Key Points
-
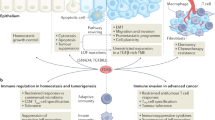
Signal transducer and activator of transcription (STAT) proteins have dual roles: they transduce signals through the cytoplasm and function as transcription factors in the nucleus. Although some STAT proteins such as STAT1 increase anti-tumour immunity, STAT3 and others induce cancer-promoting inflammation.
-
STAT3 signalling is a major intrinsic pathway for cancer inflammation owing to its frequent activation in malignant cells and key role in regulating many genes crucial for cancer inflammation in the tumour microenvironment.
-
Persistent activation of STAT3, and to a lesser extent STAT5, in diverse human cancers increases proliferation, survival, angiogenesis and metastasis, while also inhibiting anti-tumour immunity.
-
Many STAT3-regulated genes encode cytokines and growth factors, the receptors of which in turn activate the same STAT3 pathways, thereby propagating a stable feedforward loop between tumour cells and non-transformed stromal cells, including myeloid cells and T cells, promoting inflammatory responses that further support tumour growth and survival.
-
Interleukin-6 (IL-6)–Janus kinase (JAK)–STAT3 signalling is important for cancers resulting from the activation of the intrinsic inflammatory pathway owing to genetic or epigenetic changes in tumour cells. Extrinsic environmental inflammatory factors such as sunlight, pathogens and chemical carcinogens can also activate STAT3 through different mechanisms.
-
STAT3 interacts with nuclear factor-κB (NF-κB) at multiple levels and is activated by several NF-κB-regulated gene products, including IL-6. These two transcription factors regulate a multitude of genes important for STAT3 activation and cancer-promoting inflammation.
-
STAT1-driven anti-tumour immune responses and STAT3-mediated immune modulatory pathways can be mutually antagonistic, suggesting that therapeutic interventions targeting specific STATs can tip this balance to convert tumour-promoting inflammation to anti-tumour immune responses. Therefore, STAT3 has emerged as a crucial target for cancer therapy and STAT3 inhibitors are actively being developed.
-
Several tyrosine kinase inhibitors already in the clinic reduce STAT3 signalling by various mechanisms, thereby inducing tumour cell apoptosis and modulating inflammation in the tumour microenvironment in favour of therapeutic responses.
Abstract
Commensurate with their roles in regulating cytokine-dependent inflammation and immunity, signal transducer and activator of transcription (STAT) proteins are central in determining whether immune responses in the tumour microenvironment promote or inhibit cancer. Persistently activated STAT3 and, to some extent, STAT5 increase tumour cell proliferation, survival and invasion while suppressing anti-tumour immunity. The persistent activation of STAT3 also mediates tumour-promoting inflammation. STAT3 has this dual role in tumour inflammation and immunity by promoting pro-oncogenic inflammatory pathways, including nuclear factor-κB (NF-κB) and interleukin-6 (IL-6)–GP130–Janus kinase (JAK) pathways, and by opposing STAT1- and NF-κB-mediated T helper 1 anti-tumour immune responses. Consequently, STAT3 is a promising target to redirect inflammation for cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
Catlett-Falcone, R. et al. Constitutive activation of Stat3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity 10, 105–115 (1999). This report was the first to document that IL-6–JAK–STAT3 signalling, which increases inflammation-induced cancer, is important for the growth and survival of human tumour cells.
Bollrath, J. et al. gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell 15, 91–102 (2009).
Kujawski, M. et al. Stat3 mediates myeloid cell-dependent tumor angiogenesis in mice. J. Clin. Invest. 118, 3367–3377 (2008). This study demonstrated that STAT3 is a crucial mediator for myeloid cell-induced tumour angiogenesis.
Kortylewski, M. et al. Regulation of the IL-23 and IL-12 balance by Stat3 signaling in the tumor microenvironment. Cancer Cell 15, 114–123 (2009).
Wang, L., Yi, T., Kortylewski, M., Pardoll, D., Zeng, D., Yu, H. IL-17 can promote tumor growth through an IL-6/Stat3 signaling pathway. J. Exp. Med. 206, 1457–1464 (2009).
Grivennikov, S. et al. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell 15, 103–113 (2009). Studies reported in references 3 and 7 identified STAT3 as a central node for inflammation-induced cancer, suggesting that it is a crucial target for treating and preventing cancer-promoting inflammation.
Shain, K. H. et al. β1 integrin adhesion enhances IL-6-mediated STAT3 signaling in myeloma cells: implications for microenvironment influence on tumor survival and proliferation. Cancer Res. 69, 1009–1015 (2009).
Ara, T. et al. Interleukin-6 in the bone marrow microenvironment promotes the growth and survival of neuroblastoma cells. Cancer Res. 69, 329–337 (2009).
Gao, S. P. et al. Mutations in the EGFR kinase domain mediate STAT3 activation via IL-6 production in human lung adenocarcinomas. J. Clin. Invest. 117, 3846–3856 (2007).
Olcaydu, D. et al. A common JAK2 haplotype confers susceptibility to myeloproliferative neoplasms. Nature Genet. 41, 450–454 (2009).
Rebouissou, S. et al. Frequent in-frame somatic deletions activate gp130 in inflammatory hepatocellular tumours. Nature 457, 200–204 (2009). This study provides excellent examples of genetic mutations activating the IL-6–GP130–JAK–STAT3 pathway leading to inflammation-associated human cancer.
Ernst, M. et al. STAT3 and STAT1 mediate IL-11-dependent and inflammation-associated gastric tumorigenesis in gp130 receptor mutant mice. J. Clin. Invest. 118, 1727–1738 (2008).
Arredondo, J., Chernyavsky, A. I., Jolkovsky, D. L., Pinkerton, K. E. & Grando, S. A. Receptor-mediated tobacco toxicity: cooperation of the Ras/Raf-1/MEK1/ERK and JAK-2/STAT-3 pathways downstream of alpha7 nicotinic receptor in oral keratinocytes. FASEB J. 20, 2093–2101 (2006).
Gu, M., Singh, R. P., Dhanalakshmi, S., Agarwal, C. & Agarwal, R. Silibinin inhibits inflammatory and angiogenic attributes in photocarcinogenesis in SKH-1 hairless mice. Cancer Res. 67, 3483–3491 (2007).
Aziz, M. H., Manoharan, H. T. & Verma, A. K. Protein kinase C epsilon, which sensitizes skin to sun's UV radiation-induced cutaneous damage and development of squamous cell carcinomas, associates with Stat3. Cancer Res. 67, 1385–1394 (2007).
Bronte-Tinkew, D. M. et al. Helicobacter pylori cytotoxin-associated gene A activates the signal transducer and activator of transcription 3 pathway in vitro and in vivo. Cancer Res. 69, 632–639 (2009).
Landen, C. N. Jr, et al. Neuroendocrine modulation of signal transducer and activator of transcription-3 in ovarian cancer. Cancer Res. 67, 10389–10396 (2007).
Samavati, L. et al. STAT3 tyrosine phosphorylation is critical for interleukin 1β and interleukin-6 production in response to lipopolysaccharide and live bacteria. Mol. Immunol. 46, 1867–1877 (2009).
Chan, K. S. et al. Disruption of Stat3 reveals a critical role in both the initiation and the promotion stages of epithelial carcinogenesis. J. Clin. Invest. 114, 720–728 (2004).
Migone, T. S. et al. Constitutively activated Jak-STAT pathway in T cells transformed with HTLV-I. Science 269, 79–81 (1995).
Sano, S. et al. Signal transducer and activator of transcription 3 is a key regulator of keratinocyte survival and proliferation following UV irradiation. Cancer Res. 65, 5720–5729 (2005).
Karin, M. & Greten, F. R. NF-κB: linking inflammation and immunity to cancer development and progression. Nature Rev. Immunol. 5, 749–759 (2005).
Pikarsky, E. et al. NF-κB functions as a tumour promoter in inflammation-associated cancer. Nature 431, 461–466 (2004).
Baud, V. & Karin, M. Is NF-κB a good target for cancer therapy? Hopes and pitfalls. Nature Rev. Drug Discov. 8, 33–40 (2009).
Darnell, J. E. Jr. Transcription factors as targets for cancer therapy. Nature Rev. Cancer 2, 740–749 (2002).
Yu, H. & Jove, R. The STATs of cancer — new molecular targets come of age. Nature Rev. Cancer 4, 97–105 (2004).
Yu, H., Kortylewski, M. & Pardoll, D. Crosstalk between cancer and immune cells: role of STAT3 in the tumour microenvironment. Nature Rev. Immunol. 7, 41–51 (2007).
Zhong, Z., Wen, Z. & Darnell, J. E. Jr. Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science 264, 95–98 (1994).
Darnell, J. E. Jr, Kerr, I. M. & Stark, G. R. Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 264, 1415–1421 (1994).
Bromberg, J. F. et al. Stat3 as an oncogene. Cell 98, 295–303 (1999). This report was the first to demonstrate that Stat3 has intrinsic oncogenic potential.
Lee, H. et al. Persistently activated Stat3 maintains constitutive NF-κB activity in tumors. Cancer Cell 15, 283–293 (2009). This study revealed how STAT3 directly interacts with RELA and facilitates its acetylation by p300, leading to prolonged NF-κB nuclear retention in both tumour cells and tumour-associated immune cells.
Lu, T. & Stark, G. R. Cytokine overexpression and constitutive NFκB in cancer. Cell Cycle 3, 1114–1117 (2004).
Welte, T. et al. STAT3 deletion during hematopoiesis causes Crohn's disease-like pathogenesis and lethality: a critical role of STAT3 in innate immunity. Proc. Natl Acad. Sci. USA 100, 1879–1884 (2003).
Yang, J. et al. Unphosphorylated STAT3 accumulates in response to IL-6 and activates transcription by binding to NFκB. Genes Dev. 21, 1396–1408 (2007).
Basseres, D. S. & Baldwin, A. S. Nuclear factor-κB and inhibitor of κB kinase pathways in oncogenic initiation and progression. Oncogene 25, 6817–6830 (2006).
Haura, E. B., Turkson, J. & Jove, R. Mechanisms of disease: insights into the emerging role of signal transducers and activators of transcription in cancer. Nature. Clin. Pract. Oncol. 2, 315–324 (2005).
Heinrich, P. C., Behrmann, I., Muller-Newen, G., Schaper, F. & Graeve, L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem. J. 334 (Pt 2), 297–314 (1998).
Ogura, H. et al. Interleukin-17 promotes autoimmunity by triggering a positive-feedback loop via interleukin-6 induction. Immunity 29, 628–636 (2008).
Akira, S. et al. Molecular cloning of APRF, a novel IFN-stimulated gene factor 3 p91-related transcription factor involved in the gp130-mediated signaling pathway. Cell 77, 63–71 (1994).
Dunn, G. P. et al. A critical function for type I interferons in cancer immunoediting. Nature Immunol. 6, 722–729 (2005).
Blattman, J. N. & Greenberg, P. D. Cancer immunotherapy: a treatment for the masses. Science 305, 200–205 (2004).
Pardoll, D. M. Spinning molecular immunology into successful immunotherapy. Nature Rev. Immunol. 2, 227–238 (2002).
Kaplan, D. H. et al. Demonstration of an interferon γ-dependent tumor surveillance system in immunocompetent mice. Proc. Natl Acad. Sci. USA 95, 7556–7561 (1998).
Koebel, C. M. et al. Adaptive immunity maintains occult cancer in an equilibrium state. Nature 450, 903–907 (2007). This paper demonstrates the important role of adaptive immunity in controlling tumour outgrowth.
Galon, J. et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960–1964 (2006).
Dunn, G. P., Koebel, C. M. & Schreiber, R. D. Interferons, immunity and cancer immunoediting. Nature Rev. Immunol. 6, 836–848 (2006).
Sasaki, K. et al. Stat6 signaling suppresses VLA-4 expression by CD8+ T cells and limits their ability to infiltrate tumor lesions in vivo. J. Immunol. 181, 104–108 (2008).
Ostrand-Rosenberg, S. & Sinha, P. Myeloid-derived suppressor cells: linking inflammation and cancer. J. Immunol. 182, 4499–4506 (2009).
Kortylewski, M. et al. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nature Med. 11, 1314–1321 (2005). This was the first report to show that STAT3 is persistently activated in tumour-associated immune cells, leading to the suppression of anti-tumour innate and adaptive immune responses.
Cheng, F. et al. A critical role for Stat3 signaling in immune tolerance. Immunity 19, 425–436 (2003).
Cheng, P. et al. Inhibition of dendritic cell differentiation and accumulation of myeloid-derived suppressor cells in cancer is regulated by S100A9 protein. J. Exp. Med. 205, 2235–49 (2008).
Ho, H. H. & Ivashkiv, L. B. Role of STAT3 in type I interferon responses. Negative regulation of STAT1-dependent inflammatory gene activation. J. Biol. Chem. 281, 14111–14118 (2006).
Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nature Rev. Immunol. 3, 133–146 (2003).
Matsumura, Y. et al. Selective expansion of foxp3-positive regulatory T cells and immunosuppression by suppressors of cytokine signaling 3-deficient dendritic cells. J. Immunol. 179, 2170–2179 (2007).
Chen, Z. et al. Selective regulatory function of Socs3 in the formation of IL-17-secreting T cells. Proc. Natl Acad. Sci. USA 103, 8137–8142 (2006).
Chen, Z. & O'Shea, J. J. Th17 cells: a new fate for differentiating helper T cells. Immunol. Res. 41, 87–102 (2008).
Wu, S. et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nature Med. 15, 1016–1022 (2009).
Schindler, C., Levy, D. E. & Decker, T. JAK-STAT signaling: from interferons to cytokines. J. Biol. Chem. 282, 20059–20063 (2007).
Takeda, K. & Akira, S. STAT family of transcription factors in cytokine-mediated biological responses. Cytokine Growth Factor Rev. 11, 199–207 (2000).
Stark, G. R., Kerr, I. M., Williams, B. R., Silverman, R. H. & Schreiber, R. D. How cells respond to interferons. Annu. Rev. Biochem. 67, 227–264 (1998).
Darnell, J. E., Jr. Studies of IFN-induced transcriptional activation uncover the Jak-Stat pathway. J. Interferon Cytokine Res. 18, 549–554 (1998).
Taga, T. & Kishimoto, T. Gp130 and the interleukin-6 family of cytokines. Annu. Rev. Immunol. 15, 797–819 (1997).
Hirano, T., Ishihara, K. & Hibi, M. Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene 19, 2548–2556 (2000).
Levy, D. E. & Darnell, J. E., Jr. Stats: transcriptional control and biological impact. Nature Rev. Mol. Cell Biol. 3, 651–662 (2002).
Heinrich, P. C. et al. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem. J. 374, 1–20 (2003).
Chin, Y. E. et al. Cell growth arrest and induction of cyclin-dependent kinase inhibitor p21 WAF1/CIP1 mediated by STAT1. Science 272, 719–722 (1996).
Shen, Y., Devgan, G., Darnell, J. E., Jr, & Bromberg, J. F. Constitutively activated Stat3 protects fibroblasts from serum withdrawal and UV-induced apoptosis and antagonizes the proapoptotic effects of activated Stat1. Proc. Natl Acad. Sci. USA 98, 1543–1548 (2001).
Leung, S., Qureshi, S. A., Kerr, I. M., Darnell, J. E., Jr & Stark, G. R. Role of STAT2 in the alpha interferon signaling pathway. Mol. Cell. Biol. 15, 1312–1317 (1995).
Li, Y. et al. Activation of the signal transducers and activators of the transcription 3 pathway in alveolar epithelial cells induces inflammation and adenocarcinomas in mouse lung. Cancer Res. 67, 8494–8503 (2007).
Judd, L. M. et al. STAT3 activation regulates growth, inflammation, and vascularization in a mouse model of gastric tumorigenesis. Gastroenterology 131, 1073–1085 (2006).
Ogata, H. et al. Deletion of the SOCS3 gene in liver parenchymal cells promotes hepatitis-induced hepatocarcinogenesis. Gastroenterology 131, 179–193 (2006).
Dauer, D. J. et al. Stat3 regulates genes common to both wound healing and cancer. Oncogene 24, 3397–3408 (2005).
Grandis, J. R. et al. Requirement of Stat3 but not Stat1 activation for epidermal growth factor receptor- mediated cell growth in vitro. J. Clin. Invest. 102, 1385–1392 (1998).
Trevino, J. G. et al. Src activation of Stat3 is an independent requirement from NF-κB activation for constitutive IL-8 expression in human pancreatic adenocarcinoma cells. Angiogenesis 9, 101–110 (2006).
Bowman, T. et al. Stat3-mediated Myc expression is required for Src transformation and PDGF-induced mitogenesis. Proc. Natl Acad. Sci. USA 98, 7319–7324 (2001).
Yu, C. L. et al. Enhanced DNA-binding activity of a Stat3-related protein in cells transformed by the Src oncoprotein. Science 269, 81–83 (1995). This was the first report of persistent activation of STAT3 signalling by a specific oncoprotein in transformed cells.
Lo, H. W. et al. Nuclear interaction of EGFR and STAT3 in the activation of the iNOS/NO pathway. Cancer Cell 7, 575–589 (2005).
Lin, T. S., Mahajan, S. & Frank, D. A. STAT signaling in the pathogenesis and treatment of leukemias. Oncogene 19, 2496–2504 (2000).
Battle, T. E. & Frank, D. A. The role of STATs in apoptosis. Curr. Mol. Med. 2, 381–392 (2002).
Bruns, H. A. & Kaplan, M. H. The role of constitutively active Stat6 in leukemia and lymphoma. Crit. Rev. Oncol. Hematol. 57, 245–253 (2006).
Skinnider, B. F., Kapp, U. & Mak, T. W. The role of interleukin 13 in classical Hodgkin lymphoma. Leuk. Lymphoma 43, 1203–1210 (2002).
Huang, M. et al. Inhibition of Bcr-Abl kinase activity by PD180970 blocks constitutive activation of Stat5 and growth of CML cells. Oncogene 21, 8804–8816 (2002).
Xie, T. X. et al. Activation of stat3 in human melanoma promotes brain metastasis. Cancer Res. 66, 3188–3196 (2006).
Sumimoto, H., Imabayashi, F., Iwata, T. & Kawakami, Y. The BRAF-MAPK signaling pathway is essential for cancer-immune evasion in human melanoma cells. J. Exp. Med. 203, 1651–1656 (2006).
Wang, T. et al. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nature Med. 10, 48–54 (2004).
Dalwadi, H. et al. Cyclooxygenase-2-dependent activation of signal transducer and activator of transcription 3 by interleukin-6 in non-small cell lung cancer. Clin. Cancer Res. 11, 7674–7682 (2005).
Russo, V., Martienssen, R. A, and Riggs, A. D. Epigenetic Mechanisms of Gene Regulation. 662 (Cold Spring Harbor Laboratory Press, USA, 1996).
Zhang, Q., Wang, H. Y., Liu, X. & Wasik, M. A. STAT5A is epigenetically silenced by the tyrosine kinase NPM1-ALK and acts as a tumor suppressor by reciprocally inhibiting NPM1-ALK expression. Nature Med. 13, 1341–1348 (2007).
Choudhari, S. R. et al. Deactivation of Akt and STAT3 signaling promotes apoptosis, inhibits proliferation, and enhances the sensitivity of hepatocellular carcinoma cells to an anticancer agent, Atiprimod. Mol. Cancer Ther. 6, 112–121 (2007).
Sun, S. & Steinberg, B. M. PTEN is a negative regulator of STAT3 activation in human papillomavirus-infected cells. J. Gen. Virol. 83, 1651–1658 (2002).
Muromoto, R. et al. Epstein-Barr virus-derived EBNA2 regulates STAT3 activation. Biochem. Biophys. Res. Commun. 378, 439–443 (2009).
Kortylewski, M. et al. Toll-like receptor 9 activation of signal transducer and activator of transcription 3 constrains its agonist-based immunotherapy. Cancer Res. 69, 2497–2505 (2009).
Chiron, D., Bekeredjian-Ding, I., Pellat-Deceunynck, C., Bataille, R. & Jego, G. Toll-like receptors: lessons to learn from normal and malignant human B cells. Blood 112, 2205–2213 (2008).
Greten, F. R. et al. IKKβ links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118, 285–296 (2004).
Groblewska, M. et al. Serum interleukin 6 (IL-6) and C-reactive protein (CRP) levels in colorectal adenoma and cancer patients. Clin. Chem. Lab. Med. 46, 1423–1428 (2008).
Naugler, W. E. et al. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science 317, 121–124 (2007).
Matsukawa, A. et al. Stat3 in resident macrophages as a repressor protein of inflammatory response. J. Immunol. 175, 3354–3359 (2005).
Pollard, J. W. Tumour-educated macrophages promote tumour progression and metastasis. Nature Rev. Cancer 4, 71–78 (2004).
Chang, H. C. et al. Impaired development of human Th1 cells in patients with deficient expression of STAT4. Blood 113, 5887–5890 (2009).
Takeda, K. et al. Enhanced Th1 activity and development of chronic enterocolitis in mice devoid of Stat3 in macrophages and neutrophils. Immunity 10, 39–49 (1999).
Kubo, M., Hanada, T. & Yoshimura, A. Suppressors of cytokine signaling and immunity. Nature Immunol. 4, 1169–1176 (2003).
Zou, W. Regulatory T cells, tumour immunity and immunotherapy. Nature Rev. Immunol. 6, 295–307 (2006).
Gerosa, F. et al. Differential regulation of interleukin 12 and interleukin 23 production in human dendritic cells. J. Exp. Med. 205, 1447–1461 (2008).
Langowski, J. L. et al. IL-23 promotes tumour incidence and growth. Nature 442, 461–465 (2006).
Numasaki, M. et al. Interleukin-17 promotes angiogenesis and tumor growth. Blood 101, 2620–2627 (2003).
Kryczek, I., Wei, S., Szeliga, W., Vatan, L. & Zou, W. Endogenous IL-17 contributes to reduced tumor growth and metastasis. Blood 114, 357–359 (2009).
Ghosh, S., May, M. J. & Kopp, E. B. NF-κB and Rel proteins: evolutionarily conserved mediators of immune responses. Annu. Rev. Immunol. 16, 225–260 (1998).
Karin, M., Cao, Y., Greten, F. R. & Li, Z. W. NF-κB in cancer: from innocent bystander to major culprit. Nature Rev. Cancer 2, 301–310 (2002).
Chen, L. F. & Greene, W. C. Shaping the nuclear action of NF-κB. Nature Rev. Mol. Cell Biol. 5, 392–401 (2004).
Wang, J. et al. Distinct roles of different NF-κB subunits in regulating inflammatory and T cell stimulatory gene expression in dendritic cells. J. Immunol. 178, 6777–6788 (2007).
Chiarle, R. et al. Stat3 is required for ALK-mediated lymphomagenesis and provides a possible therapeutic target. Nature Med. 11, 623–629 (2005).
Milner, J. D. et al. Impaired TH17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 452, 773–776 (2008). This paper demonstrates that STAT3 dominant-negative mutation in individuals with hyper-IgE syndrome is associated with impaired T H 17 cell differentiation.
Minegishi, Y. et al. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature 448, 1058–1062 (2007).
Al Khatib, S. et al. Defects along the TH17 differentiation pathway underlie genetically distinct forms of the hyper IgE syndrome. J. Allergy Clin. Immunol. 124, 342–348, (2009).
Sen, B., Saigal, B., Parikh, N., Gallick, G. & Johnson, F. M. Sustained Src inhibition results in signal transducer and activator of transcription 3 (STAT3) activation and cancer cell survival via altered Janus-activated kinase-STAT3 binding. Cancer Res. 69, 1958–1965 (2009).
Paez-Ribes, M. et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15, 220–231 (2009).
Turkson, J. et al. Novel peptidomimetic inhibitors of signal transducer and activator of transcription 3 dimerization and biological activity. Mol. Cancer Ther. 3, 261–269 (2004).
Mandal, P. K. et al. Conformationally constrained peptidomimetic inhibitors of signal transducer and activator of transcription. 3: Evaluation and molecular modeling. J. Med. Chem. 52, 2429–2442 (2009).
Turkson, J. et al. Phosphotyrosyl peptides block Stat3-mediated DNA binding activity, gene regulation, and cell transformation. J. Biol. Chem. 276, 45443–45455 (2001).
Siddiquee, K. et al. Selective chemical probe inhibitor of Stat3, identified through structure-based virtual screening, induces antitumor activity. Proc. Natl Acad. Sci. USA 104, 7391–7396 (2007).
Schust, J., Sperl, B., Hollis, A., Mayer, T. U. & Berg, T. Stattic: a small-molecule inhibitor of STAT3 activation and dimerization. Chem. Biol. 13, 1235–1242 (2006).
Turkson, J. et al. Inhibition of constitutive signal transducer and activator of transcription 3 activation by novel platinum complexes with potent antitumor activity. Mol. Cancer Ther. 3, 1533–1542 (2004).
Turkson, J. et al. A novel platinum compound inhibits constitutive Stat3 signaling and induces cell cycle arrest and apoptosis of malignant cells. J. Biol. Chem. 280, 32979–32988 (2005).
Ling, X. & Arlinghaus, R. B. Knockdown of STAT3 expression by RNA interference inhibits the induction of breast tumors in immunocompetent mice. Cancer Res. 65, 2532–2536 (2005).
Leong, P. L. et al. Targeted inhibition of Stat3 with a decoy oligonucleotide abrogates head and neck cancer cell growth. Proc. Natl Acad. Sci. USA 100, 4138–4143 (2003).
Barton, B. E., Karras, J. G., Murphy, T. F., Barton, A. & Huang, H. F. Signal transducer and activator of transcription 3 (STAT3) activation in prostate cancer: Direct STAT3 inhibition induces apoptosis in prostate cancer lines. Mol. Cancer Ther. 3, 11–20 (2004).
Kortylewski, M. et al. In vivo delivery of siRNA to immune cells by conjugation to a TLR9 agonist enhances antitumor immune responses. Nature Biotechnol. 27, 925–932 (2009).
Sen, M. et al. Lack of toxicity of a STAT3 decoy oligonucleotide. Cancer Chemother. Pharmacol. 63, 983–995 (2009).
Blaskovich, M. A. et al. Discovery of JSI-124 (cucurbitacin I), a selective Janus kinase/signal transducer and activator of transcription 3 signaling pathway inhibitor with potent antitumor activity against human and murine cancer cells in mice. Cancer Res. 63, 1270–1279 (2003).
Kotha, A. et al. Resveratrol inhibits Src and Stat3 signaling and induces the apoptosis of malignant cells containing activated Stat3 protein. Mol. Cancer Ther. 5, 621–629 (2006).
Nam, S. et al. Indirubin derivatives inhibit Stat3 signaling and induce apoptosis in human cancer cells. Proc. Natl Acad. Sci. USA 102, 5998–6003 (2005).
Ghoreschi, K., Laurence, A. & O'Shea, J. J. Selectivity and therapeutic inhibition of kinases: to be or not to be? Nature Immunol. 10, 356–360 (2009).
Pesu, M. et al. Therapeutic targeting of Janus kinases. Immunol. Rev. 223, 132–142 (2008).
Kong, L. Y. et al. A novel inhibitor of signal transducers and activators of transcription 3 activation is efficacious against established central nervous system melanoma and inhibits regulatory T cells. Clin. Cancer Res. 14, 5759–5768 (2008).
Nefedova, Y. et al. Regulation of dendritic cell differentiation and antitumor immune response in cancer by pharmacologic-selective inhibition of the janus-activated kinase 2/signal transducers and activators of transcription 3 pathway. Cancer Res. 65, 9525–9535 (2005).
Gozgit, J. M. et al. Effects of the JAK2 inhibitor, AZ960, on Pim/BAD/BCL-xL survival signaling in the human JAK2 V617F cell line SET-2. J. Biol. Chem. 283, 32334–32343 (2008).
Yang, F. et al. Sorafenib inhibits signal transducer and activator of transcription 3 signaling associated with growth arrest and apoptosis of medulloblastomas. Mol. Cancer Ther. 7, 3519–3526 (2008).
Xin, H. et al. Sunitinib inhibition of Stat3 induces renal cell carcinoma tumor cell apoptosis and reduces immunosuppressive cells. Cancer Res. 69, 2506–2513 (2009).
Ozao-Choy, J. et al. The novel role of tyrosine kinase inhibitor in the reversal of immune suppression and modulation of tumor microenvironment for immune-based cancer therapies. Cancer Res. 69, 2514–2522 (2009).
Ko, J. S. et al. Sunitinib mediates reversal of myeloid-derived suppressor cell accumulation in renal cell carcinoma patients. Clin. Cancer Res. 15, 2148–2157 (2009).
Caprioli, F. et al. Autocrine regulation of IL-21 production in human T lymphocytes. J. Immunol. 180, 1800–1807 (2008).
Donnelly, R. P., Dickensheets, H. & Finbloom, D. S. The interleukin-10 signal transduction pathway and regulation of gene expression in mononuclear phagocytes. J. Interferon Cytokine Res. 19, 563–573 (1999).
Lamprecht, B. et al. Aberrant expression of the Th2 cytokine IL-21 in Hodgkin lymphoma cells regulates STAT3 signaling and attracts Treg cells via regulation of MIP-3α. Blood 112, 3339–3347 (2008).
Takatori, H., Kanno, Y., Chen, Z. & O'Shea, J. J. New complexities in helper T cell fate determination and the implications for autoimmune diseases. Mod. Rheumatol. 18, 533–541 (2008).
Stout, B. A., Bates, M. E., Liu, L. Y., Farrington, N. N. & Bertics, P. J. IL-5 and granulocyte-macrophage colony-stimulating factor activate STAT3 and STAT5 and promote Pim-1 and cyclin D3 protein expression in human eosinophils. J. Immunol. 173, 6409–6417 (2004).
Zorn, E. et al. IL-2 regulates FOXP3 expression in human CD4+CD25+ regulatory T cells through a STAT-dependent mechanism and induces the expansion of these cells in vivo. Blood 108, 1571–1579 (2006).
Finkelman, F. D. et al. Interleukin-4- and interleukin-13-mediated host protection against intestinal nematode parasites. Immunol. Rev. 201, 139–155 (2004).
Schuringa, J. J., Timmer, H., Luttickhuizen, D., Vellenga, E. & Kruijer, W. c-Jun and c-Fos cooperate with STAT3 in IL-6-induced transactivation of the IL-6 respone element (IRE). Cytokine 14, 78–87 (2001).
Lo, H. W. et al. Epidermal growth factor receptor cooperates with signal transducer and activator of transcription 3 to induce epithelial-mesenchymal transition in cancer cells via up-regulation of TWIST gene expression. Cancer Res. 67, 9066–9076 (2007).
Wu, Y., Diab, I., Zhang, X., Izmailova, E. S. & Zehner, Z. E. Stat3 enhances vimentin gene expression by binding to the antisilencer element and interacting with the repressor protein, ZBP-89. Oncogene 23, 168–178 (2004).
Liu, H. et al. Serine phosphorylation of STAT3 is essential for Mcl-1 expression and macrophage survival. Blood 102, 344–352 (2003).
Madamanchi, N. R., Li, S., Patterson, C. & Runge, M. S. Thrombin regulates vascular smooth muscle cell growth and heat shock proteins via the JAK-STAT pathway. J. Biol. Chem. 276, 18915–18924 (2001).
Bartoli, M. et al. VEGF differentially activates STAT3 in microvascular endothelial cells. FASEB J. 17, 1562–1564 (2003).
Gao, H., Priebe, W., Glod, J. & Banerjee, D. Activation of signal transducers and activators of transcription 3 and focal adhesion kinase by stromal cell-derived factor 1 is required for migration of human mesenchymal stem cells in response to tumor cell-conditioned medium. Stem Cells 27, 857–865 (2009).
Leung, T. H., Hoffmann, A. & Baltimore, D. One nucleotide in a κB site can determine cofactor specificity for NF-κB dimers. Cell 118, 453–464 (2004).
Burysek, L., Syrovets, T. & Simmet, T. The serine protease plasmin triggers expression of MCP-1 and CD40 in human primary monocytes via activation of p38 MAPK and janus kinase (JAK)/STAT signaling pathways. J. Biol. Chem. 277, 33509–33517 (2002).
Acknowledgements
We would like to thank members of our laboratories who have made major discoveries in the topics discussed here. We also thank the many laboratories that have contributed important publications relevant to the topics in this Review but which could not be cited owing to space limitations. Special thanks go to L. Wang and J. Deng for helping to prepare the figures and references, R. Buettner for compiling data on STAT3 inhibitors, and M. Kortylewski for critical reading of the manuscript.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- TH1
-
A TH1 response is mediated by CD4+ T cells and promoted by type 1 interferons and IL-12, this response mediates its effect through cytokines, particularly IFNγ, promoting cellular immune responses against intracellular infections and tumours.
- Innate immune cells
-
Innate immune cells include natural killer cells, macrophages, neutrophils and dendritic cells, which provide immediate non-specific defence against pathogens. These cells identify and destroy virus-, bacteria- and fungus-infected cells and malignant cells.
- Myeloid-derived suppressor cells
-
MDSCs. Heterogenous and plastic cells. When isolated from normal bone marrow, they do not exhibit imunosuppresive effects. However, when exposed to the tumour microenvironment, they inhibit both CD4+ and CD8+ T cells.
- TH17 T cells
-
Defined by the secretion of IL-17A but not IFNγ or IL-4. TH17 cells have been implicated in protective immunity to intestinal and pulmonary bacterial infections, as well as pathological immunity in several autoimmune diseases.
- Type 1 IFNs
-
Include multiple family members of the IFNα group and the related IFNβ. All type 1 IFNs bind to a single receptor, termed the type 1 or IFNα receptor. They mediate direct anti-viral activity against infected cells, and can also inhibit tumour growth and promote anti-tumour immune responses.
- Natural killer cell
-
A type of lymphocyte that protects against infectious microbes and kills tumour cells through the recognition of specific cell membrane molecules that are upregulated under conditions of cell stress, such as infection or carcinogenic transformation.
- Adaptive immunity
-
Mediated by antigen-specific T lymphocytes and antibodies produced by B cells. It takes longer to develop than innate immunity but has greater antigen specificity and includes the development of immunological memory.
- Dendritic cells
-
Specialized and the most efficient antigen-presenting cells, which can activate T cells and thereby induce antigen-specific immune responses.
- CD8+ T cell
-
The T cell subset from which cytolytic T cells develop. These in turn directly recognize target cells based on the surface expression of antigenic peptide complexed with MHC I molecules and kill their targets by injecting granzymes that induce apoptosis.
- Lamina propria
-
A constituent of the moist linings of mucous membranes, which line different tubes of the body, including the gastrointestinal tract.
- Hyper-IgE syndrome
-
A rare immune and connective tissue disorder characterized by dermatitis, cyst-forming pneumonia and increased serum levels of immunoglobulin E antibody. In some patients it is caused by autosomal dominant STAT3 mutations.
- Decoy DNA-binding sites
-
DNA oligonucleotides with sequence-specific binding sites that sequester their cognate binding proteins, thereby preventing them from binding to the regulatory sequences of nuclear genes.
Rights and permissions
About this article
Cite this article
Yu, H., Pardoll, D. & Jove, R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 9, 798–809 (2009). https://doi.org/10.1038/nrc2734
Issue Date:
DOI: https://doi.org/10.1038/nrc2734
This article is cited by
-
Chronic restraint stress promotes oral squamous cell carcinoma development by inhibiting ALDH3A1 via stress response hormone
BMC Oral Health (2024)
-
Syntaxin6 contributes to hepatocellular carcinoma tumorigenesis via enhancing STAT3 phosphorylation
Cancer Cell International (2024)
-
Single-cell RNA sequencing analysis of vestibular schwannoma reveals functionally distinct macrophage subsets
British Journal of Cancer (2024)
-
Medicinal chemistry perspective of JAK inhibitors: synthesis, biological profile, selectivity, and structure activity relationship
Molecular Diversity (2024)
-
Synthesis and pharmacological evaluation of polyamine-diarylidenyl piperidone derivatives as potent antitumor agents
Monatshefte für Chemie - Chemical Monthly (2024)