Abstract
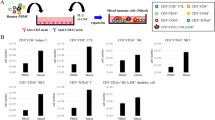
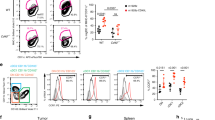
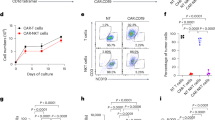
Natural killer T (NKT) lymphocytes mediate a rapid reaction to the glycolipid drug α-galactosylceramide (αGalCer), which triggers release of large amounts of cytokines into the serum within 12 h, starting with interleukin 4 (IL-4). When αGalCer is administered to mice on dendritic cells (DCs) instead, the response is more prolonged (>4 days) and marked by a large expansion in IFN-γ–producing NKT cells as well as greater resistance to metastases of the B16 melanoma. Nevertheless, DCs from mice given free αGalCer are able to induce strong IFN-γ–producing NKT responses when transferred to naïve mice, but not when transferred to αGalCer-treated recipients. In the latter, the NKT cells are anergized and can respond to glycolipid only in the presence of supplemental IL-2. Therefore, when αGalCer is selectively targeted to DCs, mice develop a stronger, more prolonged and effector type of NKT response, but this response can be blocked by the induction of anergy after presentation of αGalCer on other cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
26 August 2002
Acknowledgments were updated with note and PDF was appended with note.
References
Bendelac, A., Rivera, M.N., Park, S.-H. & Roark, J.H. Mouse CD1-specific NK1 T cells. Annu. Rev. Immunol. 15, 535–562 (1997).
MacDonald, H.R. CD1d-glycolipid Tetramers. A new tool to monitor natural killer T cells in health and disease. J. Exp. Med. 192, 15–20 (2000).
Godfrey, D.I., Hammond, K.J., Poulton, L.D., Smyth, M.J. & Baxter, A.G. NKT cells: facts, functions and fallacies. Immunol. Today 21, 573–583 (2000).
Hong, S. et al. The natural killer T-cell ligand α-galactosylceramide prevents autoimmune diabetes in non-obese diabetic mice. Nature Med. 7, 1052–1056 (2001).
Sharif, S. et al. Activation of natural killer T cells by α-galactosylceramide treatment prevents the onset and recurrence of autoimmune Type 1 diabetes. Nature Med. 7, 1057–1062 (2001).
Wang, B., Geng, Y.B. & Wang, C.R. CD1-restricted NKT cells protect nonobese diabetic mice from developing diabetes. J. Exp. Med. 194, 313–320 (2001).
Pal, E. et al. Costimulation-dependent modulation of experimental autoimmune encephalomyelitis by ligand stimulation of Vα14 NK T cells. J. Immunol. 166, 662–668 (2001).
Jahng, A.W. et al. Activation of natural killer T cells potentiates or prevents experimental autoimmune encephalomyelitis. J. Exp. Med. 194, 1789–1799 (2001).
Singh, A.K. et al. Natural killer T cell activation protects mice against experimental autoimmune encephalomyelitis. J. Exp. Med. 194, 1801–1811 (2001).
Kawano, T. et al. Natural killer-like nonspecific tumor cell lysis mediated by specific ligand-activated Vα14 NKT cells. Proc. Natl. Acad. Sci. USA 95, 5690–5693 (1998).
Cui, J. et al. Requirement for Vα14 NKT cells in IL-12-mediated rejection of tumors. Science 278, 1623–1626 (1997).
Toura, I. et al. Inhibition of experimental tumor metastasis by dendritic cells pulsed with α-galactosylceramide. J. Immunol. 163, 2387–2391 (1999).
Shin, T. et al. Inhibition of tumor metastasis by adoptive transfer of IL-12-activated Vα14 NKT cells. Int. J. Cancer 91, 523–528 (2001).
Nakagawa, R. et al. Mechanisms of the antimetastatic effect in the liver and of the hepatocyte injury induced by α-galactosylceramide in mice. J. Immunol. 166, 6578–6584 (2001).
Smyth, M.J. et al. Sequential production of interferon-γ by NK1. 1+ T cells and natural killer cells is essential for the antimetastatic effect of α- galactosylceramide. Blood 99, 1259–1266 (2002).
Smyth, M.J. et al. Differential tumor surveillance by natural killer (NK) and NKT cells. J. Exp. Med. 191, 661–668 (2000).
Smyth, M.J., Taniguchi, M. & Street, S.E. The anti-tumor activity of IL-12: mechanisms of innate immunity that are model and dose dependent. J. Immunol. 165, 2665–2670 (2000).
Takeda, K. et al. Relative contribution of NK and NKT cells to the anti-metastatic activities of IL-12. Int. Immunol. 12, 909–914 (2000).
Gonzalez-Aseguinolaza, G. et al. α-galactosylceramide-activated Vα14 natural killer T cells mediate protection against murine malaria. Proc. Natl. Acad. Sci. USA 97, 8461–8466 (2000).
Kakimi, K., Guidotti, L.G., Koezuka, Y. & Chisari, F.V. Natural killer T cell activation inhibits hepatitis B virus replication in vivo. J. Exp. Med. 192, 921–930 (2000).
Ishikawa, H. et al. CD4+ Vα14 NKT cells play a crucial role in an early stage of protective immunity against infection with Leishmania major. Int. Immunol. 12, 1267–1274 (2000).
Grant, E.P. et al. Molecular recognition of lipid antigens by T cell receptors. J. Exp. Med. 189, 195–205 (1999).
Kawano, T. et al. CD1d-restricted and TCR-mediated activation of Vα14 NKT cells by glycosylceramides. Science 278, 1626–1629 (1997).
Burdin, N. et al. Selective ability of mouse CD1 to present glycolipids: α-galactosylceramide specifically stimulates Vα14+ NK T lymphocytes. J. Immunol. 161, 3271–3281 (1998).
Brossay, L. et al. CD1d-mediated recognition of an α-galactosylceramide by natural killer T Cells is highly conserved through mammalian evolution. J. Exp. Med. 188, 1521–1528 (1998).
Spada, F.M., Koezuka, Y. & Porcelli, S.A. CD1d-restricted recognition of synthetic glycolipid antigens by human natural killer T cells. J. Exp. Med. 188, 1529–1534 (1998).
Matsuda, J.L. & Kronenberg, M. Presentation of self and microbial lipids by CD1 molecules. Curr. Opin. Immunol. 13, 19–25 (2001).
Morita, M. et al. Structure-activity relationship of α-galactosylceramides against B16-bearing mice. J. Med. Chem. 38, 2176 (1995).
Zeng, Z.-H. et al. Crystal structure of mouse CD1: an MHC- like fold with a large hydrophobic binding groove. Science 277, 339–345 (1997).
Benlagha, K., Weiss, A., Beavis, A., Teyton, L. & Bendelac, A. In vivo identification of glycolipid antigen-specific T cells using fluorescent CD1d tetramers. J. Exp. Med. 191, 1895–1904 (2000).
Matsuda, J.L. et al. Tracking the response of natural killer T cells to a glycolipid antigen using CD1d tetramers. J. Exp. Med. 192, 741–754 (2000).
Tomura, M. et al. A novel function of Vα14+ CD4+ NKT cells: stimulation of IL-12 production by antigen-presenting cells in the innate immune system. J. Immunol. 163, 93–101 (1999).
Kitamura, H. et al. The natural killer T (NKT) cell ligand α-galactosylceramide demonstrates its immunopotentiating effect by inducing interleukin (IL)-12 production by dendritic cells and IL-12 receptor expression on NKT cells. J. Exp. Med. 189, 1121–1128 (1999).
Yang, O.O. et al. CD1d on myeloid dendritic cells stimulates cytokine secretion from and cytolytic activity of Vα24JαQ T cells: a feedback mechanism for immune regulation. J. Immunol. 165, 3756–3762 (2000).
Trobonjaca, Z., Leithauser, F., Moller, P., Schirmbeck, R. & Reimann, J. Activating immunity in the liver. I. liver dendritic cells (but not hepatocytes) are potent activators of IFN-γ release by liver NKT cells. J. Immunol. 167, 1413–1422 (2001).
Kadowaki, N. et al. Distinct cytokine profiles of neonatal natural killer T cells after expansion with subsets of dendritic cells. J. Exp. Med. 193, 1221–1226 (2001).
Takahashi, T. et al. Analysis of human Vα24+ CD4+ NKT cells activated by α-glycosylceramide-pulsed monocyte-derived dendritic cells. J. Immunol. 164, 4458–4464 (2000).
Nishimura, T. et al. The interface between innate and acquired immunity: glycolipid antigen presentation by CD1d-expressing dendritic cells to NKT cells induces the differentiation of antigen-specific cytotoxic T lymphocytes. Int. Immunol. 12, 987–994 (2000).
Inaba, K., Metlay, J.P., Crowley, M.T. & Steinman, R.M. Dendritic cells pulsed with protein antigens in vitro can prime antigen-specific, MHC-restricted T cells in situ. J. Exp. Med. 172, 631–640 (1990).
Eberl, G. & MacDonald, H.R. Rapid death and regeneration of NKT cells in anti-CD3e or IL-12-treated mice: a major role for bone marrow in NKT cell homoestasis. Immunity 9, 345–353 (1998).
Osman, Y. et al. Activation of hepatic NKT cells and subsequent liver injury following administration of α-galactosylceramide. Eur. J. Immunol. 30, 1919–1928 (2000).
Hayakawa, Y. et al. Critical contribution of IFN-γ and NK cells, but not perforin- mediated cytotoxicity, to anti-metastatic effect of α-galactosylceramide. Eur. J. Immunol. 31, 1720–1727 (2001).
Muller, G. et al. Fetal calf serum-free generation of functionally active murine dendritic cells suitable for in vivo therapeutic approaches. J. Invest. Dermatol. 114, 142–149 (2000).
Inaba, K. et al. The formation of immunogenic MHC class II- peptide ligands in lysosomal compartments of dendritic cells is regulated by inflammatory stimuli. J. Exp. Med. 191, 927–936 (2000).
Granucci, F. et al. Inducible IL-2 production by dendritic cells revealed by global gene expression analysis. Nature Immunol. 2, 882–888 (2001).
Dunne, J. et al. Selective expansion and partial activation of human NK cells and NK receptor-positive T cells by IL-2 and IL-15. J. Immunol. 167, 3129–3138 (2001).
Wilson, S.B. & Byrne, M.C. Gene expression in NKT cells: defining a functionally distinct CD1d- restricted T cell subset. Curr. Opin. Immunol. 13, 555–561 (2001).
Nakui, M. et al. Potentiation of antitumor effect of NKT cell ligand, α- galactosylceramide by combination with IL-12 on lung metastasis of malignant melanoma cells. Clin. Exp. Metastasis 18, 147–153 (2000).
Carnaud, C. et al. Cutting edge: Cross-talk between cells of the innate immune system: NKT cells rapidly activate NK cells. J. Immunol. 163, 4647–4650 (1999).
Inaba, K. et al. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J. Exp. Med. 176, 1693–1702 (1992).
Acknowledgements
We thank M. Dhodapkar for critical reading of the manuscript. (*see note below)
*Note: In the AOP version of this article some text was incorrect. The acknowledgments should read: We thank S. Sidobre for the preparation of CD1d tetramers and M. Dhodapkar for critical reading of the manuscript. These errors have been corrected in the HTML version and will appear correctly in print. The PDF version available online has been appended.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R. M. S. has equity in a company that is developing the use of dendritic cells for clinical applications.
Rights and permissions
About this article
Cite this article
Fujii, Si., Shimizu, K., Kronenberg, M. et al. Prolonged IFN-γ–producing NKT response induced with α-galactosylceramide–loaded DCs. Nat Immunol 3, 867–874 (2002). https://doi.org/10.1038/ni827
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni827
This article is cited by
-
Regeneration of invariant natural killer T (iNKT) cells: application of iPSC technology for iNKT cell-targeted tumor immunotherapy
Inflammation and Regeneration (2023)
-
An immunostimulatory glycolipid that blocks SARS-CoV-2, RSV, and influenza infections in vivo
Nature Communications (2023)
-
Zeb2 regulates differentiation of long-lived effector of invariant natural killer T cells
Communications Biology (2023)
-
A randomised controlled trial of long NY-ESO-1 peptide-pulsed autologous dendritic cells with or without alpha-galactosylceramide in high-risk melanoma
Cancer Immunology, Immunotherapy (2023)
-
Infiltration of CD1a-positive dendritic cells in advanced laryngeal cancer correlates with unfavorable outcomes post-laryngectomy
BMC Cancer (2021)