Abstract
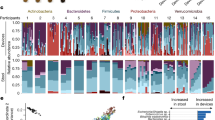
Little is known about how colonic transit time relates to human colonic metabolism and its importance for host health, although a firm stool consistency, a proxy for a long colonic transit time, has recently been positively associated with gut microbial richness. Here, we show that colonic transit time in humans, assessed using radio-opaque markers, is associated with overall gut microbial composition, diversity and metabolism. We find that a long colonic transit time associates with high microbial richness and is accompanied by a shift in colonic metabolism from carbohydrate fermentation to protein catabolism as reflected by higher urinary levels of potentially deleterious protein-derived metabolites. Additionally, shorter colonic transit time correlates with metabolites possibly reflecting increased renewal of the colonic mucosa. Together, this suggests that a high gut microbial richness does not per se imply a healthy gut microbial ecosystem and points at colonic transit time as a highly important factor to consider in microbiome and metabolomics studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Le Chatelier, E. et al. Richness of human gut microbiome correlates with metabolic markers. Nature 500, 541–546 (2013).
Lozupone, C. A., Stombaugh, J. I., Gordon, J. I., Jansson, J. K. & Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 489, 220–230 (2012).
Vandeputte, D. et al. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut 65, 57–62 (2015).
Tigchelaar, E. F. et al. Gut microbiota composition associated with stool consistency. Gut 65, 540–542 (2016).
Wang, Y. T. et al. Regional gastrointestinal transit and pH studied in 21 healthy volunteers using the wireless motility capsule: influence of age, gender, study country and testing protocol. Aliment. Pharmacol. Ther. 42, 761–772 (2015).
Nyangale, E. P., Mottram, D. S. & Gibson, G. R. Gut microbial activity, implications for health and disease: the potential role of metabolite analysis. J. Proteome Res. 11, 5573–5585 (2012).
Tremaroli, V. & Bäckhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 489, 242–249 (2012).
Davila, A.-M. et al. Intestinal luminal nitrogen metabolism: role of the gut microbiota and consequences for the host. Pharmacol. Res. 68, 95–107 (2013).
Russell, W. R., Hoyles, L., Flint, H. J. & Dumas, M.-E. Colonic bacterial metabolites and human health. Curr. Opin. Microbiol. 16, 246–254 (2013).
Andriamihaja, M. et al. The deleterious metabolic and genotoxic effects of the bacterial metabolite p-cresol on colonic epithelial cells. Free Radic. Biol. Med. 85, 219–227 (2015).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nature Rev. Microbiol. 12, 661–672 (2014).
Shafi, T. et al. Free levels of selected organic solutes and cardiovascular morbidity and mortality in hemodialysis patients: results from the retained organic solutes and clinical outcomes (ROSCO) investigators. PLoS ONE 10, e0126048 (2015).
Gabriele, S. et al. Urinary p-cresol is elevated in young French children with autism spectrum disorder: a replication study. Biomarkers 19, 463–470 (2014).
Ibrügger, S. et al. Two randomized cross-over trials assessing the impact of dietary gluten or wholegrain on the gut microbiome and host metabolic health. J. Clin. Trials 4, 1000178 (2014).
Falony, G. et al. Population-level analysis of gut microbiome variation. Science 352, 560–564 (2016).
Arumugam, M. et al. Enterotypes of the human gut microbiome. Nature 473, 174–180 (2011).
Roager, H. M., Licht, T. R., Poulsen, S. K., Larsen, T. M. & Bahl, M. I. Microbial enterotypes, inferred by the prevotella-to-bacteroides ratio, remained stable during a 6-month randomized controlled diet intervention with the new nordic diet. Appl. Environ. Microbiol. 80, 1142–1149 (2014).
Claus, S. P. et al. Systemic multicompartmental effects of the gut microbiome on mouse metabolic phenotypes. Mol. Syst. Biol. 4, 219 (2008).
He, X. & Slupsky, C. M. Metabolic fingerprint of dimethyl sulfone (DMSO2) in microbial-mammalian co-metabolism. J. Proteome Res. 13, 5281–5292 (2014).
Kilkkinen, A. et al. Use of oral antimicrobials decreases serum enterolactone concentration. Am. J. Epidemiol. 155, 472–477 (2002).
Topp, H., Sander, G., Heller-Schöch, G. & Schöch, G. Determination of 7-methylguanine, N2,N2-dimethylguanosine, and pseudouridine in ultrafiltrated serum of healthy adults by high-performance liquid chromatography. Anal. Biochem. 161, 49–56 (1987).
Topp, H. & Schöch, G. Whole-body degradation rates of transfer-, ribosomal-, and messenger ribonucleic acids and resting metabolic rate in 3- to 18-year-old humans. Pediatr. Res. 47, 163–163 (2000).
Mirvish, S. S., Medalie, J., Linsell, C. A., Yousuf, E. & Reyad, S. 7-methylguanine and other minor urinary purines: values for normal subjects from Israel, Gaza, and Kenya, and for patients with cancer of various organs or cirrhosis of the liver. Cancer 27, 736–743 (1971).
Johansson, M. E. V., Larsson, J. M. H. & Hansson, G. C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host–microbial interactions. Proc. Natl Acad. Sci. USA 108(Suppl), 4659–4665 (2011).
Barrios, C. et al. Gut-microbiota-metabolite axis in early renal function decline. PLoS ONE 10, e0134311 (2015).
Pimentel, M. et al. Methane, a gas produced by enteric bacteria, slows intestinal transit and augments small intestinal contractile activity. Am. J. Physiol. Gastrointest. Liver Physiol. 290, G1089–G1095 (2006).
Soret, R. et al. Short-chain fatty acids regulate the enteric neurons and control gastrointestinal motility in rats. Gastroenterology 138, 1772–1782 (2010).
Degen, L. P. & Phillips, S. F. How well does stool form reflect colonic transit? Gut 39, 109–113 (1996).
Macfarlane, G. T., Cummings, J. H., Macfarlane, S. & Gibson, G. R. Influence of retention time on degradation of pancreatic enzymes by human colonic bacteria grown in a 3-stage continuous culture system. J. Appl. Bacteriol. 67, 520–527 (1989).
Macfarlane, S., Quigley, M., Hopkins, M., Newton, D. F. & Macfarlane, G. Polysaccharide degradation by human intestinal bacteria during growth under multi-substrate limiting conditions in a three-stage continuous culture system. FEMS Microbiol. Ecol. 26, 231–243 (1998).
Cummings, J. H., Hill, M. J., Bone, E. S., Branch, W. J. & Jenkins, D. J. The effect of meat protein and dietary fiber on colonic function and metabolism. II: Bacterial metabolites in feces and urine. Am. J. Clin. Nutr. 32, 2094–2101 (1979).
Benus, R. F. J. et al. Association between Faecalibacterium prausnitzii and dietary fibre in colonic fermentation in healthy human subjects. Br. J. Nutr. 104, 693–700 (2010).
Manach, C., Scalbert, A., Morand, C., Remesy, C. & Jimenez, L. Polyphenols: food sources and bioavailability. Am. J. Clin. Nutr. 79, 727–747 (2004).
van Duynhoven, J. et al. Metabolic fate of polyphenols in the human superorganism. Proc. Natl Acad. Sci. USA 108(Suppl), 4531–4538 (2011).
Gross, G. et al. In vitro bioconversion of polyphenols from black tea and red wine/grape juice by human intestinal microbiota displays strong interindividual variability. J. Agric. Food Chem. 58, 10236–10246 (2010).
Bode, L. M. et al. In vivo and in vitro metabolism of trans-resveratrol by human gut microbiota. Am. J. Clin. Nutr. 97, 295–309 (2013).
Shimotoyodome, A., Meguro, S., Hase, T., Tokimitsu, I. & Sakata, T. Decreased colonic mucus in rats with loperamide-induced constipation. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 126, 203–212 (2000).
Toden, S., Bird, A. R., Topping, D. L. & Conlon, M. A. Resistant starch attenuates colonic DNA damage induced by higher dietary protein in rats. Nutr. Cancer 51, 45–51 (2005).
Ten Bruggencate, S. J. M., Bovee-Oudenhoven, I. M. J., Lettink-Wissink, M. L. G., Katan, M. B. & Van Der Meer, R. Dietary fructo-oligosaccharides and inulin decrease resistance of rats to salmonella: protective role of calcium. Gut 53, 530–535 (2004).
Rao, J. N. & Wang, J.-Y. in Molecule to Function to Disease (eds Granger, N., Granger, J. & Princeton, N. ) 11–114 (Morgan & Claypool, 2011).
Sakata, T. Effects of indigestible dietary bulk and short chain fatty acids on the tissue weight and epithelial cell proliferation rate of the digestive tract in rats. J. Nutr. Sci. Vitaminol. (Tokyo) 32, 355–362 (1986).
Goodlad, R. A. et al. Effects of an elemental diet, inert bulk and different types of dietary fibre on the response of the intestinal epithelium to refeeding in the rat and relationship to plasma gastrin, enteroglucagon, and PYY concentrations. Gut 28, 171–180 (1987).
Lewis, S. J. & Heaton, K. W. Increasing butyrate concentration in the distal colon by accelerating intestinal transit. Gut 41, 245–251 (1997).
Timmons, J., Chang, E. T., Wang, J.-Y. & Rao, J. N. Polyamines and gut mucosal homeostasis. J. Gastrointest. Dig. Syst. 2(Suppl 7), 001 (2012).
Earle, K. A. et al. Quantitative imaging of gut microbiota spatial organization. Cell Host Microbe 18, 478–488 (2015).
Guérin, A. et al. Risk of developing colorectal cancer and benign colorectal neoplasm in patients with chronic constipation. Aliment. Pharmacol. Ther. 40, 83–92 (2014).
Schadt, S., Chen, L.-Z. & Bischoff, D. Evaluation of relative LC/MS response of metabolites to parent drug in LC/nanospray ionization mass spectrometry: potential implications in MIST assessment. J. Mass Spectrom. 46, 1281–1286 (2011).
McKeown, C., Hisle-Gorman, E., Eide, M., Gorman, G. H. & Nylund, C. M. Association of constipation and fecal incontinence with attention-deficit/hyperactivity disorder. Pediatrics 132, e1210-5 (2013).
Pang, K. H. & Croaker, G. D. H. Constipation in children with autism and autistic spectrum disorder. Pediatr. Surg. Int. 27, 353–358 (2011).
Wu, M.-J. et al. Colonic transit time in long-term dialysis patients. Am. J. Kidney Dis. 44, 322–327 (2004).
Waller, P. A. et al. Dose–response effect of Bifidobacterium lactis HN019 on whole gut transit time and functional gastrointestinal symptoms in adults. Scand. J. Gastroenterol. 46, 1057–1064 (2011).
Abrahamsson, H. & Antov, S. Accuracy in assessment of colonic transit time with particles: how many markers should be used? Neurogastroenterol. Motil. 22, 1164–1169 (2010).
Wesolowska-Andersen, A. et al. Choice of bacterial DNA extraction method from fecal material influences community structure as evaluated by metagenomic analysis. Microbiome 2, 19 (2014).
Godon, J. J., Zumstein, E., Dabert, P., Habouzit, F. & Moletta, R. Molecular microbial diversity of an anaerobic digestor as determined by small-subunit rDNA sequence analysis. Appl. Environ. Microbiol. 63, 2802–2813 (1997).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 41, e1 (2013).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nature Methods 10, 996–998 (2013).
Haas, B. J. et al. Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res. 21, 494–504 (2011).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nature Methods 7, 335–336 (2010).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461 (2010).
McDonald, D. et al. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J. 6, 610–618 (2012).
Caporaso, J. G. et al. PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics 26, 266–267 (2010).
Price, M. N., Dehal, P. S. & Arkin, A. P. FastTree: computing large minimum evolution trees with profiles instead of a distance matrix. Mol. Biol. Evol. 26, 1641–1650 (2009).
Chen, Y. et al. Combination of injection volume calibration by creatinine and MS signals’ normalization to overcome urine variability in LC-MS-based metabolomics studies. Anal. Chem. 85, 7659–7665 (2013).
Want, E. J. et al. Global metabolic profiling procedures for urine using UPLC-MS. Nature Protoc. 5, 1005–1018 (2010).
Smith, C. A., Want, E. J., O'Maille, G., Abagyan, R. & Siuzdak, G. XCMS: processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal. Chem. 78, 779–787 (2006).
Wishart, D. S. et al. HMDB 3.0—The human metabolome database in 2013. Nucleic Acids Res. 41, D801–D807 (2013).
Kim, S. ppcor: an R package for a fast calculation to semi-partial correlation coefficients. Commun. Stat. Appl. Methods 22, 665–674 (2015).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate—a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 57, 289–300 (1995).
Warnes, G. R. et al. gplots: various R programming tools for plotting data. R package version 2.16.0 (CRAN, 2015); http://cran.r-project.org/package=gplots
Oksanen, J. et al. vegan: community ecology package. R package version 2.3.1 (CRAN, 2015); https://cran.r-project.org/package=vegan
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Acknowledgements
The authors thank K.V. Vibefelt for helping out with DNA extraction and N. Bicen for performing the PCR and sequencing. The sequencing was carried out by the DTU in-house facility (DTU Multi-Assay Core, DMAC), Technical University of Denmark. This work was funded by the Danish Council for Strategic Research (grant no. 11-116163; Center for Gut, Grain and Greens), by the Technical University of Denmark and by the personal Danisco Award (to T.R.L.). The Novo Nordisk Foundation Center for Basic Metabolic Research is an independent research centre at the University of Copenhagen and is partly funded by an unrestricted donation from the Novo Nordisk Foundation.
Author information
Authors and Affiliations
Contributions
M.I.B., R.J.G., H.V., T.H., T.S.-P., O.P., L.L., M.K., R.G. and T.R.L. assembled the cohort and developed protocols and infrastructure to obtain the biological samples and clinical metadata. M.H.S. measured the colonic transit time. L.B.S.H., V.C. and M.D.D. prepared the faecal samples, extracted DNA and performed 16S rRNA gene sequencing. L.B.S.H. performed the 16S data analyses, with contributions from V.C. H.M.R. and H.L.F. prepared the urine samples, performed the urine metabolic profiling and identified the urinary metabolites. H.M.R. performed the statistical correlation analyses with contributions from L.B.S.H., D.R.P. and H.B.N. H.M.R., M.I.B., H.B.N., L.B.S.H., T.S.-P., R.G., M.K. and T.R.L. interpreted the data. H.M.R. and T.R.L. wrote the manuscript and all authors read, revised and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Tables 1-6, Supplementary Figures 1-14 and Supplementary References. (PDF 2035 kb)
Rights and permissions
About this article
Cite this article
Roager, H., Hansen, L., Bahl, M. et al. Colonic transit time is related to bacterial metabolism and mucosal turnover in the gut. Nat Microbiol 1, 16093 (2016). https://doi.org/10.1038/nmicrobiol.2016.93
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.93