Abstract
The glucose transporter GLUT1 at the blood-brain barrier (BBB) mediates glucose transport into the brain. Alzheimer's disease is characterized by early reductions in glucose transport associated with diminished GLUT1 expression at the BBB. Whether GLUT1 reduction influences disease pathogenesis remains, however, elusive. Here we show that GLUT1 deficiency in mice overexpressing amyloid β-peptide (Aβ) precursor protein leads to early cerebral microvascular degeneration, blood flow reductions and dysregulation and BBB breakdown, and to accelerated amyloid β-peptide (Aβ) pathology, reduced Aβ clearance, diminished neuronal activity, behavioral deficits, and progressive neuronal loss and neurodegeneration that develop after initial cerebrovascular degenerative changes. We also show that GLUT1 deficiency in endothelium, but not in astrocytes, initiates the vascular phenotype as shown by BBB breakdown. Thus, reduced BBB GLUT1 expression worsens Alzheimer's disease cerebrovascular degeneration, neuropathology and cognitive function, suggesting that GLUT1 may represent a therapeutic target for Alzheimer's disease vasculo-neuronal dysfunction and degeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Zlokovic, B.V. The blood-brain barrier in health and chronic neurodegenerative disorders. Neuron 57, 178–201 (2008).
Zlokovic, B.V. Neurovascular pathways to neurodegeneration in Alzheimer's disease and other disorders. Nat. Rev. Neurosci. 12, 723–738 (2011).
Simpson, I.A., Carruthers, A. & Vannucci, S.J. Supply and demand in cerebral energy metabolism: the role of nutrient transporters. J. Cereb. Blood Flow Metab. 27, 1766–1791 (2007).
Deng, D. et al. Crystal structure of the human glucose transporter GLUT1. Nature 510, 121–125 (2014).
Allen, A. & Messier, C. Plastic changes in the astrocyte GLUT1 glucose transporter and beta-tubulin microtubule protein following voluntary exercise in mice. Behav. Brain Res. 240, 95–102 (2013).
Choeiri, C., Staines, W., Miki, T., Seino, S. & Messier, C. Glucose transporter plasticity during memory processing. Neuroscience 130, 591–600 (2005).
Zeller, K., Rahner-Welsch, S. & Kuschinsky, W. Distribution of Glut1 glucose transporters in different brain structures compared to glucose utilization and capillary density of adult rat brains. J. Cereb. Blood Flow Metab. 17, 204–209 (1997).
Pearson, T.S., Akman, C., Hinton, V.J., Engelstad, K. & De Vivo, D.C. Phenotypic spectrum of glucose transporter type 1 deficiency syndrome (Glut1 DS). Curr. Neurol. Neurosci. Rep. 13, 342 (2013).
Wang, D. et al. A mouse model for Glut-1 haploinsufficiency. Hum. Mol. Genet. 15, 1169–1179 (2006).
Ullner, P.M. et al. Murine Glut-1 transporter haploinsufficiency: postnatal deceleration of brain weight and reactive astrocytosis. Neurobiol. Dis. 36, 60–69 (2009).
Zheng, P.-P. et al. Glut1/SLC2A1 is crucial for the development of the blood-brain barrier in vivo. Ann. Neurol. 68, 835–844 (2010).
Nguyen, L.N. et al. Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid. Nature 509, 503–506 (2014).
Ben-Zvi, A. et al. Mfsd2a is critical for the formation and function of the blood-brain barrier. Nature 509, 507–511 (2014).
Zhao, Z. & Zlokovic, B.V. Blood-brain barrier: a dual life of MFSD2A? Neuron 82, 728–730 (2014).
Landau, S.M. et al. Amyloid deposition, hypometabolism, and longitudinal cognitive decline. Ann. Neurol. 72, 578–586 (2012).
Winkler, E.A., Sagare, A.P. & Zlokovic, B.V. The pericyte: a forgotten cell type with important implications for Alzheimer's disease? Brain Pathol. 24, 371–386 (2014).
Ossenkoppele, R. et al. Differential effect of APOE genotype on amyloid load and glucose metabolism in AD dementia. Neurology 80, 359–365 (2013).
Protas, H.D. et al. Posterior cingulate glucose metabolism, hippocampal glucose metabolism, and hippocampal volume in cognitively normal, late-middle-aged persons at 3 levels of genetic risk for Alzheimer disease. JAMA Neurol. 70, 320–325 (2013).
Mosconi, L. et al. Amyloid and metabolic positron emission tomography imaging of cognitively normal adults with Alzheimer's parents. Neurobiol. Aging 34, 22–34 (2013).
Landau, S.M. et al. Comparing predictors of conversion and decline in mild cognitive impairment. Neurology 75, 230–238 (2010).
Mosconi, L. et al. FDG-PET changes in brain glucose metabolism from normal cognition to pathologically verified Alzheimer's disease. Eur. J. Nucl. Med. Mol. Imaging 36, 811–822 (2009).
Mosconi, L. et al. Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer's disease. J. Nucl. Med. 47, 1778–1786 (2006).
Niwa, K., Kazama, K., Younkin, S.G., Carlson, G.A. & Iadecola, C. Alterations in cerebral blood flow and glucose utilization in mice overexpressing the amyloid precursor protein. Neurobiol. Dis. 9, 61–68 (2002).
Jagust, W.J. et al. Diminished glucose transport in Alzheimer's disease: dynamic PET studies. J. Cereb. Blood Flow Metab. 11, 323–330 (1991).
Piert, M., Koeppe, R.A., Giordani, B., Berent, S. & Kuhl, D.E. Diminished glucose transport and phosphorylation in Alzheimer's disease determined by dynamic FDG-PET. J. Nucl. Med. 37, 201–208 (1996).
Simpson, I.A., Chundu, K.R., Davies-Hill, T., Honer, W.G. & Davies, P. Decreased concentrations of GLUT1 and GLUT3 glucose transporters in the brains of patients with Alzheimer's disease. Ann. Neurol. 35, 546–551 (1994).
Mooradian, A.D., Chung, H.C. & Shah, G.N. GLUT-1 expression in the cerebra of patients with Alzheimer's disease. Neurobiol. Aging 18, 469–474 (1997).
Kalaria, R.N. & Harik, S.I. Reduced glucose transporter at the blood-brain barrier and in cerebral cortex in Alzheimer disease. J. Neurochem. 53, 1083–1088 (1989).
Horwood, N. & Davies, D.C. Immunolabelling of hippocampal microvessel glucose transporter protein is reduced in Alzheimer's disease. Virchows Arch. 425, 69–72 (1994).
Hsiao, K. et al. Correlative memory deficits, Aβ elevation, and amyloid plaques in transgenic mice. Science 274, 99–102 (1996).
Young, C.D. et al. Modulation of glucose transporter 1 (GLUT1) expression levels alters mouse mammary tumor cell growth in vitro and in vivo. PLoS ONE 6, e23205 (2011).
Deane, R. et al. RAGE mediates amyloid-beta peptide transport across the blood-brain barrier and accumulation in brain. Nat. Med. 9, 907–913 (2003).
Paris, D. et al. Impaired angiogenesis in a transgenic mouse model of cerebral amyloidosis. Neurosci. Lett. 366, 80–85 (2004).
Paris, D. et al. Inhibition of angiogenesis by Aβ peptides. Angiogenesis 7, 75–85 (2004).
Abdul Muneer, P.M., Alikunju, S., Szlachetka, A.M., Murrin, L.C. & Haorah, J. Impairment of brain endothelial glucose transporter by methamphetamine causes blood-brain barrier dysfunction. Mol. Neurodegener. 6, 23 (2011).
Iadecola, C. et al. SOD1 rescues cerebral endothelial dysfunction in mice overexpressing amyloid precursor protein. Nat. Neurosci. 2, 157–161 (1999).
Niwa, K. et al. Aβ1–40-related reduction in functional hyperemia in mouse neocortex during somatosensory activation. Proc. Natl. Acad. Sci. USA 97, 9735–9740 (2000).
Paul, J., Strickland, S. & Melchor, J.P. Fibrin deposition accelerates neurovascular damage and neuroinflammation in mouse models of Alzheimer's disease. J. Exp. Med. 204, 1999–2008 (2007).
Deane, R. et al. LRP/amyloid beta-peptide interaction mediates differential brain efflux of Aβ isoforms. Neuron 43, 333–344 (2004).
Obermoeller-McCormick, L.M. et al. Dissection of receptor folding and ligand-binding property with functional minireceptors of LDL receptor-related protein. J. Cell Sci. 114, 899–908 (2001).
Uchida, Y. et al. A study protocol for quantitative targeted absolute proteomics (QTAP) by LC-MS/MS: application for inter-strain differences in protein expression levels of transporters, receptors, claudin-5, and marker proteins at the blood-brain barrier in ddY, FVB, and C57BL/6J mice. Fluids Barriers CNS 10, 21 (2013).
Bell, R.D. et al. SRF and myocardin regulate LRP-mediated amyloid-beta clearance in brain vascular cells. Nat. Cell Biol. 11, 143–153 (2009).
Llorente-Cortés, V., Costales, P., Bernués, J., Camino-Lopez, S. & Badimon, L. Sterol regulatory element-binding protein-2 negatively regulates low density lipoprotein receptor-related protein transcription. J. Mol. Biol. 359, 950–960 (2006).
Bell, R.D. et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 485, 512–516 (2012).
Irizarry, M.C., McNamara, M., Fedorchak, K., Hsiao, K. & Hyman, B.T. APPSw transgenic mice develop age-related A beta deposits and neuropil abnormalities, but no neuronal loss in CA1. J. Neuropathol. Exp. Neurol. 56, 965–973 (1997).
Benarroch, E.E. Brain glucose transporters: implications for neurologic disease. Neurology 82, 1374–1379 (2014).
Vannucci, S.J. Developmental expression of GLUT1 and GLUT3 glucose transporters in rat brain. J. Neurochem. 62, 240–246 (1994).
Bolaños, J.P., Delgado-Esteban, M., Herrero-Mendez, A., Fernandez-Fernandez, S. & Almeida, A. Regulation of glycolysis and pentose-phosphate pathway by nitric oxide: impact on neuronal survival. Biochim. Biophys. Acta 1777, 789–793 (2008).
Sagare, A.P. et al. Pericyte loss influences Alzheimer-like neurodegeneration in mice. Nat. Commun. 4, 2932 (2013).
Seidner, G. et al. GLUT-1 deficiency syndrome caused by haploinsufficiency of the blood-brain barrier hexose carrier. Nat. Genet. 18, 188–191 (1998).
Bell, R.D. et al. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 68, 409–427 (2010).
Maeda, K., Mies, G., Oláh, L. & Hossmann, K.A. Quantitative measurement of local cerebral blood flow in the anesthetized mouse using intraperitoneal [14C]iodoantipyrine injection and final arterial heart blood sampling. J. Cereb. Blood Flow Metab. 20, 10–14 (2000).
Chow, N. et al. Serum response factor and myocardin mediate arterial hypercontractility and cerebral blood flow dysregulation in Alzheimer's phenotype. Proc. Natl. Acad. Sci. USA 104, 823–828 (2007).
Wu, Z. et al. Role of the MEOX2 homeobox gene in neurovascular dysfunction in Alzheimer disease. Nat. Med. 11, 959–965 (2005).
Zlokovic, B.V., Mackic, J.B., Wang, L., McComb, J.G. & McDonough, A. Differential expression of Na,K-ATPase alpha and beta subunit isoforms at the blood-brain barrier and the choroid plexus. J. Biol. Chem. 268, 8019–8025 (1993).
Menon, P. et al. Impaired spine formation and learning in GPCR kinase 2 interacting protein-1 (GIT1) knockout mice. Brain Res. 1317, 218–226 (2010).
LaRue, B. et al. Method for measurement of the blood-brain barrier permeability in the perfused mouse brain: application to amyloid-beta peptide in wild type and Alzheimer's Tg2576 mice. J. Neurosci. Methods 138, 233–242 (2004).
Shibata, M. et al. Clearance of Alzheimer's amyloid-β1–40 peptide from brain by LDL receptor–related protein-1 at the blood-brain barrier. J. Clin. Invest. 106, 1489–1499 (2000).
Fowler, S.C. et al. A force-plate actometer for quantitating rodent behaviors: illustrative data on locomotion, rotation, spatial patterning, stereotypies, and tremor. J. Neurosci. Methods 107, 107–124 (2001).
Deacon, R.M.J. Assessing nest building in mice. Nat. Protoc. 1, 1117–1119 (2006).
Schildge, S., Bohrer, C., Beck, K. & Schachtrup, C. Isolation and culture of mouse cortical astrocytes. J. Vis. Exp. doi:10.3791/50079 (19 January 2013).10.3791/50079
Baskurt, O.K. et al. Comparison of three commercially available ektacytometers with different shearing geometries. Biorheology 46, 251–264 (2009).
Agarwal, N. et al. Familial polycythemia caused by a novel mutation in the beta globin gene: essential role of P50 in evaluation of familial polycythemia. Int. J. Med. Sci. 4, 232–236 (2007).
Acknowledgements
We thank E. Schauwecker for most helpful discussion. This research was supported by US National Institutes of Health grants AG039452, AG023084 and NS034467 to B.V.Z. and U01 HL087947 and R01DK092065 to E.D.A.
Author information
Authors and Affiliations
Contributions
E.A.W. contributed to manuscript preparation, experimental design and analysis, and conducted experiments. Y.N. and A.P.S. contributed to experimental design, data analysis and interpretation, and conducted experiments. S.V.R., R.D.B., D.P., J.D.S., S.H., J.S.S., P.K., A.R.N., R.B.W., J.M., E.Z. and Z.Z. conducted and analyzed experiments. H.J.M. contributed to hematological analysis and data interpretation. E.D.A. provided Slc2a1loxP/loxP mice and contributed to project design. J.S. generated Slc2a1loxP/+Tie2-Cre mice. D.C.D.V. provided Slc2a1+/− mice and contributed to project design. B.V.Z. supervised and designed all experiments and analysis, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Slc2a1 haploinsufficiency leads to reductions in GLUT1 transporter and hypoglycorrhachia.
(a) Representative confocal microscopy analysis showing GLUT1 immunodetection (red) and lectin-positive capillaries (green) in 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Colocalization, yellow. Scale bar, 100 µm. (b) Quantification of GLUT1-positive immunofluorescent signal normalized to vascular density in cortex and hippocampus in 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01. (c) Representative immunoblotting of GLUT1 protein levels in isolated cerebral microvessels from 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. (d) Quantification of GLUT1 protein expression in isolated cerebral microvessels from 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01. (e) Quantification of fasting glucose values in blood plasma and cerebrospinal fluid (CSF) from 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01. (f) Quantification of CSF-to-blood plasma ratios in 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01.
Supplementary Figure 2 Blood-brain barrier GLUT1 reductions do not alter systemic hemodynamic or physiologic parameters in Slc2a1+/–, Slc2a1+/+APPSw/0 and Slc2a1+/–APPSw/0 mice.
(a-f) Mean arterial blood pressure (a), respiratory rate (b), heart rate (c), body temperature (d), hematocrit (e) and bodyweight (f) in 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01. (g-i) Arterial blood gas values showing arterial pH (g), PCO2 (h), and PO2 (i) in 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01.
Supplementary Figure 3 GLUT1 reductions lead to progressive breakdown of the blood-brain barrier (BBB) with aging.
(a) Representative immunoblotting of extravascular IgG in brain tissue of 6-month-old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. β-actin was used as loading control. (b) Quantification of relative protein abundance of extravascular IgG (c) Representative confocal microscopy analysis of plasma-derived IgG (red) and lectin-positive capillaries (white) in 6-month-old Slc2a1+/+,Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Scale bar, 20 µm. Mean ± SEM, n=3-5 mice per group; *p<0.05 or **p<0.01.
Supplementary Figure 4 GLUT1 reductions in Slc2a1+/–APPSw/0 mice do not lead to enhanced APP production and/or processing or reduced Aβ enzymatic degradation.
(a) Represenative immunoblotting of human amyloid precursor protein (APP) and β-secretase (BACE1) in 16-month old Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Glyceraldehyde phosphate dehydrogenase (GAPDH) was used as loading control. (b-c) Relative protein abundance of human APP (b) and BACE1 (c) using densitometry analysis from immunoblots in 16-month old Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; ns, non-significant. (d-g) Represenative immunoblotting of Aβ degradative enzymes neprilysin (d) and insulin degrading enzyme (IDE) (f) in 16-month old Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Glyceraldehyde phosphate dehydrogenase (GAPDH) was used as loading control. Relative protein abundance of neprilysin (e) and IDE (g) using densitometry analysis from immunoblots in 16-month old Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-5 mice per group; ns, non-significant.
Supplementary Figure 5 Re-expression of LRP1 and GLUT1 in the hippocampus of Slc2a1+/–APPSw/0 mice using adenovirus-mediated transfer with Ad.mLRP1 and Ad.Slc2a1.
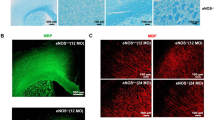
(a) Representative immunoblotting analysis of LRP-515 and mLRP1 levels in 10-month old Slc2a1+/−APPSw/0 mouse hippocampus injected with Ad.Ctrl and Ad.mLRP1. (b) Representative confocal microscopy analysis of LRP1 (green) and lectin-positive (red) microvessels in 10-month old Slc2a1+/−APPSw/0 mouse hippocampus injected with Ad.Ctrl and Ad.mLRP1. Co-localization (yellow) denotes vascular LRP1. Scale bar, 20 µm. (c) Quantification of microvascular LRP1-positive area in 10-month old Slc2a1+/−APPSw/0 mice after hippocampal injection with Ad.Ctrl and Ad.mLRP1. Mean ± SEM, n=3 mice; *p<0.05. (d) Representative confocal microscopy analysis of GLUT1 (green) and lectin-positive (red) microvessels in 10-month old Slc2a1+/−APPSw/0 mouse hippocampus injected with Ad.Ctrl and Ad.Slc2a1. Co-localization (yellow) denotes vascular GLUT1. Scale bar, 20 µm. (e) Quantification of microvascular GLUT1-positive microvessels in 10-month old Slc2a1+/−APPSw/0 mice after hippocampal injection with Ad.Ctrl and Ad.Slc2a1. Mean ± SEM, n=3 mice; **p<0.01. (f) Representative confocal microscopy analysis of LRP1 (green) and lectin-positive (red) microvessels in 10-month old Slc2a1+/−APPSw/0 mouse hippocampus injected with Ad.Ctrl and Ad.Slc2a1. Co-localization (yellow) denotes vascular LRP1. Scale bar, 20 µm. (g) Quantification of microvascular LRP1-positive microvessels in 10-month old Slc2a1+/−APPSw/0 mice after hippocampal injection with Ad.Ctrl and Ad.Slc2a1. Mean ± SEM, n=3 mice; *p<0.05.
Supplementary Figure 6 GLUT1, LRP1 and SREBP2 protein levels in brain endothelial cells isolated from Slc2a1+/+ and Slc2a1+/– mice.
(a) Representative immunoblotting (upper panel) and LRP1 (LRP-85) and GLUT1 protein abundance relative to β-actin determined by scanning densitometry (lower panel) in Slc2a1+/+ brain endothelial cells (BEC) transfected with siControl or Slc2a1 siRNA (siSlc2a1). (b) Real-time quantitative PCR (qPCR) for Lrp1 mRNA relative to GAPDH in Slc2a1+/+ BEC transfected with siControl or siSlc2a1. (c) Representative immunoblotting (upper panel) and quantification of LRP1 and GLUT1 protein abundance relative to β-actin (lower panel) in Slc2a1+/− BEC after transduction with either Ad.Ctrl or Ad.Slc2a1 to re-express GLUT1. (d) Representative immunoblotting (upper panel) and quantification of LRP1 and GLUT1 protein abundance relative to β-actin (lower panel) in Slc2a1+/+ BEC transfected with siControl or Lrp1 siRNA (siLRP1). (e) Representative immunoblotting for GLUT1, SREBP2 and LRP1 (LRP-85) in Slc2a1+/+ BEC transfected with siControl, siSlc2a1 or Srebp2 siRNA (siSrebp2). (f-h) Quantification of GLUT1 (f) SREBP2 (g) and LRP1 (h) protein abundance relative to β-actin from experiments as shown in e. (i-k) Representative immunoblotting (upper panels) and quantification (lower panels) of GLUT1 (i), SREBP2 (j) and LRP1 (k) protein abundance relative to β-actin in Slc2a1+/+ and Slc2a1+/− BEC. Mean ± SEM, n=3 independent cultures per group; *p<0.05, **p<0.01.
Supplementary Figure 7 GLUT1 isoforms in brain microvessels, capillary-depleted brain homogenates, brain endothelial cells and astrocytes in Slc2a1+/+ and Slc2a1+/– mice.
Representative immunoblotting of GLUT1 endothelial 55 kDa isoform in brain microvessels and GLUT1 astrocytic 45 kDa isoform in capillary-depleted brain homogenates from Slc2a1+/+ control mice determined at (a) short 15 s and (b) longer 3 min exposure times (left panel) with quantification relative to β-actin (right panel). Of note, the levels of 45 kDa isoform in capillary-depleted brain homogenates are low and undetectable over short exposure times as shown in a vs. b. (c) Representative immunoblotting of CD31 (endothelial marker), Tuj-1 (neuronal marker) and GFAP (astrocytic marker) in brain microvessels and capillary-depleted brains from Slc2a1+/+ mice. (d-e) Representative immunoblotting (upper panel) and quantification of GLUT1 levels (lower panel) in brain microvessels (d) and capillary-depleted brains (e) from Slc2a1+/+ and Slc2a1+/− mice. (f-g) Representative immunoblotting (upper panel) and quantification of GLUT2 (f) and GLUT3 (g) protein abundance relative to β-actin (lower panel) in capillary-depleted brains from Slc2a1+/+ and Slc2a1+/− mice. Notably, GLUT2 and GLUT3 were not found in brain microvessels (not shown), as reported1,3,52. (h) Representative immunoblotting (upper panel) and quantification of GLUT1 55 kDa and 45 kDa isoforms abundance (lower panel) relative to β-actin in brain endothelial cells (BEC) and astrocytes isolated from Slc2a1+/+ mice. (i) Representative immunoblotting (upper panel) and quantification of GLUT1 55 kDa protein abundance (lower panel) relative to β-actin in BEC from Slc2a1+/+ and Slc2a1+/− mice. (j) Representative immunoblotting (upper panel) and quantification of GLUT1 45 kDa protein abundance and GLUT2 protein abundance (lower panel) relative to β-actin in astrocytes from Slc2a1+/+ and Slc2a1+/− mice. GLUT2 was not present in BEC (not shown), as reported1,3,52. Mean ± SEM, n=3-4 mice per group (b, d-g) and 3 independent cultures per group (h-j); **p<0.01.
Supplementary Figure 8 GLUT1 expression in Slc2a1loxP/+;Tie2-Cre+/0 mice and Slc2a1loxP/+;GFAP-Cre+/0 mice with specific GLUT1 partial deletions from endothelium and astrocytes, respectively.
(a) Representative confocal microscopy analysis showing GLUT1 immunodetection (red) and lectin-positive endothelial vascular profiles (green) in 2-week-old Slc2a1lox/+, Slc2a1lox/+Tie2-Cre+/0, and Slc2a1lox/+GFAP-Cre+/0 mice. Colocalization (yellow) denotes endothelial GLUT1. Scale bar, 25 µm. (b) Quantification of GLUT1-positive immunofluorescent endothelial signal expressed as GLUT1-positive area (percentage) occupying lectin-positive endothelial capillary profiles in the cortex and hippocampus in 2-week-old Slc2a1lox/+, Slc2a1lox/+Tie2-Cre+/0, and Slc2a1lox/+GFAP-Cre+/0 mice. Mean ± SEM, n=4-5 mice per group; **p<0.01. (c-d) Representative immunoblotting of GLUT1 protein abundance relative to β-actin in brain microvessels after 15 s short exposure (c) and capillary-depleted brain homogenates after 3 min longer exposure (d) in 2-week-old Slc2a1lox/+, Slc2a1lox/+Tie2-Cre+/0, and Slc2a1lox/+GFAP-Cre+/0 mice. Mean ± SEM, n=3-4 mice per group; *p<0.05.
Supplementary Figure 9 Slc2a1 haplosufficiency does not affect red blood cell (RBC) indices and hemoglobin (Hb) oxygen saturation or dissociation in Slc2a1+/–, Slc2a1+/+APPSw/0 and Slc2a1+/–APPSw/0 mice.
(a) Quantification of Glut1 membrane protein levels in circulating RBCs from 2 month old Slc2a1+/− mice. (b) Representative bright field microscopy showing RBC morphology in 2 month old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. (c) Graph showing RBC deformability profiles, a measure of mechanical properties, in 2 month old Slc2a1+/+ (green), Slc2a1+/− (blue), Slc2a1+/+APPSw/0 (red) and Slc2a1+/−APPSw/0 (purple) mice. (d-j) Quantification of RBC hemoglobin (d), mean corpuscular volume (e), mean corpuscular hemoglobin (MCH) (f), mean corpuscular hemoglobin concentration (MCHC) (g), red cell distribution width (RDW) (h), mean arterial oxygen saturation (SaO2) (i), and mean P50 value of oxygen dissociation (j), in 2 month old Slc2a1+/+, Slc2a1+/−, Slc2a1+/+APPSw/0 and Slc2a1+/−APPSw/0 mice. Mean ± SEM, n=3-4 mice per group. ns, nonsignificant.
Supplementary Figure 10 GLUT1 deficiency leads to early cerebral microvascular degeneration, BBB breakdown, hypoperfusion, metabolic stress and accelerated accumulation of Aβ preceding neuronal functional and degenerative changes.
GLUT1 deficiency in endothelium leads to early cerebral microvascular degeneration (blue), limited BBB glucose uptake into the brain (blue), and disrupted Aβ metabolism (red) resulting in progressive BBB breakdown (blue), perfusion deficit (blue), potentially hypometabolic state (blue) and impaired vascular clearance of Aβ (red) via disruption of LRP1 (red). Ultimately, early microvascular degenerative changes resulting in accumulation of blood-borne toxins through a disrupted BBB, chronic perfusion and metabolic stress during limited glucose availability, as well as accumulation of neurotoxic Aβ species converge at the neuronal interface leading to neuronal dysfunction (blue), neuronal injury and neurodegeneration. Importantly, vascular damage precedes and helps drive neuronal changes as can be seen on the time scale.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Tables 1 and 2 (PDF 1911 kb)
Rights and permissions
About this article
Cite this article
Winkler, E., Nishida, Y., Sagare, A. et al. GLUT1 reductions exacerbate Alzheimer's disease vasculo-neuronal dysfunction and degeneration. Nat Neurosci 18, 521–530 (2015). https://doi.org/10.1038/nn.3966
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3966
This article is cited by
-
The therapeutic potential of probucol and probucol analogues in neurodegenerative diseases
Translational Neurodegeneration (2024)
-
Neuropathogenesis-on-chips for neurodegenerative diseases
Nature Communications (2024)
-
Brain metabolism in Alzheimer’s disease: biological mechanisms of exercise
Translational Neurodegeneration (2023)
-
Downregulation of the glucose transporter GLUT 1 in the cerebral microvasculature contributes to postoperative neurocognitive disorders in aged mice
Journal of Neuroinflammation (2023)
-
Anti-malaria drug artesunate prevents development of amyloid-β pathology in mice by upregulating PICALM at the blood-brain barrier
Molecular Neurodegeneration (2023)