Abstract
During metastatic progression, circulating cancer cells become lodged within the microvasculature of end organs, where most die from mechanical deformation. Although this phenomenon was first described over a half-century ago, the mechanisms enabling certain cells to survive this metastasis-suppressive barrier remain unknown. By applying whole-transcriptome RNA-sequencing technology to isogenic cancer cells of differing metastatic capacities, we identified a mutation encoding a truncated form of the pannexin-1 (PANX1) channel, PANX11–89, as recurrently enriched in highly metastatic breast cancer cells. PANX11–89 functions to permit metastatic cell survival during traumatic deformation in the microvasculature by augmenting ATP release from mechanosensitive PANX1 channels activated by membrane stretch. PANX1-mediated ATP release acts as an autocrine suppressor of deformation-induced apoptosis through P2Y-purinergic receptors. Finally, small-molecule therapeutic inhibition of PANX1 channels is found to reduce the efficiency of breast cancer metastasis. These data suggest a molecular basis for metastatic cell survival on microvasculature-induced biomechanical trauma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Zeidman, I. The fate of circulating tumors cells. I. Passage of cells through capillaries. Cancer Res. 21, 38–39 (1961).
Weiss, L., Nannmark, U., Johansson, B. R. & Bagge, U. Lethal deformation of cancer cells in the microcirculation: a potential rate regulator of hematogenous metastasis. Int. J. Cancer 50, 103–107 (1992).
Wong, C. W. et al. Apoptosis: an early event in metastatic inefficiency. Cancer Res. 61, 333–338 (2001).
Kienast, Y. et al. Real-time imaging reveals the single steps of brain metastasis formation. Nat. Med. 16, 116–122 (2010).
Weiss, L. Biomechanical interactions of cancer cells with the microvasculature during hematogenous metastasis. Cancer Metastasis Rev. 11, 227–235 (1992).
Weiss, L., Harlos, J. P. & Elkin, G. Mechanism of mechanical trauma to Ehrlich ascites tumor cells in vitro and its relationship to rapid intravascular death during metastasis. Int. J. Cancer 44, 143–148 (1989).
Chen, D. et al. LIFR is a breast cancer metastasis suppressor upstream of the Hippo-YAP pathway and a prognostic marker. Nat. Med. 18, 1511–1517 (2012).
DeRose, Y. S. et al. Tumor grafts derived from women with breast cancer authentically reflect tumor pathology, growth, metastasis and disease outcomes. Nat. Med. 17, 1514–1520 (2011).
Korpal, M. et al. Direct targeting of Sec23a by miR-200s influences cancer cell secretome and promotes metastatic colonization. Nat. Med. 17, 1101–1108 (2011).
Liu, C. et al. The microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Nat. Med. 17, 211–215 (2011).
Min, J. et al. An oncogene-tumor suppressor cascade drives metastatic prostate cancer by coordinately activating Ras and nuclear factor-κB. Nat. Med. 16, 286–294 (2010).
Peinado, H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18, 883–891 (2012).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Nik-Zainal, S. et al. The life history of 21 breast cancers. Cell 149, 994–1007 (2012).
Wang, Y. et al. Clonal evolution in breast cancer revealed by single nucleus genome sequencing. Nature 512, 155–160 (2014).
Minn, A. J. et al. Genes that mediate breast cancer metastasis to lung. Nature 436, 518–524 (2005).
Tavazoie, S. F. et al. Endogenous human microRNAs that suppress breast cancer metastasis. Nature 451, 147–152 (2008).
Bao, L., Locovei, S. & Dahl, G. Pannexin membrane channels are mechanosensitive conduits for ATP. FEBS Lett. 572, 65–68 (2004).
Locovei, S., Bao, L. & Dahl, G. Pannexin 1 in erythrocytes: function without a gap. Proc. Natl Acad. Sci. USA 103, 7655–7659 (2006).
Ransford, G. A. et al. Pannexin 1 contributes to ATP release in airway epithelia. Am. J. Respir. Cell Mol. Biol. 41, 525–534 (2009).
Seminario-Vidal, L. et al. Rho signaling regulates pannexin 1-mediated ATP release from airway epithelia. J. Biol. Chem. 286, 26277–26286 (2011).
Chekeni, F. B. et al. Pannexin 1 channels mediate ‘find-me’ signal release and membrane permeability during apoptosis. Nature 467, 863–867 (2010).
Sandilos, J. K. & Bayliss, D. A. Physiological mechanisms for the modulation of pannexin 1 channel activity. J. Physiol. 590, 6257–6266 (2012).
MacVicar, B. A. & Thompson, R. J. Non-junction functions of pannexin-1 channels. Trends Neurosci. 33, 93–102 (2010).
Thompson, R. J. et al. Activation of pannexin-1 hemichannels augments aberrant bursting in the hippocampus. Science 322, 1555–1559 (2008).
Thompson, R. J., Zhou, N. & MacVicar, B. A. Ischemia opens neuronal gap junction hemichannels. Science 312, 924–927 (2006).
Schenk, U. et al. Purinergic control of T cell activation by ATP released through pannexin-1 hemichannels. Sci. Signal. 1, ra6 (2008).
Corriden, R. & Insel, P. A. Basal release of ATP: an autocrine-paracrine mechanism for cell regulation. Sci. Signal. 3, re1 (2010).
Schumacher, D., Strilic, B., Sivaraj, K. K., Wettschureck, N. & Offermanns, S. Platelet-derived nucleotides promote tumor-cell transendothelial migration and metastasis via P2Y2 receptor. Cancer Cell 24, 130–137 (2013).
Gulbransen, B. D. et al. Activation of neuronal P2X7 receptor-pannexin-1 mediates death of enteric neurons during colitis. Nat. Med. 18, 600–604 (2012).
Silverman, W., Locovei, S. & Dahl, G. Probenecid, a gout remedy, inhibits pannexin 1 channels. Am. J. Physiol. Cell Physiol. 295, C761–C767 (2008).
Bruzzone, R., Barbe, M. T., Jakob, N. J. & Monyer, H. Pharmacological properties of homomeric and heteromeric pannexin hemichannels expressed in Xenopus oocytes. J. Neurochem. 92, 1033–1043 (2005).
Ma, W., Hui, H., Pelegrin, P. & Surprenant, A. Pharmacological characterization of pannexin-1 currents expressed in mammalian cells. J. Pharmacol. Exp. Ther. 328, 409–418 (2009).
Qian, B. et al. A distinct macrophage population mediates metastatic breast cancer cell extravasation, establishment and growth. PLoS ONE 4, e6562 (2009).
Gupta, G. P. et al. Mediators of vascular remodelling co-opted for sequential steps in lung metastasis. Nature 446, 765–770 (2007).
Li, A. et al. Mechanisms of ATP release, the enabling step in purinergic dynamics. Cell Physiol. Biochem. 28, 1135–1144 (2011).
Belete, H. A., Hubmayr, R. D., Wang, S. & Singh, R. D. The role of purinergic signaling on deformation induced injury and repair responses of alveolar epithelial cells. PLoS ONE 6, e27469 (2011).
Zhang, M., Piskuric, N. A., Vollmer, C. & Nurse, C. A. P2Y2 receptor activation opens pannexin-1 channels in rat carotid body type II cells: potential role in amplifying the neurotransmitter ATP. J. Physiol. 590, 4335–4350 (2012).
Ambrosi, C. et al. Pannexin1 and Pannexin2 channels show quaternary similarities to connexons and different oligomerization numbers from each other. J. Biol. Chem. 285, 24420–24431 (2010).
Sandilos, J. K. et al. Pannexin 1, an ATP release channel, is activated by caspase cleavage of its pore-associated C-terminal autoinhibitory region. J. Biol. Chem. 287, 11303–11311 (2012).
Hu, X. C. & Chow, L. W. Fine needle aspiration may shed breast cells into peripheral blood as determined by RT-PCR. Oncology 59, 217–222 (2000).
Murthy, S. M. et al. The influence of surgical trauma on experimental metastasis. Cancer 64, 2035–2044 (1989).
Demicheli, R., Retsky, M. W., Hrushesky, W. J., Baum, M. & Gukas, I. D. The effects of surgery on tumor growth: a century of investigations. Ann. Oncol. 19, 1821–1828 (2008).
Png, K. J., Halberg, N., Yoshida, M. & Tavazoie, S. F. A microRNA regulon that mediates endothelial recruitment and metastasis by cancer cells. Nature 481, 190–194 (2012).
Trapnell, C., Pachter, L. & Salzberg, S. L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009).
Chen, S. & Liu, J. S. Statistical applications of the Poisson-binomial and conditional Bernoulli distributions. Stat. Sin. 7, 875–892 (1997).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 57, 289–300 (1995).
Goya, R. et al. SNVMix: predicting single nucleotide variants from next-generation sequencing of tumors. Bioinformatics 26, 730–736 (2010).
Koboldt, D. C. et al. VarScan: variant detection in massively parallel sequencing of individual and pooled samples. Bioinformatics 25, 2283–2285 (2009).
Ding, L. et al. Genome remodelling in a basal-like breast cancer metastasis and xenograft. Nature 464, 999–1005 (2010).
Adzhubei, I. A. et al. A method and server for predicting damaging missense mutations. Nat. Methods 7, 248–249 (2010).
Pellegatti, P., Falzoni, S., Pinton, P., Rizzuto, R. & Di Virgilio, F. A novel recombinant plasma membrane-targeted luciferase reveals a new pathway for ATP secretion. Mol. Biol. Cell 16, 3659–3665 (2005).
Biserni, A. et al. Identification of apoptotic cells in reporter mice using modified luciferin. TOP (Transgenic Oper. Prod.) SRL (2010); http://www.promega.com/resources/pubhub/identification-of-apoptotic-cells-in-reporter-mice-using-modified-luciferin
Xu, J. Preparation, culture, and immortalization of mouse embryonic fibroblasts. Curr. Protoc. Mol. Biol. 28.1, 8 (2005).
Pencheva, N. et al. Convergent multi-miRNA targeting of ApoE drives LRP1/LRP8-dependent melanoma metastasis and angiogenesis. Cell 151, 1068–1082 (2012).
Acknowledgements
We thank C. Alarcon, N. Pencheva, A. Nguyen, J. Ross and Saeed Tavazoie for comments on previous versions of this manuscript. We thank C. D. Allis, B. Chait, T. Kapoor and V. Ruta for their insightful comments on this work. We thank C. Zhao of the Rockefeller Genomics Resource Center and J. Xiang of the Weill Cornell Medical College Genomics Resources Core Facility for assistance with next-generation RNA sequencing. We thank S. Chandarlapaty at MSKCC, USA, for providing the BT549, MDA-MB-468 and HCC1806 breast cancer cell lines. We thank V. Shestopalov at University of Miami, USA, for providing the PANX1-null mice. J. Massague at MSKCC, USA, generously provided metastatic sub-lines. P.W.F. is a biomedical fellow in the Weill Cornell/Rockefeller/Sloan-Kettering Tri-Institutional MD-PhD Program supported by NIH MSTP grant GM07739 and an Anderson Cancer Center Fellow at Rockefeller University. N.H. is an Anderson Cancer Center Fellow at Rockefeller University. S.F.T. is a Department of Defense (DOD) Era of Hope Scholar, a DOD Scholar-Innovator Award Recipient and a Rita Allen Foundation Scholar.
Author information
Authors and Affiliations
Contributions
S.F.T. conceived the project and supervised all research. P.W.F. and S.F.T. wrote the manuscript. P.W.F. and S.F.T. designed the experiments. P.W.F., S.Z., N.H. and C.M. performed the experiments. T.D.S., H.G., P.W.F. and O.E. designed and performed the computational analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
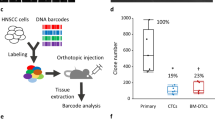
Supplementary Figure 3 The discovery of recurrent mutations enriched in highly metastatic breast cancer.
(a) Schematic of the systematic discovery framework used to identify nSNVs enriched by allelic frequency in highly metastatic CN-LM1A and MDA-LM2 human breast cancer cells. (b) Recurrent and non-neutral mutations identified to be significantly enriched in highly metastatic breast cancer cells by a one-tailed Student’s t-test (P < 0.05); n = 4. (c) Allele-specific RNA-seq of the PANX1 C268T allele in biological triplicates of the CN34 and MDA-MB-231 parental breast cancer cells and their respective lung metastatic derivatives, CN-LM1A and MDA-LM2. Mean enrichment was quantified by counting the increase in frequency of the PANX1 C268T allele in the metastatic sub-lines as compared to the corresponding parental lines; n = 3. (d) Sanger sequencing of the PANX1 mutant allele from genomic DNA of each parental and metastatic line. (e) Metaphase spread of four single-cell subclones generated from the MDA-LM2 cell population showing the intraclonal genetic heterogeneity (varying number of chromosomes) of in vivo selected highly metastatic derivative cell lines. Fractional enrichment of nSNVs in metastatic derivatives can be understood in the context of such heterogeneity. n represents biological replicates. Experiments b–d are representative and were replicated with two independent metastatic breast cancer cell lines.
Supplementary Figure 4 PANX11–89 augments PANX1 channel-mediated extracellular ATP release from metastatic breast cancer cells.
(a) The % inhibition of extracellular ATP release from metastatic derivative sub-lines at one minute was measured in the presence of three independent PANX1 inhibitors (Probenecid, Cbx and 10Panx1) at varying concentrations (2 mM, 500 μM and 100 μM, respectively). (b) Increasing concentrations of ATP (0, 50, 100, 500 nM) were measured using the Cell-Titer Glo luciferase assay. (c) Quantification of PANX1-mediated ATP release from the MDA-LM2 sub-line pretreated for 10 min with PBS, 2 mM probenecid (Prob), 500 μM CBX, or 100 μM 10Panx1 peptide; n = 4. (d) Extracellular ATP release from PANX1-null mouse embryonic fibroblasts (PANX1 KO MEFs) transfected with 5 μg human full-length PANX1 or 5 μg human full-length PANX1 and 2.5 μg human PANX11–89; n = 7. (e) Extracellular ATP release from PANX1 KO MEFs transfected with 5 μg human PANX11–89 or 5 μg vector control; n = 8. (f) PANX1-mediated extracellular ATP release from BT549 breast cancer cells expressing PANX11–89 or control vector; n = 8. (g) PANX1-mediated extracellular ATP release from MDA-MB-468 breast cancer cells expressing PANX11–89 or control vector; n = 8. (h) Quantitative RT-PCR analysis of wild-type PANX1 transcript expression in HCC1806 cells expressing PANX11–89 transfected with two independent siRNAs that specifically target full-length PANX1; n = 4. (i) CN34 and CN-LM1A total PANX1 mRNA expression quantified by RNA-seq; FPKM values averaged over two rounds (4 technical replicates) of RNA-seq for each cell line; n = 2. (j) Time-course measurements of ATP release from MDA-MB-231 parental cells and the MDA-LM2 metastatic derivatives sub-lines pretreated with Cbx (500 μM) or PBS for 10 min; n = 4. (k) Quantitative bioluminescence imaging of lung metastasis after the injection of 4 × 104 MDA-LM2 breast cancer cells pretreated with 100 μM 10Panx1 or scrambled peptide, into NOD scid (NS) mice; n = 5. (l) Day 42 quantification of metastatic foci from H&E-stained lungs (left) and representative lung images from vimentin-stained lungs (right) of mice injected with MDA-LM2 cells pretreated with 10Panx1 or scrambled peptide; n = 5. Scale bar, 1 mm. Error bars, s.e.m., ns, nonsignificant, ∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001 by a one-tailed Student’s t-test. n represents biological replicates c–h,l,j, number of mice in k and number of lungs in l. Experiments c–g and i–l were replicated twice with two independent cell lines. Experiments h and i are single experiments with biological replicates. Bioluminescent and histological images are representative of the median.
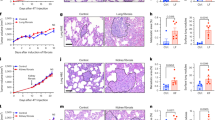
Supplementary Figure 5 PANX1 activity promotes the metastatic dissemination of breast cancer cells by enhancing early survival in the target organ.
(a) Daily quantitative imaging plot of lung bioluminescence subsequent to the injection of 4 × 104 metastatic MDA-LM2 breast cancer cells pretreated (30 min) with 100 μM 10Panx1 or scrambled peptides, into NS mice; n = 7. (b) Lungs from mice were extracted at day 3, sectioned and stained for vimentin and the numbers of vimentin-positive cancer cells were quantified; n = 7. Scale bar, 0.25 mm. (c) Quantification of proliferation over 5 days for CN-LM1A and MDA-LM2 cells over-expressing the autoinhibitory C-terminal domain of PANX1 or control vector; n = 4. (d) Quantification of 24 h cell survival for CN-LM1A and MDA-LM2 cells pretreated for 15 min with 100 μM 10Panx1 or scrambled peptide; n = 8. (e) Quantification of 24 h invasion for CN-LM1A and MDA-LM2 cells in the presence of 100 μM 10Panx1 or scrambled peptide; n = 4. (f) Quantification of 36 h anchorage-independent cancer cell survival for CN-LM1A cells in the presence of 100 μM 10Panx1 or scrambled peptide; n = 4. (g) Quantification of 24 h trans-endothelial migration for MDA-LM2 cells in the presence of 100 μM 10Panx1 or scrambled peptide; n = 4. (h) In vivo quantification of luciferase-based caspase-3/7 activity at 3 and 6 h after tail-vein injection of 4 × 104 MDA-LM2 breast cancer cells, pretreated with 100 μM 10Panx1 or scrambled peptide, into NS mice; n = 5. Error bars, s.e.m., ∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001 by a one-tailed Student’s t-test. n represents number of mice in a,h, number of mouse lungs in b, and biological replicates in c–g. Experiments a–e and h were replicated with at least two times in two independent cell lines. Experiments f and g are single experiments with biological replicates. Bioluminescent and histological images are representative of the median.
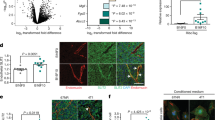
Supplementary Figure 6 PANX11–89 augments ATP release through an interaction with PANX1 to promote breast cancer cell survival in the pulmonary vasculature.
(a) Quantification of viable, trypan blue-negative CN-LM1A cells with 30 min Boyden chamber centrifugation (3,800 r.p.m.) after cells were pre-treated for 10 min with 500 μM CBX or PBS; n = 4. (b) Quantification of extracellular ATP release from CN-LM1A cells expressing the extracellular ATP hydrolase CD39 or control vector; n = 8. (c) Daily quantitative imaging of lung bioluminescence for three days subsequent to the injection of 1 × 105 CN-LM1A breast cancer cells expressing CD39 or control vector, into NS mice; n = 6. (d) Co-immunoprecipiation of Flag-tagged full-length PANX11−426 and endogenous PANX11–89 from CN-LM1A and MDA-LM2 cells. Anti-PANX1 N-terminal antibody detected a band similar in size to that of Flag-tagged PANX11–89 expressed in HEK293T cells. The presence of this band in the metastatic sub-lines suggests that endogenous PANX11–89 associates with Flag-tagged full-length PANX11−426. The multiple bands representing full-length PANX1 represent the previously described glycosylated forms of PANX1. (e) Co-immunoprecipitation of PANX11–89-Flag from HEK293T cells co-transfected with full-length PANX11−426. Anti-PANX1 N-terminal antibody was used to detect the associated PANX1 species. The multiple full-length PANX1 bands represent the previously described glycosylated forms of PANX1. The input lysates were immunoblotted for PANX1. (f) Increasing concentrations of DSP (dithiobis[succinimidyl propionate]) crosslinker were applied to HEK293T cells expressing Flag-tagged PANX1 before lysis. PANX1 complexes were detected using anti-FLAG M2 antibody. (g) Anti-PANX1 and anti-RFP immunoblotting of DSP crosslinked lysates from HEK293T cells expressing PANX1-EGFP, PANX1-EGFP and PANX11–89-mRFP or PANX11–89-mRFP. Molecular weights are indicated. (h) Quantification of PANX1-mediated ATP release from HEK293T cells transfected with 5 μg control vector, 5 μg wild-type PANX1 (DVVD), 2.5 μg wild-type PANX1 and 2.5 μg PANX11–89, 5 μg caspase resistant full-length PANX1 (AVVA), or 2.5 μg caspase resistant full-length PANX1 and 2.5 μg PANX11–89; n = 4. Error bars, s.e.m., ∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001 by a one-tailed Student’s t-test. n represents biological replicates in a,b,h, and number of mice in c. Experiments a–d were replicated at least two times with at least two independent cell lines. Experiments e–h are single experiments with biological replicates. Bioluminescent and images are representative of the median.
Supplementary Figure 7 PANX1 is a druggable target promoting breast cancer metastasis on cancer cell entry into the lung.
(a) Quantitative imaging of lung bioluminescence 18 h post tail-vein injection of 1 × 106 MDA-MB-468 breast cancer cells, expressing PANX11–89 or a control vector, into NSG mice; n = 6. (b) In vivo quantification of luciferase-based caspase-3/7 activity at 3 and 6 h post tail-vein injection of 1 × 106 MDA-MB-468 breast cancer cells, expressing either PANX11–89 or a control vector, into NS mice; n = 6. (c) Quantification of viable, trypan blue-negative MDA-MDA-468 cells expressing PANX11–89 or a control vector after 1 h extreme hypotonic (12.5% PBS) stretch in the presence of succinate buffer or apyrase (2 U ml−1); n = 4. (d) Quantitative bioluminescence imaging of lung metastasis after tail-vein injection of 1 × 105 CN-LM1A breast cancer cells pretreated for 30 min with CBX (500 μM) or PBS vehicle into NS mice; n = 4 (vehicle), n = 6 (Cbx). (e) Day 35 quantification of metastatic foci (left) and representative lung images (right) from H&E stained lungs of mice injected with CN-LM1A cells pretreated with CBX or PBS vehicle; n = 4 (vehicle), n = 6 (Cbx). Scale bar, 0.5 mm. (f) Mouse body weight before and after daily i.p. injections of CBX (25 mg kg−1) or an equivalent volume of PBS vehicle for seven days. n = 10. (g) 4-week representative images of vimentin-stained lungs from mice treated with two 25 mg kg−1 doses of intravenous CBX or vehicle control. Arrowheads indicate vimentin-positive metastatic foci. Scale bar, 0.5 mm. Error bars, s.e.m., ns, nonsignificant; ∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001 by a one-tailed Student’s t-test. n represents number of mice in a,b,d,f, number of mouse lungs in e and biological replicates in c. Experiments a–c were replicated at least two times with at least two independent cell lines. Therapeutic PANX1 inhibition experiments were performed using differing protocols with two independent triple negative breast cancer cell lines. Bioluminescent and histological images are representative of the median.
Supplementary information
Supplementary Information
Supplementary Information (PDF 627 kb)
Rights and permissions
About this article
Cite this article
Furlow, P., Zhang, S., Soong, T. et al. Mechanosensitive pannexin-1 channels mediate microvascular metastatic cell survival. Nat Cell Biol 17, 943–952 (2015). https://doi.org/10.1038/ncb3194
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3194