabbit monoclonal antibody to progesterone receptor (SP2): findings of the UK National External Quality Assessment Scheme for Immunocytochemistry and FISH highlight the need for correct validation of antibodies on introduction to the laboratory. Am. J. Clin. Pathol. 129, 398–409 (2008).
Urruticoechea, A., Smith, I. E. & Dowsett, M. Proliferation marker Ki-67 in early breast cancer. J. Clin. Oncol. 23, 7212–7220 (2005).
van 't Veer, L. J. et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 415, 530–536 (2002).
van de Vijver, M. J. et al. A gene expression signature as a predictor of survival in breast cancer. N. Engl. J. Med. 347, 1999–2009 (2002).
Buyse, M. et al. Validation and clinical utility of a 70-gene prognostic signature for women with node-negative breast cancer. J. Natl Cancer Inst. 99, 1183–1192 (2006).
Paik, S. et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N. Engl. J. Med. 351, 2817–2826 (2004).
Goldstein, L. J. et al. Prognostic utility of the 21-gene assay in hormone receptor–positive operable breast cancer compared with classical clinicopathologic features. J. Clin. Oncol. 26, 4063–4071 (2008).
Paik, S. et al. Expression of the 21 genes in the Recurrence Score assay and tamoxifen clinical benefit in the NSABP study B-14 of node negative, estrogen receptor positive breast cancer [abstract]. J. Clin. Oncol. 2005 ASCO Annual Meeting Proceedings 23 (June 1 Suppl.), a510 (2005).
Berry, D. A. et al. Estrogen-receptor status and outcomes of modern chemotherapy for patients with node-positive breast cancer. JAMA 295, 1658–1667 (2006).
Carey, L. A. et al. The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin. Cancer Res. 13, 2329–2334 (2007).
Wülfing, P. et al. HER2-positive circulating tumor cells indicate poor clinical outcome in stage I to III breast cancer patients. Clin. Cancer Res. 12, 1715–1720 (2006).
Simmons, C. et al. Does confirmatory tumor biopsy alter the management of breast cancer patients with distant metastases? Ann. Oncol. 20, 1499–1504 (2009).
Liedtke, C. et al. Prognostic impact of discordance between triple-receptor measurements in primary and recurrent breast cancer. Ann. Oncol. 20, 1953–1958 (2009).
Navin, N. et al. Inferring tumor progression from genomic heterogeneity. Genome Res. 20, 68–80 (2010).
Di Leo, A. et al. A meta-analysis of phase III trials evaluating the predictive value of HER2 and topoisomerase II alpha in early breast cancer patients treated with CMF or anthracycline-based adjuvant therapy [abstract]. Breast Cancer Res. Treat. 107, a705 (2008).
Egawa, C. et al. Increased expression of BRCA1 mRNA predicts favorable response to anthracycline-containing chemotherapy in breast cancers. Breast Cancer Res. Treat. 78, 45–50 (2003).
Pusztai, L. et al. Clinical subtype-derived p53 gene signature is predictive of prognosis and response to chemotherapy in ER-positive but not in ER-negative breast cancers [abstract]. Cancer Res. 69 (24 Suppl.), a6122 (2009).
Pusztai, L. et al. Evaluation of microtubule-associated protein-Tau expression as a prognostic and predictive marker in the NSABP-B 28 randomized clinical trial. J. Clin. Oncol. 27, 4287–4292 (2009).
Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365, 1687–1717 (2005).
Viale, G. & Bottiglieri, L. Pathological definition of triple negative breast cancer. Eur. J. Cancer 45 (Suppl. 1), 5–10 (2009).
Chia, S. et al. Human epidermal growth factor receptor 2 overexpression as a prognostic factor in a large tissue microarray series of node-negative breast cancers. J. Clin. Oncol. 26, 5697–5704 (2008).
Curigliano, G. et al. Clinical relevance of HER2 overexpression/amplification in patients with small tumor size and node-negative breast cancer. J. Clin. Oncol. 27, 5693–5699 (2009).
Straver, M. E. et al. The 70-gene signature as a response predictor for neoadjuvant chemotherapy in breast cancer. Breast Cancer Res. Treat. 119, 551–558 (2010).
Knauer, M. et al. The predictive value of the 70-gene signature for adjuvant chemotherapy in early breast cancer. Breast Cancer Res. Treat. 120, 655–661 (2010).
Rodenhuis, S. et al. A simple system for grading the response of breast cancer to neoadjuvant chemotherapy. Ann. Oncol. 21, 481–487 (2010).
Paik, S. et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J. Clin. Oncol. 24, 3726–3734 (2006).
Albain, K. S. et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol. 11, 55–65 (2010).
Parker, J. S. et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 27, 1160–1167 (2009).
Braun, S. et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N. Engl. J. Med. 353, 793–802 (2005).
Allard, W. J. et al. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin. Cancer Res. 10, 6897–6904 (2004).
Hayes, D. F. & Smerage, J. Is there a role for circulating tumor cells in the management of breast cancer? Clin. Cancer Res. 14, 3646–3650 (2008).
Pachmann, K. et al. Monitoring the response of circulating epithelial tumor cells to adjuvant chemotherapy in breast cancer allows detection of patients at risk of early relapse. J. Clin. Oncol. 26, 1208–1215 (2008).
Stathopoulos, E. N. et al. Detection of CK-19 mRNA-positive cells in the peripheral blood of breast cancer patients with histologically and immunohistochemically negative axillary lymph nodes. Ann. Oncol. 16, 240–246 (2005).
Xenidis, N. et al. Predictive and prognostic value of peripheral blood cytokeratin-19 mRNA-positive cells detected by real-time polymerase chain reaction in node-negative breast cancer patients. J. Clin. Oncol. 24, 3756–3762 (2006).
Ignatiadis, M. et al. Prognostic value of the molecular detection of circulating tumor cells using a multimarker reverse transcription-PCR assay for cytokeratin 19, mammaglobin A, and HER2 in early breast cancer. Clin. Cancer Res. 14, 2593–2600 (2008).
Pierga, J. Y. et al. Circulating tumor cell detection predicts early metastatic relapse after neoadjuvant chemotherapy in large operable and locally advanced breast cancer in a phase II randomized trial. Clin. Cancer Res. 14, 7004–7010 (2008).
Xenidis, N. et al. Clinical relevance of circulating CK-19 mRNA-positive cells detected during the adjuvant tamoxifen treatment in patients with early breast cancer. Ann. Oncol. 18, 1623–1631 (2007).
Denkert, C. et al. Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J. Clin. Oncol. 28, 105–113 (2010).
Schmidt, M. et al. The humoral immune system has a key prognostic impact in node-negative breast cancer. Cancer Res. 68, 5405–5413 (2008).
Desmedt, C. et al. Biological processes associated with breast cancer clinical outcome depend on the molecular subtypes. Clin. Cancer Res. 14, 5158–5165 (2008).
Teschendorff, A. E., Miremadi, A., Pinder, S. E., Ellis, I. O. & Caldas, C. An immune response gene expression module identifies a good prognosis subtype in estrogen receptor negative breast cancer. Genome Biol. 8, R157 (2007).
Farmer, P. et al. A stroma-related gene signature predicts resistance to neoadjuvant chemotherapy in breast cancer. Nat. Med. 15, 68–74 (2009).
Author information
Authors and Affiliations
Contributions
L. Santarpia contributed to researching the data for this article and review/editing of the manuscript. A. Di Leo and C. Oakman contributed equally to researching the data for this article, the discussion, writing, and review/editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Oakman, C., Santarpia, L. & Di Leo, A. Breast cancer assessment tools and optimizing adjuvant therapy. Nat Rev Clin Oncol 7, 725–732 (2010). https://doi.org/10.1038/nrclinonc.2010.170
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2010.170