Abstract
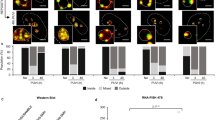
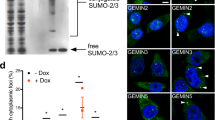
The survival motor neurons (smn) gene in mice is essential for embryonic viability. In humans, mutation of the telomeric copy of the SMN1 gene causes spinal muscular atrophy, an autosomal recessive disease. Here we report that the SMN protein interacts with the zinc-finger protein ZPR1 and that these proteins colocalize in small subnuclear structures, including gems and Cajal bodies. SMN and ZPR1 redistribute from the cytoplasm to the nucleus in response to serum. This process is disrupted in cells from patients with Werdnig–Hoffman syndrome (spinal muscular atrophy type I) that have SMN1 mutations. Similarly, decreased ZPR1 expression prevents SMN localization to nuclear bodies. Our data show that ZPR1 is required for the localization of SMN in nuclear bodies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Munsat, T. L. & Davies, K. E., eds International SMA consortium meeting, 26–28 June 1992, Bonn, Germany. Neuromuscul Disord. 2, 423–428 (1992).
Wirth, B. An update of the mutation spectrum of the survival motor neuron gene (SMN1) in autosomal recessive spinal muscular atrophy (SMA). Hum. Mutat. 15, 228–237 (2000).
Lefebvre, S. et al. Identification and characterization of a spinal muscular atrophy- determining gene. Cell 80, 155–165 (1995).
Schrank, B. et al. Inactivation of the survival motor neuron gene, a candidate gene for human spinal muscular atrophy, leads to massive cell death in early mouse embryos. Proc. Natl Acad. Sci. USA 94, 9920–9925 (1997).
Miguel-Aliaga, I. et al. The Caenorhabditis elegans orthologue of the human gene responsible for spinal muscular atrophy is a maternal product critical for germline maturation and embryonic viability. Hum. Mol. Genet. 8, 2133–2143 (1999).
Monani, U. R. et al. A single nucleotide difference that alters splicing patterns distinguishes the SMA gene SMN1 from the copy gene SMN2. Hum. Mol. Genet. 8, 1177–1183 (1999).
Lorson, C. L., Hahnen, E., Androphy, E. J. & Wirth, B. A single nucleotide in the SMN gene regulates splicing and is responsible for spinal muscular atrophy. Proc. Natl Acad. Sci. USA 96, 6307–6311 (1999).
Frugier, T. et al. Nuclear targeting defect of SMN lacking the C-terminus in a mouse model of spinal muscular atrophy. Hum. Mol. Genet. 9, 849–858 (2000).
Hsieh-Li, H. M. et al. A mouse model for spinal muscular atrophy. Nature Genet. 24, 66–70 (2000).
Monani, U. R. et al. The human centromeric survival motor neuron gene (SMN2) rescues embryonic lethality in Smn−/− mice and results in a mouse with spinal muscular atrophy. Hum. Mol. Genet. 9, 333–339 (2000).
Liu, Q. & Dreyfuss, G. A novel nuclear structure containing the survival of motor neurons protein. EMBO J. 15, 3555–3565 (1996).
Matera, A. G. & Frey, M. R. Coiled bodies and gems: Janus or gemini? Am. J. Hum. Genet. 63, 317–321 (1998).
Carvalho, T. et al. The spinal muscular atrophy disease gene product, SMN: A link between snRNP biogenesis and the Cajal (coiled) body. J. Cell Biol. 147, 715–728 (1999).
Young, P. J., Le, T. T., thi Man, N., Burghes, A. H. & Morris, G. E. The relationship between SMN, the spinal muscular atrophy protein, and nuclear coiled bodies in differentiated tissues and cultured cells. Exp. Cell Res. 256, 365–374 (2000).
Liu, Q., Fischer, U., Wang, F. & Dreyfuss, G. The spinal muscular atrophy disease gene product, SMN, and its associated protein SIP1, are in a complex with spliceosomal snRNP proteins. Cell 90, 1013–1021 (1997).
Fischer, U., Liu, Q. & Dreyfuss, G. The SMN–SIP1 complex has an essential role in spliceosomal snRNP biogenesis. Cell 90, 1023–1029 (1997).
Pellizzoni, L., Kataoka, N., Charroux, B. & Dreyfuss, G. A novel function for SMN, the spinal muscular atrophy disease gene product, in pre-mRNA splicing. Cell 95, 615–624 (1998).
Pagliardini, S. et al. Subcellular localization and axonal transport of the survival motor neuron (SMN) protein in the developing rat spinal cord. Hum. Mol. Genet. 9, 47–56 (2000).
Coovert, D. D. et al. The survival motor neuron protein in spinal muscular atrophy. Hum. Mol. Genet. 6, 1205–1214 (1997).
Lefebvre, S. et al. Correlation between severity and SMN protein level in spinal muscular atrophy. Nature Genet. 16, 265–269 (1997).
Galcheva-Gargova, Z. et al. Binding of zinc-finger protein ZPR1 to the epidermal growth factor receptor. Science 272, 1797–1802 (1996).
Galcheva-Gargova, Z. et al. The cytoplasmic zinc-finger protein ZPR1 accumulates in the nucleolus of proliferating cells. Mol. Biol. Cell 9, 2963–2971 (1998).
Gangwani, L., Mikrut, M., Galcheva-Gargova, Z. & Davis, R. J. Interaction of ZPR1 with translation elongation factor-1α in proliferating cells. J. Cell Biol. 143, 1471–1484 (1998).
Adams, M. D. et al. The genome sequence of Drosophila melanogaster. Science 287, 2185–2195 (2000).
The C. elegans sequencing consortium. Genome sequence of the nematode C. elegans: a platform for investigating biology. Science 282, 2012–2018 (1998).
Bertrandy, S. et al. The RNA-binding properties of SMN: deletion analysis of the zebrafish orthologue defines domains conserved in evolution. Hum. Mol. Genet. 8, 775–782 (1999).
Owen, N., Doe, C. L., Mellor, J. & Davies, K. E. Characterization of the Schizosaccharomyces pombe orthologue of the human survival motor neuron (SMN) protein. Hum. Mol. Genet. 9, 675–684 (2000).
Charroux, B. et al. Gemin3: A novel DEAD box protein that interacts with SMN, the spinal muscular atrophy gene product, and is a component of gems. J. Cell Biol. 147, 1181–1194 (1999).
Lorson, C. L. et al. SMN oligomerization defect correlates with spinal muscular atrophy severity. Nature Genet. 19, 63–66 (1998).
Charroux, B. et al. Gemin4. A novel component of the smn complex that is found in both gems and nucleoli. J. Cell Biol. 148, 1177–1186 (2000).
Iwahashi, H. et al. Synergistic anti-apoptotic activity between Bcl-2 and SMN implicated in spinal muscular atrophy. Nature 390, 413–417 (1997).
Strasswimmer, J. et al. Identification of survival motor neuron as a transcriptional activator- binding protein. Hum. Mol. Genet. 8, 1219–1226 (1999).
Williams, B. Y., Hamilton, S. L. & Sarkar, H. K. The survival motor neuron protein interacts with the transactivator FUSE binding protein from human fetal brain. FEBS Lett. 470, 207–210 (2000).
Giesemann, T. et al. A role for polyproline motifs in the spinal muscular atrophy protein SMN. Profilins bind to and colocalize with smn in nuclear gems. J. Biol. Chem. 274, 37908–37914 (1999).
Coovert, D. D. et al. Does the survival motor neuron protein (SMN) interact with Bcl-2? J. Med. Genet. 37, 536–539 (2000).
Meister, G. et al. Characterization of a nuclear 20S complex containing the survival of motor neurons (SMN) protein and a specific subset of spliceosomal Sm proteins. Hum. Mol. Genet. 9, 1977–1986 (2000).
Pellizzoni, L., Charroux, B. & Dreyfuss, G. SMN mutants of spinal muscular atrophy patients are defective in binding to snRNP proteins. Proc. Natl Acad. Sci. USA 96, 11167–11172 (1999).
Gall, J. G., Bellini, M., Wu, Z. & Murphy, C. Assembly of the nuclear transcription and processing machinery: Cajal bodies (coiled bodies) and transcriptosomes. Mol. Biol. Cell 10, 4385–4402 (1999).
Lewis, J. D. & Tollervey, D. Like attracts like: getting RNA processing together in the nucleus. Science 288, 1385–1389 (2000).
Sleeman, J. E. & Lamond, A. I. Nuclear organization of pre-mRNA splicing factors. Curr. Opin. Cell Biol. 11, 372–377 (1999).
Francis, J. W., Sandrock, A. W., Bhide, P. G., Vonsattel, J. P. & Brown, R. H. Jr Heterogeneity of subcellular localization and electrophoretic mobility of survival motor neuron (SMN) protein in mammalian neural cells and tissues. Proc. Natl Acad. Sci. USA 95, 6492–6497 (1998).
Harlow, E. & Lane, D. Antibodies: A Laboratory Manual, (Cold Spring Harbor Press, Cold Spring Harbor, 1988).
Andrade, L. E., Tan, E. M. & Chan, E. K. Immunocytochemical analysis of the coiled body in the cell cycle and during cell proliferation. Proc. Natl Acad. Sci. USA 90, 1947–1951 (1993).
Zillmann, M., Zapp, M. L. & Berget, S. M. Gel electrophoretic isolation of splicing complexes containing U1 small nuclear ribonucleoprotein particles. Mol. Cell. Biol. 8, 814–821 (1988).
Acknowledgements
We thank E. K. L. Chan and G. Dreyfuss for providing reagents, and K. Gemme for administrative assistance. This study was supported by a grant from the National Cancer Institute. R.J.D. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gangwani, L., Mikrut, M., Theroux, S. et al. Spinal muscular atrophy disrupts the interaction of ZPR1 with the SMN protein. Nat Cell Biol 3, 376–383 (2001). https://doi.org/10.1038/35070059
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35070059
This article is cited by
-
The phospho-landscape of the survival of motoneuron protein (SMN) protein: relevance for spinal muscular atrophy (SMA)
Cellular and Molecular Life Sciences (2022)
-
Revisiting the role of mitochondria in spinal muscular atrophy
Cellular and Molecular Life Sciences (2021)
-
Deregulation of ZPR1 causes respiratory failure in spinal muscular atrophy
Scientific Reports (2017)
-
Identification and Expression Pattern of a ZPR1 Gene in Wild Tomato (Solanum Pennellii)
Plant Molecular Biology Reporter (2013)
-
Spinal Muscular Atrophy and the Antiapoptotic Role of Survival of Motor Neuron (SMN) Protein
Molecular Neurobiology (2013)