Abstract
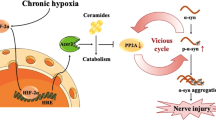
Hypoxia, especially the chronic type, leads to disruptive results in the brain that may contribute to the pathogenesis of some neurodegenerative diseases such as Alzheimer’s disease (AD). The ventrolateral medulla (VLM) contains clusters of interneurons, such as the pre-Bötzinger complex (preBötC), that generate the main respiratory rhythm drive. We hypothesized that exposing animals to chronic sustained hypoxia (CSH) might develop tauopathy in the brainstem, consequently changing the rhythmic manifestations of respiratory neurons. In this study, old (20–22 months) and young (2–3 months) male rats were subjected to CSH (10 ± 0.5% O2) for ten consecutive days. Western blotting and immunofluorescence (IF) staining were used to evaluate phosphorylated tau. Mitochondrial membrane potential (MMP or ∆ψm) and reactive oxygen species (ROS) production were measured to assess mitochondrial function. In vivo diaphragm’s electromyography (dEMG) and local field potential (LFP) recordings from preBötC were employed to assess the respiratory factors and rhythmic representation of preBötC, respectively. Findings showed that ROS production increased significantly in hypoxic groups, associated with a significant decline in ∆ψm. In addition, tau phosphorylation elevated in the brainstem of hypoxic groups. On the other hand, the power of rhythms declined significantly in the preBötC of hypoxic rats, parallel with changes in the respiratory rate, total respiration time, and expiration time. Moreover, there was a positive and statistically significant correlation between LFP rhythm’s power and inspiration time. Our data showed that besides CSH, aging also contributed to mitochondrial dysfunction, tau hyperphosphorylation, LFP rhythms’ power decline, and changes in respiratory factors.












Similar content being viewed by others
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- preBötC:
-
The pre-Bötzinger complex
- VLM:
-
Ventrolateral medulla
- CSH:
-
Chronic sustained hypoxia
- ROS:
-
Reactive oxygen species
- dEMG:
-
Diaphragm muscle electromyography
- LFP:
-
Local field potential
- MMP:
-
Mitochondrial membrane potential
- MAP:
-
Microtubule-associated protein
- GSK3β:
-
Glycogen synthase kinase-3β
- CDK5:
-
Cyclin-dependent kinase 5
- NFTs:
-
Neurofibrillary tangles
- PHF:
-
Paired helical filaments
- CIH:
-
Chronic intermittent hypoxia
- OSAS:
-
Obstructive sleep apnea syndrome
- PP2A:
-
Protein phosphatase A2
- VRG:
-
Ventral respiratory group
- rVRG:
-
Ventral respiratory group, rostral part
- EEG:
-
Electroencephalography
- 4V:
-
Fourth ventricle
- Sp5:
-
Spinal trigeminal tract
- Sp5I:
-
Interpolar nucleus of spinal trigeminal nucleus
- AmbC:
-
Compact part of the ambiguous nucleus
- 12N:
-
Hypoglossal nucleus
- NTS:
-
Nucleus tractus solitarius
- IOD:
-
Inferior olive dorsal nucleus
- IOPr:
-
Inferior olive principal nucleus
- IOM:
-
Inferior olive medial nucleus
- py:
-
Pyramidal tract
- IF:
-
Immunofluorescence
- mtDNA:
-
Mitochondrial DNA
- PHFD:
-
Post hypoxic frequency decline
- VAH:
-
Ventilatory acclimatization to hypoxia
References
Thomas LW, Ashcroft M (2019) Exploring the molecular interface between hypoxia-inducible factor signalling and mitochondria. Cell Mol Life Sci 76(9):1759–1777
Abe H, Semba H, Takeda N (2017) The roles of hypoxia signaling in the pathogenesis of cardiovascular diseases. J Atheroscler Thromb 24(9):884–894
Brent MB (2022) A review of the skeletal effects of exposure to high altitude and potential mechanisms for hypobaric hypoxia-induced bone loss. Bone 154:116258
Asmaro D, Mayall J, Ferguson S (2013) Cognition at altitude: impairment in executive and memory processes under hypoxic conditions. Aviat Space Environ Med 84(11):1159–1165
Kent BD, Mitchell PD, McNicholas WT (2011) Hypoxemia in patients with COPD: cause, effects, and disease progression. Int J Chron Obstruct Pulmon Dis 6:199
Small DH, Cappai R (2006) Alois Alzheimer and Alzheimer’s disease: a centennial perspective. J Neurochem 99(3):708–710
Giacobini E, Gold G (2013) Alzheimer disease therapy—moving from amyloid-β to tau. Nat Rev Neurol 9(12):677–686
Pîrşcoveanu DFV, Pirici I, Tudorică V, Bălşeanu TA, Albu VC, Bondari S, Bumbea AM, Pîrşcoveanu M (2017) Tau protein in neurodegenerative diseases - a review. Rom J Morphol Embryol 58(4):1141–1150
Wagner U et al (1996) Cellular phosphorylation of tau by GSK-3 beta influences tau binding to microtubules and microtubule organisation. J Cell Sci 109(Pt 6):1537–1543
Kimura T, Ishiguro K, Hisanaga S (2014) Physiological and pathological phosphorylation of tau by Cdk5. Front Mol Neurosci 7:65
Wang Y, Mandelkow E (2016) Tau in physiology and pathology. Nat Rev Neurosci 17(1):5–21
Chen K (2020) Phosphorylation of the tau protein in neurodegenerative disease. Biomed J Sci Tech Res 26(4)
Mietelska-Porowska A et al (2014) Tau protein modifications and interactions: their role in function and dysfunction. Int J Mol Sci 15(3):4671–4713
Duquette A et al (2021) Similarities and differences in the pattern of tau hyperphosphorylation in physiological and pathological conditions: impacts on the elaboration of therapies to prevent tau pathology. Front Neurol 11:607680
Kazim SF et al (2022) Chronic intermittent hypoxia enhances pathological tau seeding, propagation, and accumulation and exacerbates Alzheimer-like memory and synaptic plasticity deficits and molecular signatures. Biol Psychiatry 91(4):346–358
Raz L et al (2019) Hypoxia promotes tau hyperphosphorylation with associated neuropathology in vascular dysfunction. Neurobiol Dis 126:124–136
Qiu L et al (2016) Chronic cerebral hypoperfusion enhances tau hyperphosphorylation and reduces autophagy in Alzheimer’s disease mice. Sci Rep 6(1):23964
Bu X-L et al (2015) Serum amyloid-beta levels are increased in patients with obstructive sleep apnea syndrome. Sci Rep 5(1):13917
Liu H et al (2016) Chronic hypoxia facilitates Alzheimer’s disease through demethylation of γ-secretase by downregulating DNA methyltransferase 3b. Alzheimers Dement 12(2):130–143
Biswal S et al (2016) Global hypoxia induced impairment in learning and spatial memory is associated with precocious hippocampal aging. Neurobiol Learn Mem 133:157–170
Chen G-J et al (2003) Transient hypoxia causes Alzheimer-type molecular and biochemical abnormalities in cortical neurons: potential strategies for neuroprotection. J Alzheimers Dis 5:209–228
Jain K et al (2015) Hypobaric hypoxia imbalances mitochondrial dynamics in rat brain hippocampus. Neurol Res Int 2015:742059
Onyango IG, Dennis J, Khan SM (2016) Aging Dis 7(2):201–214
Wu Y, Chen M, Jiang J (2019) Mitochondrial dysfunction in neurodegenerative diseases and drug targets via apoptotic signaling. Mitochondrion 49:35–45
Rey F et al (2022) Mitochondrial dysfunctions in neurodegenerative diseases: role in disease pathogenesis, strategies for analysis and therapeutic prospects. Neural Regen Res 17(4):754
Sena LA, Chandel NS (2012) Physiological roles of mitochondrial reactive oxygen species. Mol Cell 48(2):158–167
Zhang B et al (2022) Role of mitochondrial reactive oxygen species in homeostasis regulation. Redox Rep 27(1):45–52
Melov S et al (2007) Mitochondrial oxidative stress causes hyperphosphorylation of tau. PLoS ONE 2(6):e536
Gaig C, Iranzo A (2012) Sleep-disordered breathing in neurodegenerative diseases. Curr Neurol Neurosci Rep 12(2):205–217
Jorge C et al (2020) Obstructive sleep apnoea and cognitive decline in mild-to-moderate Alzheimer’s disease. Eur Respir J 56(5):2000523
Affoo RH et al (2013) Swallowing dysfunction and autonomic nervous system dysfunction in Alzheimer’s disease: a scoping review of the evidence. J Am Geriatr Soc 61(12):2203–2213
Dutschmann M et al (2010) Upper airway dysfunction of tau-P301L mice correlates with tauopathy in midbrain and ponto-medullary brainstem nuclei. J Neurosci 30(5):1810–1821
Ebel DL, Torkilsen CG, Ostrowski TD (2017) Blunted respiratory responses in the streptozotocin-induced Alzheimer’s disease rat model. J Alzheimers Dis 56:1197–1211
Trease AJ et al (2022) Hyperphosphorylated human tau accumulates at the synapse, localizing on synaptic mitochondrial outer membranes and disrupting respiration in a mouse model of tauopathy. Front Mol Neurosci 15:852368
Smith JC et al (2007) Spatial and functional architecture of the mammalian brain stem respiratory network: a hierarchy of three oscillatory mechanisms. J Neurophysiol 98(6):3370–3387
Smith JC et al (1991) Pre-Bötzinger complex: a brainstem region that may generate respiratory rhythm in mammals. Science 254(5032):726–729
Jiang C, Lipski J (1990) Extensive monosynaptic inhibition of ventral respiratory group neurons by augmenting neurons in the Bötzinger complex in the cat. Exp Brain Res 81(3):639–648
Garcia AJ et al (2016) Chronic intermittent hypoxia alters local respiratory circuit function at the level of the preBötzinger complex. Front Neurosci 10
Kiatchoosakun P et al (2002) Effects of hypoxia on respiratory neural output and lower esophageal sphincter pressure in piglets. Pediatr Res 52(1):50–55
England SJ et al (1995) Activity of respiratory neurons during hypoxia in the chemodenervated cat. J Appl Physiol 78(3):856–861
Richter DW, Müller M (2009) Neural respiratory control during acute hypoxia. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer, Berlin Heidelberg, Berlin, pp 2601–2606
Alonso AC et al (1994) Role of abnormally phosphorylated tau in the breakdown of microtubules in Alzheimer’s disease. Proc Natl Acad Sci 91(12):5562–5566
Nishioka C et al (2022) Axonal transport impairment and its relationship with diffusion tensor imaging metrics of a murine model of p301L tau induced tauopathy. Neuroscience 498:144–154
Menkes-Caspi N et al (2015) Pathological tau disrupts ongoing network activity. Neuron 85(5):959–966
Maleysson V et al (2019) Relevance of electroencephalogram assessment in amyloid and tau pathology in rat. Behav Brain Res 359:127–134
Xu Y, Fan Q (2022) Relationship between chronic hypoxia and seizure susceptibility. CNS Neurosci Ther 28(11):1689–1705
Pamenter ME, Powell FL (2016) Time domains of the hypoxic ventilatory response and their molecular basis. Compr Physiol 6(3):1345–1385
Bayer C et al (2011) Acute versus chronic hypoxia: why a simplified classification is simply not enough. Int J Radiat Oncol Biol Phys 80(4):965–968
Hossienzadeh F et al (2013) Effect of ghrelin on brain edema induced by acute and chronic systemic hypoxia. Neurosci Lett 534:47–51
Subramanian HH, Holstege G (2013) Stimulation of the midbrain periaqueductal gray modulates preinspiratory neurons in the ventrolateral medulla in the rat in vivo. J Comp Neurol 521(13):3083–3098
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134(1):9–21
Hayes MH (1996) Statistical digital signal processing and modeling. Wiley
Soltani Zangbar H et al (2020) A potential entanglement between the spinal cord and hippocampus: theta rhythm correlates with neurogenesis deficiency following spinal cord injury in male rats. J Neurosci Res 98(12):2451–2467
Liao P-C et al (2020) Chapter 1 - isolation of mitochondria from cells and tissues. In: Pon LA, Schon EA (eds) Methods in cell biology. Academic Press, pp 3–31
Hosseini L, Vafaee MS, Badalzadeh R (2020) Melatonin and nicotinamide mononucleotide attenuate myocardial ischemia/reperfusion injury via modulation of mitochondrial function and hemodynamic parameters in aged rats. J Cardiovasc Pharmacol Ther 25(3):240–250
Liu F et al (2018) Mitochondria in ischemic stroke: new insight and implications. Aging Dis 9(5):924–937
Wang W et al (2020) Mitochondria dysfunction in the pathogenesis of Alzheimer’s disease: recent advances. Mol Neurodegener 15:1–22
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552(Pt 2):335–344
Korshunov SS, Skulachev VP, Starkov AA (1997) High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. FEBS Lett 416(1):15–18
Sugrue MM, Tatton WG (2001) Mitochondrial membrane potential in aging cells. Biol Signals Recept 10(3–4):176–188
Madamanchi NR, Runge MS (2007) Mitochondrial dysfunction in atherosclerosis. Circ Res 100(4):460–473
Ohashi S et al (2006) Protective roles of redox-active protein thioredoxin-1 for severe acute pancreatitis. Am J Physiol Gastrointest Liver Physiol 290(4):G772-81
Lobo V et al (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev 4(8):118–126
Reddy PH (2009) Amyloid beta, mitochondrial structural and functional dynamics in Alzheimer’s disease. Exp Neurol 218(2):286–292
Deshpande A et al (2006) Different conformations of amyloid beta induce neurotoxicity by distinct mechanisms in human cortical neurons. J Neurosci 26(22):6011–6018
Cabezas-Opazo FA et al (2015) Mitochondrial dysfunction contributes to the pathogenesis of Alzheimer’s disease. Oxid Med Cell Longev 2015:509654
Aon MA, Cortassa S, O’Rourke B (2010) Redox-optimized ROS balance: a unifying hypothesis. Biochim Biophys Acta 1797(6–7):865–877
Fan Q et al (2013) Aging might augment reactive oxygen species (ROS) formation and affect reactive nitrogen species (RNS) level after myocardial ischemia/reperfusion in both humans and rats. Age (Dordr) 35(4):1017–1026
Calabrese V et al (2004) Increased expression of heat shock proteins in rat brain during aging: relationship with mitochondrial function and glutathione redox state. Mech Ageing Dev 125(4):325–335
Mecocci P et al (1993) Oxidative damage to mitochondrial DNA shows marked age-dependent increases in human brain. Ann Neurol 34(4):609–616
Alquezar C, Arya S, Kao AW (2021) Tau post-translational modifications: dynamic transformers of tau function, degradation, and aggregation. Front Neurol 11:595532
Zhang C-E et al (2014) Hypoxia-induced tau phosphorylation and memory deficit in rats. Neurodegener Dis 14:107–116
Gong C-X et al (1995) Phosphatase activity toward abnormally phosphorylated τ: decrease in Alzheimer disease brain. J Neurochem 65(2):732–738
Guha S, Johnson GV, Nehrke K (2020) The crosstalk between pathological tau phosphorylation and mitochondrial dysfunction as a key to understanding and treating Alzheimer’s disease. Mol Neurobiol 57:5103–5120
Sies H, Jones DP (2020) Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol 21(7):363–383
Jones DP, Sies H (2015) The redox code. Antioxid Redox Signal 23(9):734–746
Mailloux RJ, Jin X, Willmore WG (2014) Redox regulation of mitochondrial function with emphasis on cysteine oxidation reactions. Redox Biol 2:123–139
Martínez-Reyes I, Cuezva JM (2014) The H(+)-ATP synthase: a gate to ROS-mediated cell death or cell survival. Biochem Biophys Acta 1837(7):1099–1112
Rhee SG et al (2000) Hydrogen peroxide: a key messenger that modulates protein phosphorylation through cysteine oxidation. Sci STKE 2000(53):pe1
Tonks NK (2005) Redox redux: revisiting PTPs and the control of cell signaling. Cell 121(5):667–670
Muyllaert D et al (2008) Glycogen synthase kinase-3β, or a link between amyloid and tau pathology? Genes Brain Behav 7(s1):57–66
Toral-Rios D et al (2020) GSK3β and tau protein in Alzheimer’s disease and epilepsy. Front Cell Neurosci 14:19
Liu K et al (2016) Glycation alter the process of tau phosphorylation to change tau isoforms aggregation property. Biochimica et Biophysica Acta (BBA) - Mol Basis Dis 1862(2):192–201
Sui D, Liu M, Kuo M-H (2015) In vitro aggregation assays using hyperphosphorylated tau protein. JoVE 95:e51537
Hemmings BA, Cohen P (1983) [36] Glycogen synthase kinase-3 from rabbit skeletal muscle. Methods in enzymology. Academic Press, pp 337–345
Harik SI et al (1995) Brain glucose metabolism in hypobaric hypoxia. J Appl Physiol 79(1):136–140
Wang SH et al (2009) Cadmium toxicity toward autophagy through ROS-activated GSK-3beta in mesangial cells. Toxicol Sci 108(1):124–131
Pizarro JG et al (2008) Neuroprotective effects of SB-415286 on hydrogen peroxide-induced cell death in B65 rat neuroblastoma cells and neurons. Int J Dev Neurosci 26(3–4):269–276
Chauhan NB, Siegel GJ, Feinstein DL (2005) Propentofylline attenuates tau hyperphosphorylation in Alzheimer’s Swedish mutant model Tg2576. Neuropharmacology 48(1):93–104
Strocchi P, Pession A, Dozza B (2003) Up-regulation of cDK5/p35 by oxidative stress in human neuroblastoma IMR-32 cells. J Cell Biochem 88(4):758–765
Shea TB et al (2004) Cyclin-dependent kinase 5 increases perikaryal neurofilament phosphorylation and inhibits neurofilament axonal transport in response to oxidative stress. J Neurosci Res 76(6):795–800
Smith PD, O’Hare MJ, Park DS (2004) Emerging pathogenic role for cyclin dependent kinases in neurodegeneration. Cell Cycle 3(3):289–291
Patrick GN et al (1999) Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402(6762):615–622
Gong C-X et al (1993) Phosphoprotein phosphatase activities in Alzheimer disease brain. J Neurochem 61(3):921–927
Liu R et al (2008) Phosphorylated PP2A (tyrosine 307) is associated with Alzheimer neurofibrillary pathology. J Cell Mol Med 12(1):241–257
Orellana AMM et al (2015) Age-related neuroinflammation and changes in AKT-GSK-3β and WNT/β-catenin signaling in rat hippocampus. Aging 7(12):1094–1111
Lee K-Y et al (1999) Elevated neuronal Cdc2-like kinase activity in the Alzheimer disease brain. Neurosci Res 34(1):21–29
Kelleher I et al (2007) Kinase activities increase during the development of tauopathy in htau mice. J Neurochem 103(6):2256–2267
Leroy A et al (2010) Spectroscopic studies of GSK3β phosphorylation of the neuronal tau protein and its interaction with the N-terminal domain of apolipoprotein E. J Biol Chem 285(43):33435–33444
Paudel HK et al (1993) Brain proline-directed protein kinase phosphorylates tau on sites that are abnormally phosphorylated in tau associated with Alzheimer’s paired helical filaments. J Biol Chem 268(31):23512–23518
Uematsu M et al (2018) Brainstem tau pathology in Alzheimer’s disease is characterized by increase of three repeat tau and independent of amyloid β. Acta Neuropathol Commun 6(1):1
Wai MS et al (2009) Co-localization of hyperphosphorylated tau and caspases in the brainstem of Alzheimer’s disease patients. Biogerontology 10(4):457–469
Hirano A, Zimmerman HM (1962) Alzheimer’s neurofibrillary changes. A topographic study. Arch Neurol 7:227–242
Ishii T (1966) Distribution of Alzheimer’s neurofibrillary changes in the brain stem and hypothalamus of senile dementia. Acta Neuropathol 6(2):181–187
Zhu K et al (2019) Primary age-related tauopathy in human subcortical nuclei. Front Neurosci 13:529
Liu SJ et al (2003) Overactivation of glycogen synthase kinase-3 by inhibition of phosphoinositol-3 kinase and protein kinase C leads to hyperphosphorylation of tau and impairment of spatial memory. J Neurochem 87(6):1333–1344
Buée L et al (2000) Tau protein isoforms, phosphorylation and role in neurodegenerative disorders. Brain Res Rev 33(1):95–130
Zhang Y et al (2022) Tauopathies: new perspectives and challenges. Mol Neurodegener 17(1):28
Hoover BR et al (2010) Tau mislocalization to dendritic spines mediates synaptic dysfunction independently of neurodegeneration. Neuron 68(6):1067–1081
Yoshiyama Y et al (2007) Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron 53(3):337–351
Wu M et al (2021) The role of pathological tau in synaptic dysfunction in Alzheimer’s diseases. Transl Neurodegen 10:1–11
Holton CM et al (2020) Longitudinal changes in EEG power, sleep cycles and behaviour in a tau model of neurodegeneration. Alzheimers Res Ther 12(1):84
Scarpa JR et al (2021) NREM delta power and AD-relevant tauopathy are associated with shared cortical gene networks. Sci Rep 11(1):7797
Holth JK et al (2017) Altered sleep and EEG power in the P301S tau transgenic mouse model. Ann Clin Transl Neurol 4:180–190. https://doi.org/10.1002/acn3.390
Ahnaou A et al (2020) Functional alterations in the olfactory neuronal circuit occur before hippocampal plasticity deficits in the P301S mouse model of tauopathy: implications for early diagnosis and translational research in Alzheimer’s disease. Int J Mol Sci 21(15):5431
Buzsáki G, Anastassiou CA, Koch C (2012) The origin of extracellular fields and currents—EEG, ECoG, LFP and spikes. Nat Rev Neurosci 13(6):407–420
Polich J (1997) EEG and ERP assessment of normal aging. Electroencephal Clin Neurophysiol/Evoked Potent Sect 104(3):244–256
Kreuzer M et al (2020) Spectral and entropic features are altered by age in the electroencephalogram in patients under sevoflurane anesthesia. Anesthesiology 132(5):1003–1016
Babiloni C et al (2006) Sources of cortical rhythms in adults during physiological aging: a multicentric EEG study. Hum Brain Mapp 27(2):162–172
Jyoti A et al (2015) Progressive age-related changes in sleep and EEG profiles in the PLB1Triple mouse model of Alzheimer’s disease. Neurobiol Aging 36(10):2768–2784
Agnati LF et al (1990) Cellular and synaptic alterations in the aging brain. Aging Clin Exp Res 2(1):5–25
Mostany R et al (2013) Altered synaptic dynamics during normal brain aging. J Neurosci 33(9):4094
Ishii R et al (2017) Healthy and pathological brain aging: from the perspective of oscillations, functional connectivity, and signal complexity. Neuropsychobiology 75(4):151–161
Monteau R, Hilaire G (1991) Spinal respiratory motoneurons. Prog Neurobiol 37(2):83–144
Walsh MP, Marshall JM (2006) The early effects of chronic hypoxia on the cardiovascular system in the rat: role of nitric oxide. J Physiol 575(1):263–275
Perim RR, Bonagamba LGH, Machado BH (2015) Cardiovascular and respiratory outcome of preconditioned rats submitted to chronic intermittent hypoxia. Exp Physiol 100(9):1008–1017
Kinkead R, Mitchell GS (1999) Time-dependent hypoxic ventilatory responses in rats: effects of ketanserin and 5-carboxamidotryptamine. Am J Physiol-Regul Integr Comp Physiol 277(3):R658–R666
Aaron EA, Powell FL (1993) Effect of chronic hypoxia on hypoxic ventilatory response in awake rats. J Appl Physiol 74(4):1635–1640
Dempsey JA et al (2014) Role of chemoreception in cardiorespiratory acclimatization to, and deacclimatization from, hypoxia. J Appl Physiol (1985) 116(7):858–66
Forster HV et al (1976) Ventilatory control in peripheral chemoreceptor-denervated ponies during chronic hypoxemia. J Appl Physiol 41(6):878–885
Forster HV et al (1974) Evidence of altered regulation of ventilation during exposure to hypoxia. Respir Physiol 20(3):379–392
Wilkinson KA et al (2010) Chronic hypoxia increases the gain of the hypoxic ventilatory response by a mechanism in the central nervous system. J Appl Physiol (1985) 109(2):424–30
Attems J et al (2005) Cause of death in demented and non-demented elderly inpatients; an autopsy study of 308 cases. J Alzheimers Dis 8(1):57–62
Katz DM et al (2009) Breathing disorders in Rett syndrome: progressive neurochemical dysfunction in the respiratory network after birth. Respir Physiol Neurobiol 168(1–2):101–108
Menuet C et al (2012) Isoflurane anesthesia precipitates tauopathy and upper airways dysfunction in pre-symptomatic Tau.P301L mice: possible implication for neurodegenerative diseases. Neurobiol Dis 46(1):234–43
Menuet C et al (2011) Raphé tauopathy alters serotonin metabolism and breathing activity in terminal Tau.P301L mice: possible implications for tauopathies and Alzheimer’s disease. Respir Physiol Neurobiol 178(2):290–303
Terwel D et al (2005) Changed conformation of mutant tau-P301L underlies the moribund tauopathy, absent in progressive, nonlethal axonopathy of tau-4R/2N transgenic mice. J Biol Chem 280(5):3963–3973
Wu M, Haxhiu MA, Johnson SM (2005) Hypercapnic and hypoxic responses require intact neural transmission from the pre-Bötzinger complex. Respir Physiol Neurobiol 146(1):33–46
Baux G, Simonneau M, Tauc L (1981) Action of colchicine on membrane currents and synaptic transmission in Aplysia ganglion cells. J Neurobiol 12(1):75–85
Donoso AL, Fernandez HL (1973) Effect of colchicine on long lasting synaptic modifications induced by hyperactivity. Experientia 29(9):1089–1090
Sharma G, Goodwin J (2006) Effect of aging on respiratory system physiology and immunology. Clin Interv Aging 1(3):253–260
Krumpe PE et al (1985) The aging respiratory system. Clin Geriatr Med 1(1):143–175
Sprung J, Gajic O, Warner DO (2006) Review article: age related alterations in respiratory function - anesthetic considerations. Can J Anaesth 53(12):1244–1257
Logothetis NK et al (2001) Neurophysiological investigation of the basis of the fMRI signal. Nature 412(6843):150–157
McCormick DA et al (2003) Persistent cortical activity: mechanisms of generation and effects on neuronal excitability. Cereb Cortex 13(11):1219–1231
Penttonen M et al (1998) Gamma frequency oscillation in the hippocampus of the rat: intracellular analysis in vivo. Eur J Neurosci 10(2):718–728
Anderson J et al (2000) Stimulus dependence of two-state fluctuations of membrane potential in cat visual cortex. Nat Neurosci 3(6):617–621
Logothetis NK, Wandell BA (2004) Interpreting the BOLD signal. Annu Rev Physiol 66:735–769
Henrie JA, Shapley R (2005) LFP power spectra in V1 cortex: the graded effect of stimulus contrast. J Neurophysiol 94(1):479–490
Buzsáki G, Wang XJ (2012) Mechanisms of gamma oscillations. Annu Rev Neurosci 35:203–225
Lytton WW, Sejnowski TJ (1991) Simulations of cortical pyramidal neurons synchronized by inhibitory interneurons. J Neurophysiol 66(3):1059–1079
Gray CM et al (1989) Oscillatory responses in cat visual cortex exhibit inter-columnar synchronization which reflects global stimulus properties. Nature 338(6213):334–337
Brown P (2000) Cortical drives to human muscle: the Piper and related rhythms. Prog Neurobiol 60(1):97–108
Engelhard B, Vaadia E (2014) Spatial computation with gamma oscillations. Front Syst Neurosci 8
Acknowledgements
The authors would like to express their gratitude to Pouran Karimi for her assistance in molecular analysis. Our data were derived from the thesis of Jamal Khalilpour for a Ph.D. degree in physiology.
Funding
This work was supported by grants (No. 65698) from Drug Applied Research Center, Tabriz University of Medical Science.
Author information
Authors and Affiliations
Contributions
JK and PS conceived the study and designed the experiments. JK wrote the manuscript. JK, PS, and HSZ performed the experiments with the help of ZZ. JK, PS, HSZ, MRA, and FQP interpreted data and contributed to the discussion. All authors reviewed and concurred with the final manuscript. PS takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding authors
Ethics declarations
Ethics Approval
All procedures and handling techniques were approved by the Ethical Committee for Animal Research of Tabriz University of Medical Sciences (IR.TBZMED.VCR.REC.1399.404), and surgical procedures were conducted under aseptic conditions.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khalilpour, J., Zangbar, H.S., Alipour, M.R. et al. Chronic Sustained Hypoxia Leads to Brainstem Tauopathy and Declines the Power of Rhythms in the Ventrolateral Medulla: Shedding Light on a Possible Mechanism. Mol Neurobiol 61, 3121–3143 (2024). https://doi.org/10.1007/s12035-023-03763-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03763-4