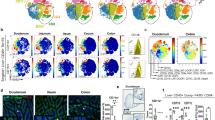
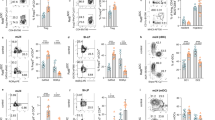
Abstract
The intestinal microbiota has a critical role in immune system and metabolic homeostasis, but it must be tolerated by the host to avoid inflammatory responses that can damage the epithelial barrier separating the host from the luminal contents1,2,3,4,5,6. Breakdown of this regulation and the resulting inappropriate immune response to commensals are thought to lead to the development of inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis7. We proposed that the intestinal immune system is instructed by the microbiota to limit responses to luminal antigens. Here we demonstrate in mice that, at steady state, the microbiota inhibits the transport of both commensal and pathogenic bacteria from the lumen to a key immune inductive site, the mesenteric lymph nodes (MLNs). However, in the absence of Myd88 or under conditions of antibiotic-induced dysbiosis, non-invasive bacteria were trafficked to the MLNs in a CCR7-dependent manner, and induced both T-cell responses and IgA production. Trafficking was carried out by CX3CR1hi mononuclear phagocytes, an intestinal-cell population previously reported to be non-migratory8. These findings define a central role for commensals in regulating the migration to the MLNs of CX3CR1hi mononuclear phagocytes endowed with the ability to capture luminal bacteria, thereby compartmentalizing the intestinal immune response to avoid inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Macpherson, A. J. & Uhr, T. Compartmentalization of the mucosal immune responses to commensal intestinal bacteria. Ann. NY Acad. Sci. 1029, 36–43 (2004)
Hooper, L. V. & Gordon, J. I. Commensal host-bacterial relationships in the gut. Science 292, 1115–1118 (2001)
Hooper, L. V., Midtvedt, T. & Gordon, J. I. How host-microbial interactions shape the nutrient environment of the mammalian intestine. Annu. Rev. Nutr. 22, 283–307 (2002)
Hooper, L. V. et al. Molecular analysis of commensal host-microbial relationships in the intestine. Science 291, 881–884 (2001)
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004)
Mazmanian, S. K., Liu, C. H., Tzianabos, A. O. & Kasper, D. L. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122, 107–118 (2005)
Balfour Sartor, R. Bacteria in Crohn’s disease: mechanisms of inflammation and therapeutic implications. J. Clin. Gastroenterol. 41 (suppl. 1). S37–S43 (2007)
Schulz, O. et al. Intestinal CD103+, but not CX3CR1+, antigen sampling cells migrate in lymph and serve classical dendritic cell functions. J. Exp. Med. 206, 3101–3114 (2009)
Macpherson, A. J. & Harris, N. L. Interactions between commensal intestinal bacteria and the immune system. Nature Rev. Immunol. 4, 478–485 (2004)
Vazquez-Torres, A. et al. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature 401, 804–808 (1999)
Griffin, A. J. & McSorley, S. J. Development of protective immunity to Salmonella, a mucosal pathogen with a systemic agenda. Mucosal Immunol. 4, 371–382 (2011)
Hapfelmeier, S. et al. Microbe sampling by mucosal dendritic cells is a discrete, MyD88-independent step in ΔinvG S. Typhimurium colitis. J. Exp. Med. 205, 437–450 (2008)
Mowat, A. M. Anatomical basis of tolerance and immunity to intestinal antigens. Nature Rev. Immunol. 3, 331–341 (2003)
Macpherson, A. J. & Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665 (2004)
Bogunovic, M. et al. Origin of the lamina propria dendritic cell network. Immunity 31, 513–525 (2009)
Varol, C. et al. Intestinal lamina propria dendritic cell subsets have different origin and functions. Immunity 31, 502–512 (2009)
Jung, S. et al. In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002)
Vallon-Eberhard, A., Landsman, L., Yogev, N., Verrier, B. & Jung, S. Transepithelial pathogen uptake into the small intestinal lamina propria. J. Immunol. 176, 2465–2469 (2006)
Jang, M. H. et al. CCR7 is critically important for migration of dendritic cells in intestinal lamina propria to mesenteric lymph nodes. J. Immunol. 176, 803–810 (2006)
Worbs, T. et al. Oral tolerance originates in the intestinal immune system and relies on antigen carriage by dendritic cells. J. Exp. Med. 203, 519–527 (2006)
Varol, C., Zigmond, E. & Jung, S. Securing the immune tightrope: mononuclear phagocytes in the intestinal lamina propria. Nature Rev. Immunol. 10, 415–426 (2010)
Rivollier, A., He, J., Kole, A., Valatas, V. & Kelsall, B. L. Inflammation switches the differentiation program of Ly6Chi monocytes from antiinflammatory macrophages to inflammatory dendritic cells in the colon. J. Exp. Med. 209, 139–155 (2012)
Johansson-Lindbom, B. et al. Functional specialization of gut CD103+ dendritic cells in the regulation of tissue-selective T cell homing. J. Exp. Med. 202, 1063–1073 (2005)
Rydstrom, A. & Wick, M. J. Monocyte recruitment, activation, and function in the gut-associated lymphoid tissue during oral Salmonella infection. J. Immunol. 178, 5789–5801 (2007)
Jung, S. et al. Analysis of fractalkine receptor CX3CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000)
Frank, D. N. et al. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl Acad. Sci. USA 104, 13780–13785 (2007)
Kaser, A., Zeissig, S. & Blumberg, R. S. Inflammatory bowel disease. Annu. Rev. Immunol. 28, 573–621 (2010)
Thiennimitr, P., Winter, S. E. & Baumler, A. J. Salmonella, the host and its microbiota. Curr. Opin. Microbiol. 15, 108–114 (2012)
Lupp, C. et al. Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe 2, 119–129 (2007)
Valdez, Y. et al. Nramp1 expression by dendritic cells modulates inflammatory responses during Salmonella Typhimurium infection. Cell. Microbiol. 10, 1646–1661 (2008)
Liu, F. & Whitton, J. L. Cutting edge: re-evaluating the in vivo cytokine responses of CD8+ T cells during primary and secondary viral infections. J. Immunol. 174, 5936–5940 (2005)
Vaishnava, S., Behrendt, C. L., Ismail, A. S., Eckmann, L. & Hooper, L. V. Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface. Proc. Natl Acad. Sci. USA 105, 20858–20863 (2008)
Napolitano, L. M., Koruda, M. J., Meyer, A. A. & Baker, C. C. The impact of femur fracture with associated soft tissue injury on immune function and intestinal permeability. Shock 5, 202–207 (1996)
Acknowledgements
We thank H. Yue and J. Hall for comments on the manuscript and members of the Littman laboratory for their suggestions. We thank the NYU Histology Core, which is supported in part by grant 5P30CA016087-32 from the National Cancer Institute. Supported by the American Cancer Society and National Institutes of Health (NIH) T32 CA009161 (G.E.D.), NIH T32 DK083256-02 (R.S.L.), Human Frontier Science Program Long-Term Fellowship (B.B.), NIH R01AI085166 (S.R.S.) and the Howard Hughes Medical Institute (D.R.L.).
Author information
Authors and Affiliations
Contributions
G.E.D. designed and performed the experiments. G.E.D. and D.R.L planned experiments and wrote the manuscript with input from all co-authors. R.S.L., J.-X.Z., S.R.S., B.B., C.G. and A.C. helped plan and perform experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-9, Supplementary Table 1 and additional references. (PDF 715 kb)
Rights and permissions
About this article
Cite this article
Diehl, G., Longman, R., Zhang, JX. et al. Microbiota restricts trafficking of bacteria to mesenteric lymph nodes by CX3CR1hi cells. Nature 494, 116–120 (2013). https://doi.org/10.1038/nature11809
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11809