Abstract
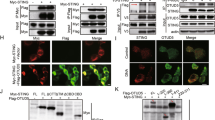
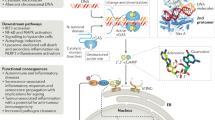
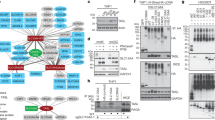
Innate immunity is stimulated not only by viral or bacterial components, but also by non-microbial danger signals (damage-associated molecular patterns)1. One of the damage-associated molecular patterns is chromosomal DNA that escapes degradation. In programmed cell death and erythropoiesis, DNA from dead cells or nuclei expelled from erythroblasts is digested by DNase II in the macrophages after they are engulfed. DNase II-/- (also known as Dnase2a-/- ) mice suffer from severe anaemia or chronic arthritis due to interferon-β (IFN-β) and tumour necrosis factor-α (TNF-α) produced from the macrophages carrying undigested DNA2,3 in a Toll-like receptor (TLR)-independent mechanism4. Here we show that Eyes absent 4 (EYA4), originally identified as a co-transcription factor, stimulates the expression of IFN-β and CXCL10 in response to the undigested DNA of apoptotic cells. EYA4 enhanced the innate immune response against viruses (Newcastle disease virus and vesicular stomatitis virus), and could associate with signalling molecules (IPS-1 (also known as MAVS), STING (TMEM173) and NLRX1). Three groups have previously shown that EYA has phosphatase activity5,6,7. We found that mouse EYA family members act as a phosphatase for both phosphotyrosine and phosphothreonine. The haloacid dehalogenase domain at the carboxy terminus contained the tyrosine-phosphatase, and the amino-terminal half carried the threonine-phosphatase. Mutations of the threonine-phosphatase, but not the tyrosine-phosphatase, abolished the ability of EYA4 to enhance the innate immune response, suggesting that EYA regulates the innate immune response by modulating the phosphorylation state of signal transducers for the intracellular pathogens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
23 July 2009
The figure key for Figure 2a was corrected on 23 July 2009.
References
Meylan, E., Tschopp, J. & Karin, M. Intracellular pattern recognition receptors in the host response. Nature 442, 39–44 (2006)
Yoshida, H., Okabe, Y., Kawane, K., Fukuyama, H. & Nagata, S. Lethal anemia caused by interferon-β produced in mouse embryos carrying undigested DNA. Nature Immunol. 6, 49–56 (2005)
Kawane, K. et al. Chronic polyarthritis caused by mammalian DNA that escapes from degradation in macrophages. Nature 443, 998–1002 (2006)
Okabe, Y., Kawane, K., Akira, S., Taniguchi, T. & Nagata, S. Toll-like receptor-independent gene induction program activated by mammalian DNA escaped from apoptotic DNA degradation. J. Exp. Med. 202, 1333–1339 (2005)
Li, X. et al. Eya protein phosphatase activity regulates Six1–Dach–Eya transcriptional effects in mammalian organogenesis. Nature 426, 247–254 (2003)
Rayapureddi, J. P. et al. Eyes absent represents a class of protein tyrosine phosphatases. Nature 426, 295–298 (2003)
Tootle, T. L. et al. The transcription factor Eyes absent is a protein tyrosine phosphatase. Nature 426, 299–302 (2003)
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006)
Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 449, 819–826 (2007)
Jemc, J. & Rebay, I. The eyes absent family of phosphotyrosine phosphatases: properties and roles in developmental regulation of transcription. Annu. Rev. Biochem. 76, 513–538 (2007)
Kim, T. K. & Maniatis, T. The mechanism of transcriptional synergy of an in vitro assembled interferon-β enhanceosome. Mol. Cell 1, 119–129 (1997)
Ohmori, Y. & Hamilton, T. A. Cooperative interaction between interferon (IFN) stimulus response element and κB sequence motifs controls IFNγ- and lipopolysaccharide-stimulated transcription from the murine IP-10 promoter. J. Biol. Chem. 268, 6677–6688 (1993)
Sharma, S. et al. Triggering the interferon antiviral response through an IKK-related pathway. Science 300, 1148–1151 (2003)
Iwamura, T. et al. Induction of IRF-3/-7 kinase and NF-κB in response to double-stranded RNA and virus infection: common and unique pathways. Genes Cells 6, 375–388 (2001)
Kawai, T. et al. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nature Immunol. 6, 981–988 (2005)
Meylan, E. et al. Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature 437, 1167–1172 (2005)
Ohto, H. et al. Cooperation of six and eya in activation of their target genes through nuclear translocation of Eya. Mol. Cell. Biol. 19, 6815–6824 (1999)
Ishikawa, H. & Barber, G. N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 455, 674–678 (2008)
Moore, C. B. et al. NLRX1 is a regulator of mitochondrial antiviral immunity. Nature 451, 573–577 (2008)
Tattoli, I. et al. NLRX1 is a mitochondrial NOD-like receptor that amplifies NF-κB and JNK pathways by inducing reactive oxygen species production. EMBO Rep. 9, 293–300 (2008)
Jin, L. et al. MPYS, a novel membrane tetraspanner, is associated with major histocompatibility complex class II and mediates transduction of apoptotic signals. Mol. Cell. Biol. 28, 5014–5026 (2008)
Cook, P. J. et al. Tyrosine dephosphorylation of H2AX modulates apoptosis and survival decisions. Nature 458, 591–596 (2009)
Das, A. K., Helps, N. R., Cohen, P. T. & Barford, D. Crystal structure of the protein serine/threonine phosphatase 2C at 2.0 Å resolution. EMBO J. 15, 6798–6809 (1996)
Lee, M. S. & Kim, Y. J. Signaling pathways downstream of pattern-recognition receptors and their cross talk. Annu. Rev. Biochem. 76, 447–480 (2007)
Michallet, M. C. et al. TRADD protein is an essential component of the RIG-like helicase antiviral pathway. Immunity 28, 651–661 (2008)
Abdelhak, S. et al. A human homologue of the Drosophila eyes absent gene underlies branchio-oto-renal (BOR) syndrome and identifies a novel gene family. Nature Genet. 15, 157–164 (1997)
Orten, D. J. et al. Branchio-oto-renal syndrome (BOR): novel mutations in the EYA1 gene, and a review of the mutational genetics of BOR. Hum. Mutat. 29, 537–544 (2008)
Mutsuddi, M. et al. Using Drosophila to decipher how mutations associated with human branchio-oto-renal syndrome and optical defects compromise the protein tyrosine phosphatase and transcriptional functions of eyes absent. Genetics 170, 687–695 (2005)
Borsani, G. et al. EYA4, a novel vertebrate gene related to Drosophila eyes absent. Hum. Mol. Genet. 8, 11–23 (1999)
Takeshita, S., Kaji, K. & Kudo, A. Identification and characterization of the new osteoclast progenitor with macrophage phenotypes being able to differentiate into mature osteoclasts. J. Bone Miner. Res. 15, 1477–1488 (2000)
Lam, K. M. & Hao, Q. Induction of lymphocyte agglutination and lysis by Newcastle disease virus. Vet. Microbiol. 15, 49–56 (1987)
Shiraishi, T. et al. Increased cytotoxicity of soluble Fas ligand by fusing isoleucine zipper motif. Biochem. Biophys. Res. Commun. 322, 197–202 (2004)
Hanayama, R. et al. Identification of a factor that links apoptotic cells to phagocytes. Nature 417, 182–187 (2002)
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000)
Ray, S. & Diamond, B. Generation of a fusion partner to sample the repertoire of splenic B cells destined for apoptosis. Proc. Natl Acad. Sci. USA 91, 5548–5551 (1994)
Kitamura, T. et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp. Hematol. 31, 1007–1014 (2003)
Morita, S., Kojima, T. & Kitamura, T. Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther. 7, 1063–1066 (2000)
Higuchi, R. in PCR Protocols: a Guide to Methods and Applications 177–188 (Academic, 1990)
Mizushima, S. & Nagata, S. pEF-BOS, a powerful mammalian expression vector. Nucleic Acids Res. 18, 5322 (1990)
Sawasaki, T., Ogasawara, T., Morishita, R. & Endo, Y. A cell-free protein synthesis system for high-throughput proteomics. Proc. Natl Acad. Sci. USA 99, 14652–14657 (2002)
Donella Deana, A. et al. An investigation of the substrate specificity of protein phosphatase 2C using synthetic peptide substrates; comparison with protein phosphatase 2A. Biochim. Biophys. Acta 1051, 199–202 (1990)
Umeda, I. O., Nakata, H. & Nishigori, H. Identification of protein phosphatase 2C and confirmation of other protein phosphatases in the ocular lenses. Exp. Eye Res. 79, 385–392 (2004)
Van Veldhoven, P. P. & Mannaerts, G. P. Inorganic and organic phosphate measurements in the nanomolar range. Anal. Biochem. 161, 45–48 (1987)
Yoneyama, M. et al. Direct triggering of the type I interferon system by virus infection: activation of a transcription factor complex containing IRF-3 and CBP/p300. EMBO J. 17, 1087–1095 (1998)
Fujisawa-Sehara, A., Hanaoka, K., Hayasaka, M., Hiromasa-Yagami, T. & Nabeshima, Y. Upstream region of the myogenin gene confers transcriptional activation in muscle cell lineages during mouse embryogenesis. Biochem. Biophys. Res. Commun. 191, 351–356 (1993)
Kawane, K. et al. Structure and promoter analysis of murine CAD and ICAD genes. Cell Death Differ. 6, 745–752 (1999)
Shapiro, D. J., Sharp, P. A., Wahli, W. W. & Keller, M. J. A high-efficiency HeLa cell nuclear transcription extract. DNA 7, 47–55 (1988)
Acknowledgements
We thank T. Fujita and M. Yoneyama for the NDV, A. Sehara-Fujisawa for the myogenin promoter, and M. Fujii and M. Harayama for secretarial assistance. This work was supported in part by Grants-in-Aid from the Ministry of Education, Science, Sports, and Culture in Japan. T.S. is a Research Assistant for Kyoto University Global COE program (Center for Frontier Medicine).
Author Contributions Y.O. screened the cDNA library, and identified EYA4 as a regulator of the innate immune reaction. T.S. produced recombinant EYA in 293T cells and wheat-germ extracts, biochemically characterized, and found its threonine-phosphatase activity. S.N. designed the research and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-14 with Legends and a Supplementary Reference. (PDF 3583 kb)
Rights and permissions
About this article
Cite this article
Okabe, Y., Sano, T. & Nagata, S. Regulation of the innate immune response by threonine-phosphatase of Eyes absent. Nature 460, 520–524 (2009). https://doi.org/10.1038/nature08138
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08138