Abstract
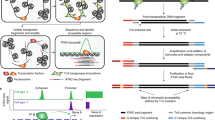
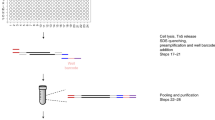
We describe an assay for transposase-accessible chromatin using sequencing (ATAC-seq), based on direct in vitro transposition of sequencing adaptors into native chromatin, as a rapid and sensitive method for integrative epigenomic analysis. ATAC-seq captures open chromatin sites using a simple two-step protocol with 500–50,000 cells and reveals the interplay between genomic locations of open chromatin, DNA-binding proteins, individual nucleosomes and chromatin compaction at nucleotide resolution. We discovered classes of DNA-binding factors that strictly avoided, could tolerate or tended to overlap with nucleosomes. Using ATAC-seq maps of human CD4+ T cells from a proband obtained on consecutive days, we demonstrated the feasibility of analyzing an individual's epigenome on a timescale compatible with clinical decision-making.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Kornberg, R.D. Chromatin structure: a repeating unit of histones and DNA. Science 184, 868–871 (1974).
Kornberg, R.D. & Lorch, Y. Chromatin structure and transcription. Annu. Rev. Cell Biol. 8, 563–587 (1992).
Mellor, J. The dynamics of chromatin remodeling at promoters. Mol. Cell 19, 147–157 (2005).
Boyle, A.P. et al. High-resolution mapping and characterization of open chromatin across the genome. Cell 132, 311–322 (2008).
Thurman, R.E. et al. The accessible chromatin landscape of the human genome. Nature 489, 75–82 (2012).
Schones, D.E. et al. Dynamic regulation of nucleosome positioning in the human genome. Cell 132, 887–898 (2008).
Valouev, A. et al. Determinants of nucleosome organization in primary human cells. Nature 474, 516–520 (2011).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Gerstein, M.B. et al. Architecture of the human regulatory network derived from ENCODE data. Nature 489, 91–100 (2012).
Goryshin, I.Y. & Reznikoff, W.S. Tn5 in vitro transposition. J. Biol. Chem. 273, 7367–7374 (1998).
Adey, A. et al. Rapid, low-input, low-bias construction of shotgun fragment libraries by high-density in vitro transposition. Genome Biol. 11, R119 (2010).
Gangadharan, S., Mularoni, L., Fain-Thornton, J., Wheelan, S.J. & Craig, N.L. DNA transposon Hermes inserts into DNA in nucleosome-free regions in vivo. Proc. Natl. Acad. Sci. USA 107, 21966–21972 (2010).
Song, L. & Crawford, G.E. DNase-seq: a high-resolution technique for mapping active gene regulatory elements across the genome from mammalian cells. Cold Spring Harb. Protoc. 2010 pdb.prot5384 (2010).
Simon, J.M., Giresi, P.G., Davis, I.J. & Lieb, J.D. Using formaldehyde-assisted isolation of regulatory elements (FAIRE) to isolate active regulatory DNA. Nat. Protoc. 7, 256–267 (2012).
The ENCODE Project Consortium. A user's guide to the Encyclopedia of DNA Elements (ENCODE). PLoS Biol. 9, e1001046 (2011).
Giresi, P.G. & Lieb, J.D. Isolation of active regulatory elements from eukaryotic chromatin using FAIRE (Formaldehyde Assisted Isolation of Regulatory Elements). Methods 48, 233–239 (2009).
Hoffman, M.M. et al. Integrative annotation of chromatin elements from ENCODE data. Nucleic Acids Res. 41, 827–841 (2013).
Prioleau, M.-N., Nony, P., Simpson, M. & Felsenfeld, G. An insulator element and condensed chromatin region separate the chicken β-globin locus from an independently regulated erythroid-specific folate receptor gene. EMBO J. 18, 4035–4048 (1999).
Ghirlando, R., Litt, M.D., Prioleau, M.-N., Recillas-Targa, F. & Felsenfeld, G. Physical properties of a genomic condensed chromatin fragment. J. Mol. Biol. 336, 597–605 (2004).
Kornberg, R.D. & Lorch, Y. Chromatin and transcription: where do we go from here. Curr. Opin. Genet. Dev. 12, 249–251 (2002).
Zhou, J., Fan, J.Y., Rangasamy, D. & Tremethick, D.J. The nucleosome surface regulates chromatin compaction and couples it with transcriptional repression. Nat. Struct. Mol. Biol. 14, 1070–1076 (2007).
Chen, K. et al. DANPOS: dynamic analysis of nucleosome position and occupancy by sequencing. Genome Res. 23, 341–351 (2013).
Kundaje, A. et al. Ubiquitous heterogeneity and asymmetry of the chromatin environment at regulatory elements. Genome Res. 22, 1735–1747 (2012).
Hesselberth, J.R. et al. Global mapping of protein-DNA interactions in vivo by digital genomic footprinting. Nat. Methods 6, 283–289 (2009).
Boyle, A.P. et al. High-resolution genome-wide in vivo footprinting of diverse transcription factors in human cells. Genome Res. 21, 456–464 (2011).
Neph, S. et al. An expansive human regulatory lexicon encoded in transcription factor footprints. Nature 489, 83–90 (2012).
Pique-Regi, R. et al. Accurate inference of transcription factor binding from DNA sequence and chromatin accessibility data. Genome Res. 21, 447–455 (2011).
Fraser, J.D., Irving, B.A., Crabtree, G.R. & Weiss, A. Regulation of interleukin-2 gene enhancer activity by the T cell accessory molecule CD28. Science 251, 313–316 (1991).
Flanagan, W.M., Corthésy, B., Bram, R.J. & Crabtree, G.R. Nuclear association of a T-cell transcription factor blocked by FK-506 and cyclosporin A. Nature 352, 803–807 (1991).
Lopez-Girona, A. et al. Lenalidomide downregulates the cell survival factor, interferon regulatory factor-4, providing a potential mechanistic link for predicting response. Br. J. Haematol. 154, 325–336 (2011).
Verstovsek, S. et al. Safety and efficacy of INCB018424, a JAK1 and JAK2 inhibitor, in myelofibrosis. N. Engl. J. Med. 363, 1117–1127 (2010).
Maurano, M.T. et al. Systematic localization of common disease-associated variation in regulatory. Science 337, 1190–1195 (2012).
Tang, F. et al. mRNA-Seq whole-transcriptome analysis of a single cell. Nat. Methods 6, 377–382 (2009).
Shalek, A.K. et al. Single-cell transcriptomics reveals bimodality in expression and splicing in immune cells. Nature 498, 236–240 (2013).
Jolma, A. et al. Multiplexed massively parallel SELEX for characterization of human transcription factor binding specificities. Genome Res. 20, 861–873 (2010).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10, R25 (2009).
Guo, Y., Mahony, S. & Gifford, D.K. High resolution genome wide binding event finding and motif discovery reveals transcription factor spatial binding constraints. PLoS Comput. Biol. 8, e1002638 (2012).
Eisen, M.B., Spellman, P.T., Brown, P.O. & Botstein, D. Cluster analysis and display of genome-wide expression patterns. Proc. Natl. Acad. Sci. USA 95, 14863–14868 (1998).
Acknowledgements
We thank members of Greenleaf and Chang labs for discussion, A. Burnet and S. Kim lab and the Stanford flow-cytometry core facility for assistance with FACS sorting, A. Schep for modeling Tn5 insertion preference, and V. Risca for graphics. This work was supported by the US National Institutes of Health (H.Y.C., W.J.G. and J.D.B.), including RC4NS073015, U01DK089532 and U19AI057229; Scleroderma Research Foundation (H.Y.C.); and California Institute for Regenerative Medicine (H.Y.C.). H.Y.C. acknowledges support as an Early Career Scientist of the Howard Hughes Medical Institute. GM12878 cells were a gift from the Snyder laboratory (Stanford University).
Author information
Authors and Affiliations
Contributions
J.D.B., P.G.G. and L.C.Z. performed the research. All authors designed experiments and interpreted the data. H.Y.C. and W.J.G. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
Stanford University has filed a provisional patent application on the methods described, and J.D.B., P.G.G., H.Y.C. and W.J.G. are named as inventors.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12 and Supplementary Tables 1 and 2 (PDF 3902 kb)
Rights and permissions
About this article
Cite this article
Buenrostro, J., Giresi, P., Zaba, L. et al. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat Methods 10, 1213–1218 (2013). https://doi.org/10.1038/nmeth.2688
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2688
This article is cited by
-
MYH1G-AS is a chromatin-associated lncRNA that regulates skeletal muscle development in chicken
Cellular & Molecular Biology Letters (2024)
-
PeNAC67-PeKAN2-PeSCL23 and B-class MADS-box transcription factors synergistically regulate the specialization process from petal to lip in Phalaenopsis equestris
Molecular Horticulture (2024)
-
Tissue-specific RNA Polymerase II promoter-proximal pause release and burst kinetics in a Drosophila embryonic patterning network
Genome Biology (2024)
-
Epigenetic reshaping through damage: promoting cell fate transition by BrdU and IdU incorporation
Cell & Bioscience (2024)
-
Epigenetic regulation and factors that influence the effect of iPSCs-derived neural stem/progenitor cells (NS/PCs) in the treatment of spinal cord injury
Clinical Epigenetics (2024)