Key Points
-
Since the accidental discovery of its biological properties over 40 years ago, cisplatin has made a major impact in the chemotherapeutic treatment of testicular and ovarian cancers and is still widely used today.
-
The initial driver for further platinum-drug development was the discovery of the severe safety issues that are raised with cisplatin, especially nephrotoxicity. This resulted in the development of carboplatin, which is, broadly speaking, equally effective to cisplatin, but with a more acceptable side-effect profile.
-
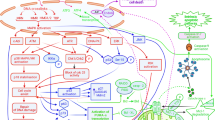
The mechanism of action of cisplatin (and carboplatin) involves covalent binding to purine DNA bases, which primarily leads to cellular apoptosis.
-
Much is now understood as to how tumours all too commonly exhibit resistance to cisplatin, either intrinsically or as acquired during courses of therapy. Major mechanisms include: decreased membrane transport of the drug, increased cytoplasmic detoxification, increased DNA repair, and increased tolerance to DNA damage.
-
The second driver for new platinum-drug development was to circumvent mechanisms of resistance, and thereby broaden the clinical utility of this class of agents. These efforts have resulted in oxaliplatin (active in patients with colorectal cancer), satraplatin (the first orally administered platinum drug, which shows promise in patients with prostate cancer) and picoplatin.
-
Improved delivery of platinum drugs to tumours is being studied in early clinical trials using liposomal-based or co-polymer-based products, as well as by the use of localized, intraperitoneal administration of cisplatin or carboplatin in patients with ovarian cancer.
-
It might also be possible to circumvent platinum-drug resistance in the clinic through modulating resistance mechanisms; for example, those involving increased glutathione or loss of DNA mismatch repair.
Abstract
The accidental discovery of the anticancer properties of cisplatin and its clinical introduction in the 1970s represent a major landmark in the history of successful anticancer drugs. Although carboplatin — a second-generation analogue that is safer but shows a similar spectrum of activity to cisplatin — was introduced in the 1980s, the pace of further improvements slowed for many years. However, in the past several years interest in platinum drugs has increased. Key developments include the elucidation of mechanisms of tumour resistance to these drugs, the introduction of new platinum-based agents (oxaliplatin, satraplatin and picoplatin), and clinical combination studies using platinum drugs with resistance modulators or new molecularly targeted drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rosenberg, B., VanCamp, L., Krigas, T. Inhibition of cell division in Escherichia coli by electrolysis products from a platinum electrode. Nature 205, 698–699 (1965).
Rosenberg, B., VanCamp, L., Trosko, J. E., Mansour, V. H. Platinum compounds: a new class of potent antitumour agents. Nature 222, 385–386 (1969).
Siddik, Z. H. Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 22, 7265–7279 (2003).
Chaney, S. G., Campbell, S. L., Temple, B., Bassett, E., Wu, Y., Faldu, M. Protein interactions with platinum–DNA adducts: from structure to function. J. Inorg. Biochem. 98, 1551–1559 (2004).
Eastman, A. Activation of programmed cell death by anticancer agents: cisplatin as a model system. Cancer Cells 2, 275–280 (1990).
Cvitkovic, E., Spaulding, J., Bethune, V., Martin, J., Whitmore, W. F. Improvement of cis-dichlorodiammineplatinum (NSC 119875): therapeutic index in an animal model. Cancer 39, 1357–1361 (1977).
Harrap, K. R. Preclinical studies identifying carboplatin as a viable cisplatin alternative. Cancer Treat. Rev. 12, 21–33 (1985).
Knox, R. J., Friedlos, F., Lydall, D. A., Roberts, J. J. Mechanism of cytotoxicity of anticancer platinum drugs: evidence that cis-diamminedichloroplatinum(II) and cis-diammine-(1,1-cyclobutanedicarboxylato)platinum(II) differ only in the kinetics of their interaction with DNA. Cancer Res. 46, 1972–1979 (1986).
Aabo, K., et al. Chemotherapy in advanced ovarian cancer: four systematic meta-analyses of individual patient data from 37 randomized trials. Advanced Ovarian Cancer Trialists' Group. Br. J. Cancer 78, 1479–1487 (1998).
Kelland, L. R., et al. Mechanism-related circumvention of cis-diamminedichloroplatinum(II) acquired resistance using two pairs of human ovarian carcinoma cell lines by ammine/amine platinum(IV) dicarboxylates. Cancer Res. 52, 3857–3864 (1992).
Gately, D. P., Howell, S. B. Cellular accumulation of the anticancer agent cisplatin: a review. Br. J. Cancer 67, 1171–1176 (1993).
Ishida, S., Lee, J., Thiele, D. J., Herskowitz, I. Uptake of the anticancer drug cisplatin mediated by the copper transporter Ctr1 in yeast and mammals. Proc. Natl Acad. Sci. USA 99, 14298–14302 (2002). This work identifies the role of the protein CTR1, normally involved in copper homeostasis, in transporting cisplatin into cells.
Katano, K., et al. Acquisition of resistance to cisplatin is accompanied by changes in the cellular pharmacology of copper. Cancer Res. 62, 6559–6565 (2002).
Holzer, A. K., Manorek, G. H., Howell, S. B. Contribution of the major copper influx transporter CTR1 to the cellular accumulation of cisplatin, carboplatin and oxaliplatin. Molec. Pharmacol. 70, 1390–1394 (2006).
Holzer, A. K., Howell, S. B. The internalization and degradation of human copper transporter 1 following cisplatin exposure. Cancer Res. 66, 10944–10952 (2006).
Safaei, R., Holzer, A. K., Katano, K., Samimi, G., Howell, S. B. The role of copper transporters in the development of resistance to Pt drugs. J. Inorg. Chem. 98, 1607–1613 (2004).
Samimi, G., et al. Increased expression of the copper efflux transporter ATP7A mediates resistance to cisplatin, carboplatin and oxaliplatin in ovarian cancer cells. Clin. Cancer Res. 10, 4661–4669 (2004).
Mistry, P., Kelland, L. R., Abel, G., Sidhar, S., Harrap, K. R. The relationships between glutathione, glutathione-S-transferase and cytotoxicity of platinum drugs and melphalan in eight human ovarian carcinoma cell lines. Br. J. Cancer 64, 215–220 (1991). One of the first preclinical studies identifying a correlation between increased levels of glutathione and resistance to cisplatin and carboplatin in ovarian carcinoma cells.
Ishikawa, T. The ATP-dependent glutathione S-conjugate export pump. Trends Biochem. Sci. 17, 463–468 (1992).
Lewis, A. D., Hayes, J. D., Wolf, C. R. Glutathione and glutathione-dependent enzymes in ovarian adenocarcinoma cell lines derived from a patient before and after the onset of drug resistance: intrinsic differences and cell cycle effects. Carcinogenesis, 9, 1283–1287 (1988).
Yang, P., Ebbert, J. O., Sun, Z., Weinshilboum, R. M. Role of the glutathione metabolic pathway in lung cancer treatment and prognosis: a review. J. Clin. Oncol. 24, 1761–1769 (2006).
Kelley, S. L. et al. Overexpression of metallothionein confers resistance to anticancer drugs. Science 241, 1813–1815 (1988).
Holford, J., Beale, P. J., Boxall, F. E., Sharp, S. Y., Kelland, L. R. Mechanisms of drug resistance to the platinum complex ZD0473 in ovarian cancer cell lines. Eur. J. Cancer 36, 1984–1990 (2000).
Johnson, S., et al. Relationship between platinum–DNA adduct formation and removal and cisplatin cytotoxicity in cisplatin-sensitive and -resistant human ovarian cancer cells. Cancer Res. 54, 5911–5916 (1994).
Ferry, K. V., Hamilton, T. C., Johnson, S. W. Increased nucleotide excision repair in cisplatin-resistant ovarian cancer cells: role of ERCC1-XPF. Biochem. Pharmacol. 60, 1305–1313 (2000).
Chang, I. Y., et al. Small interfering RNA-induced suppression of ERCC1, enhances sensitivity of human cancer cells to cisplatin. Biochem. Biophys. Res. Commun. 327, 225–233 (2005).
Dabholkar, M., Bostick-Bruton, F., Weber, C., Bohr, V. A., Egwuagu, C., Reed, E. ERCC1 and ERCC2 expression in malignant tissues from ovarian cancer patients. J. Natl. Cancer Inst. 84, 1512–1517 (1992). An original clinical translational study linking overexpression of the NER DNA-repair pathway gene ERCC1 to poor response to platinum-based chemotherapy in patients with ovarian cancer.
Reed, E. ERCC1 and clinical resistance to platinum-based therapy. Clin. Cancer Res. 11, 6100–6102 (2005).
Fink, D. et al. The role of DNA mismatch repair in platinum drug resistance. Cancer Res. 56, 4881–4886 (1996).
Zdraveski, Z. Z., Mello, J. A., Farinelli, C. K., Essigmann, J. M., Marinus, M. G. MutS preferentially recognises cisplatin-over oxaliplatin-modified DNA. J. Biol. Chem. 277, 1255–1260 (2002).
Gifford, G., Paul, J., Vasey, P. A., Kaye, S. B., Brown, R. The acquisition of hMLH1 methylation in plasma DNA after chemotherapy predicts poor survival for ovarian cancer patients. Clin. Cancer Res. 10, 4420–4426 (2004). A clinical translational study in ovarian cancer patients showing that loss of function of the DNA-mismatch-repair pathway through hypermethylation of the hMLH1 gene after chemotherapy, predicts poor survival.
Helleman, J., et al. Mismatch repair and treatment resistance in ovarian cancer. BMC Cancer 6, 201 (2006).
Bassett, E., et al. Frameshifts and deletions during in vitro translesion synthesis past Pt-DNA adducts by DNA polymerases β and η. DNA repair 1, 1003–1016 (2002).
Albertella, M. R., Green, C. M., Lehmann, A. R., O'Connor, M. J. A role for polymerase η in the cellular tolerance to cisplatin-induced damage. Cancer Res. 65, 9799–9806 (2005). Experiments indicating that inhibition of DNA polymerase η could increase the anticancer effectiveness of cisplatin.
Gadducci, A., Cosio, S., Muraca, S., Genazzani, A. R. Molecular mechanisms of apoptosis and chemosensitivity to platinum and paclitaxel in ovarian cancer: biological data and clinical implications. Eur. J. Gynaecol. Oncol. 23, 390–396 (2002).
Kelland, L. R. Overcoming resistance to platinum therapy in patients with advanced cancer. Am. J. Cancer 1, 247–255 (2002).
Kidani, Y., Inagaki, K., Iigo, M., Hoshi, A., Kuretani, K. Antitumor activity of 1,2-diaminocyclohexane-platinum complexes against sarcoma-180 ascites form. J. Med. Chem. 21, 1315–1318 (1978).
Rixe, O., et al. Oxaliplatin, tetraplatin, cisplatin and carboplatin: spectrum of activity in drug-resistant cell lines and in the cell lines of the National Cancer Institute's Anticancer Drug Screen panel. Biochem. Pharmacol. 52, 1855–1865 (1996).
Spingler, B., Whittington, D. A., Lippard, S. J. 2.4Å crystal structure of an oxaliplatin 1,2-d(GpG) intrastrand cross-link in a DNA dodecamer duplex. Inorg. Chem. 40, 5596–5602 (2001).
Raymond, E., Faivre, S., Chaney, S., Woynarowski, J., Cvitkovic, E. Cellular and molecular pharmacology of oxaliplatin. Mol. Cancer Ther. 1, 227–235 (2002).
Machover, D., et al. Two consecutive phase II studies of oxaliplatin (L-OHP) for treatment of patients with advanced colorectal carcinoma who were resistant to previous treatment with fluoropyrimidines. Ann. Oncol. 7, 95–98 (1996).
Levi, F., et al. A chronopharmacologic phase II clinical trial with 5-fluorouracil, folinic acid, and oxaliplatin using an ambulatory multichannel programmable pump. High antitumour effectiveness against metastatic colorectal cancer. Cancer 69, 893–900 (1992).
Giacchetti, S., et al. Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil–leucovorin as first-line treatment of metastatic colorectal cancer. J. Clin. Oncol. 18, 136–147 (2000).
De Gramont, A., et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J. Clin. Oncol. 18, 2938–2947 (2000).
Rothenberg, M. L., et al. Superiority of oxaliplatin and fluorouracil–leucovorin compared with either therapy alone in patients with progressive colorectal cancer after irinotecan and fluorouracil–leucovorin: interim results of a Phase III trial. J. Clin. Oncol. 21, 2059–2069 (2003).
Goldberg, R. M., et al. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J. Clin. Oncol. 22, 23–29 (2004). A key clinical phase III study showing significantly improved survival with oxaliplatin, fluorouracil/leucovorin (FOLFOX regimen) in patients with previously untreated metastatic colorectal cancer.
Kelland, L. R., et al. Preclinical antitumor evaluation of Bis-acetato-ammine-dichloro-cyclohexylamine platinum(IV): an orally active platinum drug. Cancer Res. 53, 2581–2586 (1993). The first paper demonstrating, in preclinical tumour models, the feasibility of achieving antitumour activity with an orally administered platin, JM216/satraplatin.
McKeage, M. J. et al. Schedule dependency of orally administered Bis-acetato-ammine-dichloro-cyclohexylamine-platinum(IV) (JM216) in vivo. Cancer Res. 54, 4118–4122 (1994).
Sharp, S. Y., Rogers, P. M., Kelland, L. R. Transport of cisplatin and Bis-acetato-ammine-dichlorocyclohexylamine platinum(IV) (JM216) in human ovarian carcinoma cell lines: identification of a plasma membrane protein associated with cisplatin resistance. Clin. Cancer Res. 1, 981–989 (1995).
Kelland, L. R. An update on satraplatin: the first orally available platinum anticancer drug. Exp. Opin. Invest. Drugs 9, 1373–1382 (2000).
Samimi, G., Howell, S. B. Modulation of the cellular pharmacology of JM118, the major metabolite of satraplatin, by copper influx and efflux transporters. Cancer Chemother. Pharmacol. 57, 781–788 (2006).
Silverman, A. P., Bu, W., Cohen, S. M., Lippard, S. J. 2.4-Å crystal structure of the asymmetric platinum complex [Pt(ammine)(cyclohexylamine)]2+ bound to a dodecamer DNA duplex. J. Biol. Chem. 277, 49743–49749 (2002).
Reardon, J. T., Vaisman, A., Chaney, S. G., Sancar, A. Efficient nucleotide excision repair of cisplatin, oxaliplatin, and bis-aceto-ammine-dichloro-cyclohexylamine-platinum(IV) (JM216) platinum intrastrand DNA diadducts. Cancer Res. 59, 3968–3971 (1999).
McKeage, M. J., et al. Phase I and pharmacokinetic study of an oral platinum complex given daily for 5 days in patients with cancer. J. Clin. Oncol. 15, 2691–2700 (1997).
Choy, H. Satraplatin: an orally available platinum analog for the treatment of cancer. Expert Rev. Anticancer Ther. 6, 973–982 (2006).
McKeage, M. J. Satraplatin in hormone-refractory prostate cancer and other tumour types: pharmacological properties and clinical evaluation. Drugs 67, 859–869 (2007).
Sternberg, C. N., et al. Phase III trial of Satraplatin, an oral platinum plus prednisone vs. prednisone alone in patients with hormone-refractory prostate cancer. Oncology, 68, 2–9 (2005). A pivotal clinical study demonstrating the potential clinical benefit of satraplatin in patients with hormone-refractory prostate cancer.
Holford, J., Sharp, S. Y., Murrer, B. A., Abrams, M., Kelland, L. R. In vitro circumvention of cisplatin resistance by the novel sterically hindered platinum complex AMD473. Br. J. Cancer 77, 366–373 (1998). The first studies with JM473/picoplatin, showing retention of activity in vitro against several cisplatin-resistant tumour cell lines of defined mechanisms of resistance.
Holford, J., et al. Chemical, biochemical and pharmacological activity of the novel sterically hindered platinum co-ordination complex, cis-[amminedichloro(2-methylpyridine)] platinum(II) (AMD473). Anticancer Drug Design 13, 1–18 (1998).
Sharp, S. Y., O'Neill, C. F., Rogers, P. M., Boxall, F. E, Kelland, L. R. Retention of activity by the new generation platinum agent AMD0473 in four human tumour cell lines possessing acquired resistance to oxaliplatin. Eur. J. Cancer 38, 2309–2315 (2002).
Raynaud, F. I. et al. Cis-amminedichloro(2-methylpyridine) platinum (II) (AMD473), a novel sterically hindered platinum complex: in vivo activity, toxicology and pharmacokinetics in mice. Clin. Cancer Res. 3, 2063–2074 (1997).
Rogers, P., Boxall, F. E., Allot, C. P., Stephens, T. C., Kelland, L. R. Sequence-dependent synergism between the new generation platinum agent ZD0473 and paclitaxel in cisplatin-sensitive and -resistant human ovarian carcinoma cell lines. Eur. J. Cancer 38, 1653–1660 (2002).
Gore, M. E., et al. A phase II trial of ZD0473 in platinum-pretreated ovarian cancer. Eur. J. Cancer, 38, 2416–2420 (2002).
Treat, J., et al. ZD0473 treatment in lung cancer: an overview of the clinical trial results. Eur. J. Cancer 38, S13–S18 (2002). A summary of early-phase clinical studies of picoplatin in patients with lung cancer, demonstrating potential utility in cisplatin-resistant small-cell lung cancer.
Beale, P. et al. A phase I clinical and pharmacological study of cis-diamminedichloro(2-methylpyridine) platinum II (AMD473). Br. J. Cancer 88, 1128–1134 (2003).
Brabec, V. et al. DNA modifications by a novel bifunctional trinuclear platinum phase I anticancer agent. Biochemistry 38, 6781–6790 (1999).
Akaza, H. et al. Phase II study of cis-diammine(glycolato)platinum, 254–S, in patients with advanced germ-cell testicular cancer, prostatic cancer, and transitional-cell carcinoma of the urinary tract. Cancer Chemother. Pharmacol. 31, 187–192 (1992).
Gordon, A. N., et al. Long-term survival advantage for women treated with pegylated liposomal doxorubicin compared with topotecan in a phase 3 randomized study of recurrent and refractory epithelial ovarian cancer. Gynecol. Oncol. 95, 1–8 (2004).
Gradishar, W. J., et al. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J. Clin. Oncol. 23, 7794–7803 (2005).
White, S. C. et al. Phase II study of SPI-77 (sterically stabilised liposomal cisplatin) in advanced non-small cell lung cancer. Br. J. Cancer, 95, 822–828 (2006).
Dragovich, T., Mendelson, D., Kurtin, S., Richardson, K., Von Hoff, D., Hoos, A. A Phase 2 trial of the liposomal DACH platinum L-NDDP in patients with therapy-refractory advanced colorectal cancer. Cancer Chemother. Pharmacol. 58, 759–764 (2006).
Rademaker-Lakhai, J. M., et al. A Phase I and pharmacological study of the platinum polymer AP5280 given as an intravenous infusion once every 3 weeks in patients with solid tumors. Clin. Cancer Res. 10, 3386–3395 (2004).
Rice, J. R., Gerberich, J. L., Nowotnik, D. P., Howell, S. B. Preclinical efficacy and pharmacokinetics of AP5346, a novel diaminocyclohexane-platinum tumor-targeting drug delivery system. Clin. Cancer Res. 12, 2248–2254 (2006).
Campone, M., et al. Phase I and pharmacokinetic trial of AP5346, a DACH-platinum-polymer conjugate, administered weekly for three out of every 4 weeks to advanced solid tumor patients. Cancer Chemother. Pharmacol. 17 Feb 2007 (doi:10.1007/s00280-006-0397-0).
Armstrong, D. K., et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N. Engl. J. Med. 354, 34–43 (2006). The most recent of 3 randomized trials in patients with ovarian cancer indicating an improvement in tumour response rates through intraperitoneal administration of platinum-based chemotherapy.
Morgan, A. S., et al. Tumor efficacy and bone-marrow sparing properties of TER286, a cytotoxin activated by glutathione S-transferase. Cancer Res. 58, 2568–2575 (1998).
Townsend, D. M., Shen, H., Staros, A. L., Gate, L., Tew, K. D. Efficacy of a glutathione S-transferase π-activated prodrug in platinum-resistant ovarian cancer cells. Mol. Cancer Ther. 1, 1089–1095 (2002).
Rosen, L. S., Phase I study of TLK286 (Telcyta) administered weekly in advanced malignancies. Clin. Cancer Res. 10, 3689–3698 (2004).
Kavanagh, J. J., et al. Multi-institutional phase 2 study of TLK286 (TELCYTA, a glutathione S-transferase P1–1 activated glutathione analog prodrug) in patients with platinum and paclitaxel refractory or resistant ovarian cancer. Int. J. Gynecol. Cancer 15, 593–600 (2005).
Plumb, J. A., Strathdee, G., Sludden, J., Kaye, S. B., Brown, R. Reversal of drug resistance in human tumor xenografts by 2′-deoxy-5-azacytidine-induced demethylation of the hMLH1 gene promoter. Cancer Res. 60, 6039–6044 (2000).
Hanahan, D., Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Collins, I., Workman, P. New approaches to molecular cancer therapeutics. Nature Chem. Biol. 2, 689–700 (2006).
Sandler, A., et al. Paclitaxel–carboplatin alone or with bevacizumab for non-small cell lung cancer. N. Engl. J. Med. 355, 2542–2550 (2006). The first demonstration of clinical proof of principle for the use of platinum-based chemotherapy (carboplatin) in combination with a molecularly targeted drug (bevacizumab).
McKeage, M. J., Kelland, L. R. 5,6-dimethylxanthenone-4-acetic acid (DMXAA). Clinical potential in combination with taxane-based chemotherapy. Am. J. Cancer, 5, 155–162 (2006).
Pietras, R. J., Fendly, B. M., Chazin, V. R., Pegram, M. D., Howell, S. B., Slamon, D. J. Antibody to HER-2/neu receptor blocks DNA repair after cisplatin in human breast and ovarian cancer cells. Oncogene, 9, 1829–1838 (1994).
Pegram, M. D., et al. Results of two open-label, multicenter phase II studies of docetaxel, platinum salts, and trastuzumab in HER2-positive advanced breast cancer. J. Natl. Cancer Inst. 96, 759–769 (2004).
Hurley, J., et al. Docetaxel, cisplatin and trastuzumab as primary systemic therapy for human epidermal growth factor receptor 2-positive locally advanced breast cancer. J. Clin. Oncol. 24, 1831–1838 (2006).
Bhattacharyya, A., Ear, U. S., Koller, B. H., Weichselbaum, R. R., Bishop, D. K. The breast cancer susceptibility gene BRCA1 is required for subnuclear assembly of Rad51 and survival following treatment with the DNA cross-linking agent cisplatin. J. Biol. Chem. 275, 23899–23903 (2000). A preclinical study showing that breast cancer cells harbouring BRCA1 mutations are hypersensitive to cisplatin.
Turner, N., Tutt, A., Ashworth, A. Targeting the DNA repair defect of BRCA tumours. Curr. Opin. Pharmacol. 5, 388–393 (2005).
Beuvink, I, et al. The mTOR inhibitor RAD001 sensitizes tumor cells to DNA-damaged induced apoptosis through inhibition of p21 translation. Cell 120, 747–759 (2005).
Morgan-Lappe, S., et al. RNAi-based screening of the human kinome identifies Akt-cooperating kinases: a new approach to designing efficacious multitargeted kinase inhibitors. Oncogene, 25, 1340–1348 (2006).
Roberts, D., et al. Identification of genes associated with platinum drug sensitivity and resistance in human ovarian cancer cells. Br. J. Cancer 92, 1149–1158 (2005).
Olivero, M., et al. Genes regulated by hepatocyte growth factor as targets to sensitize ovarian cells to cisplatin. Mol. Cancer Ther. 5, 1126–1135 (2006).
Davies, M. S., Berners-Price, S. J., Hambley, T. W. Rates of platination of –AG- and –GA- containing double-stranded oligonucleotides: effect of chloride concentration. J. Inorg. Biochem. 79, 167–172 (2000).
Fichtinger-Schepman, A. M., van der Veer, J. L., den Hartog, J. H., Lohman, P. H., Reedijk, J. Adducts of the antitumor drug cis-diamminedichloroplatinum(II) with DNA: formation, identification, and quantitation. Biochemistry, 24, 707–713 (1985).
Takahara, P. M., Rosenzweig, A. C., Frederick, C. A., Lippard, S. J. Crystal structure of double-stranded DNA containing the major adduct of the anticancer drug cisplatin. Nature 377, 649–652 (1995).
Huang, H., Zhu, L., Reid, B. R., Drobny, G. P., Hopkins, P. B. Solution structure of a cisplatin-induced interstrand cross-link. Science 270, 1842–1845 (1995).
Teuben, J. M., Bauer, C., Wang, A. H., Reedijk, J. Solution structure of a DNA duplex containing a cis-diammineplatinum(II) 1,3-d(GTG) intrastrand cross-link, a major adduct in cells treated with the anticancer drug carboplatin. Biochemistry 38, 12305–12312 (1999).
Einhorn, L. H. Curing metastatic testicular cancer. Proc. Natl Acad. Sci. USA 99, 4592–4595 (2002).
Walker, M. C., Parris, C. N., Masters, J. R. Differential sensitivities of human testicular and bladder tumor cell lines to chemotherapeutic drugs. J. Natl Cancer Inst. 79, 213–216 (1987).
Kelland, L. R., et al. Establishment and characterization of an in vitro model of acquired resistance to cisplatin in a human testicular nonseminomatous germ cell line. Cancer Res. 52, 1710–1716 (1992).
Koberle, B. et al. DNA repair capacity and cisplatin sensitivity of human testis tumour cells. Int. J. Cancer 70, 551–555 (1997).
Koberle, B., Masters, J. R., Hartley, J. A., Wood, R. D. Defective repair of cisplatin-induced DNA damage caused by reduced XPA protein in testicular germ cell tumours. Curr. Biol. 9, 273–276 (1999). The identification of a molecular defect in the NER DNA-repair pathway causing hypersensitivity of at least some testicular cancers to cisplatin.
Welsh, C., Day, R., McGurk, C., Masters, J. R. W., Wood, R. D. Koberle, B. Reduced levels of XPA, ERCC1 and XPF DNA repair proteins in testis tumor cell lines. Int. J. Cancer 110, 352–361 (2004).
Acknowledgements
The author wishes to thank K. Harrap and members of his laboratory, from 1987 to 1997. Studies on what became satraplatin and picoplatin during this period were supported by what is now Cancer Research UK.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
Lloyd Kelland received academic rewards as an employee of the Institute of Cancer Research (UK) from 1984 to 2001, with respect to the inventions of satraplatin and picoplatin.
Related links
Related links
FURTHER INFORMATION
Glossary
- Aquation
-
A chemical reaction in which water molecules are incorporated into a compound; in the case of cisplatin, with either displacement of one chlorine (monoaqua species) or both chlorines (diaqua species).
- 'Leaving' groups
-
During its reaction with DNA the dichloro groups of cisplatin are displaced or substituted but the two ammine groups remain intact, leading to a convention to refer to the groups within platinum cancer drugs that are displaced as 'leaving groups' (and those that remain as stable or carrier ligands).
- Prehydration
-
The process of administering large amounts of water (or fluid) to patients before chemotherapy.
- Myelosuppression
-
A decreased bone-marrow function that results in lower numbers of red blood cells, white blood cells and platelets.
- Thrombocytopenia
-
A decrease in the number of platelets in the blood.
- Carrier ligand
-
Stable groups on platinum drugs that are not replaced by substitution reactions.
- Objective response rate
-
The proportion of patients with defined tumour shrinkage; generally the sum of partial responses plus complete responses.
- Hazard ratio
-
The relative risk of experiencing a particular event; an HR of 0.6 means that one group has a 40% lower risk than the other group.
- HER2
-
An oncogene belonging to the EGFR family that has an important role in around a quarter of all breast cancers.
Rights and permissions
About this article
Cite this article
Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat Rev Cancer 7, 573–584 (2007). https://doi.org/10.1038/nrc2167
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc2167