Abstract
Objective
This study was to investigate the variables in bone marrow harvesting procedure and individual donor factors which can potentially affect the yield of mesenchymal stromal cells (MSC).
Methods
We determined the yield of MSC from bone marrow under different clinical conditions by comparing the MSC colony numbers from: (1) donors of different ages; (2) healthy donors and patients with leukemia; (3) bone marrow aspirated at different time points during marrow harvesting; (4) bone marrow harvested by different needles.
Results
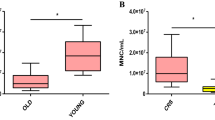
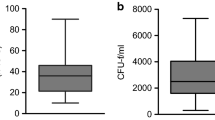
During the process of harvesting, the number of MSC significantly decreased with increase number of aspiration, from 675/ml at the initial decreased to 60/ml after 100 ml bone marrow aspirated, and 50/ml after 200 ml bone marrow aspirated. The number of MSC retrieved from leukemia patients (99/ml bone marrow) was significantly lower than that of healthy donors (708/ml bone marrow). However, there was no significant difference in growth rate. There was no significant age-related difference of MSC yielded from donors <55 years. And there was no significant difference in MSC number between the samples from single end-holed needle and those from multiple-side-hole needle.
Conclusion
The optimal bone marrow samples for MSC collection should be obtained earlier in the process of harvesting procedure. Bone marrow from donors <55 years was equally good as MSC sources. The autologous MSC from leukemia patients can be utilized for in-vitro MSC expansion.
Similar content being viewed by others
References
Fibbe WE, Noort WA. Mesenchymal stem cells and hematopoietic stem cell transplantation. Ann N Y Acad Sci 2003; 996:235–244.
Devine SM, Hoffman R. Role of mesenchymal stem cells in hematopoietic stem cell transplantation. Curr Opin Hematol 2000; 7: 358–363.
Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005; 105:1815–1822.
Beyth S, Borovsky Z, Mevorach D, et al. Human mesenchymal stem cells alter antigen-presenting cell maturation and induce T-cell unresponsiveness. Blood 2005; 105:2214–2219.
Chung NG, Jeong DC, Park SJ, et al. Cotransplantation of marrow stromal cells may prevent lethal graft-versus-host disease in major histocompatibility complex mismatched murine hematopoietic stem cell transplantation. Int J Hematol 2004; 80:370–376.
Le Blanc K, Rasmusson I, Sundberg B, et al. Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 2004; 363:1439–1441.
Le Blanc K, Ringdén O. Immunobiology of human mesenchymal stem cells and future use in hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005; 11:321–334.
Maitra B, Szekely E, Gjini K, et al. Human mesenchymal stem cells support unrelated donor hematopoietic stem cells and suppress T-cell activation. Bone Marrow Transplant 2004; 33:597–604.
Lazarus HM, Koc ON, Devine SM, et al. Cotransplantation of HLA-identical sibling culture-expanded mesenchymal stem cells and hematopoietic stem cells in hematologic malignancy patients. Biol Blood Marrow Transplant 2005; 11:389–398.
Ringdén O, Uzunel M, Rasmusson I, et al. Mesenchymal stem cells for treatment of therapy-resistant graft-versus-host disease. Transplantation 2006; 81:1390–1397.
Gonzalo-Daganzo R, Regidor C, Martín-Donaire T, et al. Results of a pilot study on the use of third-party donor mesenchymal stromal cells in cord blood transplantation in adults. Cytotherapy 2009; 11:278–288.
von Bonin M, Stölzel F, Goedecke A, et al. Treatment of refractory acute GVHD with third-party MSC expanded in platelet lysate-containing medium. Bone Marrow Transplant 2009; 43:245–251.
Li J, Law HK, Lau YL, et al. Differential damage and recovery of human mesenchymal stem cells after exposure to chemotherapeutic agents. Br J Haematol 2004; 127:326–334.
Muschler GF, Boehm C, Easley K. Aspiration to obtain osteoblast progenitor cells from human bone marrow: the influence of aspiration volume. J Bone Joint Surg Am 1997; 79:1699–1709.
Castro-Malaspina H, Gay RE, Resnick G, et al. Characterization of human-bone marrow fibroblast colony-forming cells (CFU-F) and their progeny. Blood 1980; 56:289–301.
Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284:143–147.
Galotto M, Berisso G, Delfino L, et al. Stromal damage as consequence of high-dose chemo/radiotherapy in bone marrow transplant recipients. Exp Hematol 1999; 27:1460–1466.
Lebedinskaia OV, Gorskaia IuF, Shuklina Elu, et al. Age changes in the numbers of stromal precursor cells in the animal bone marrow. Morfologiia 2004; 126:46–49.
Scutt A, Kollenkirchen U, Bertram P. Effect of age and ovariectomy on fibroblastic colony-forming unit numbers in rat bone marrow. Calcif Tissue Int 1996; 59:309–310.
Bergman RJ, Gazit D, Kahn AJ, et al. Age-related changes in osteogenic stem cells in mice. J Bone Miner Res 1996; 11:568–577.
Egrise D, Martin D, Vienne A, et al. The number of fibroblastic colonies formed from bone marrow is decreased and the in vitro proliferation rate of trabecular bone cells increased in aged rats. Bone 1992; 13: 355–361.
Bellows CG, Pei W, Jia Y, et al. Proliferation, differentiation and self-renewal of osteoprogenitors in vertebral cell populations from aged and young female rats. Mech Ageing Dev 2003; 124:747–757.
Justesen J, Stenderup K, Eriksen EF, et al. Maintenance of osteoblastic and adipocytic differentiation potential with age and osteoporosis in human marrow stromal cell cultures. Calcif Tissue Int 2002; 71:36–44.
Oreffo RO, Bennett A, Carr AJ, et al. Patients with primary osteoarthritis show no change with ageing in the number of osteogenic precursors. Scand J Rheumatol 1998; 27:415–424.
Oreffo RO, Bord S, Triffitt JT. Skeletal progenitor cells and ageing human populations. Clin Sci (Lond) 1998; 94:549–555.
Brockbank KG, Ploemacher RE, van Peer CM. An in vitro analysis of murine hemopoietic fibroblastoid progenitors and fibroblastoid cell function during aging. Mech Ageing Dev 1983; 22:11–21.
Stenderup K, Justesen J, Eriksen EF, et al. Number and proliferative capacity of osteogenic stem cells are maintained during aging and in patients with osteoporosis. J Bone Miner Res 2001; 16:1120–1129.
Katsuno M, Hirata J, Kaneko S, et al. Serial studies of bone marrow-derived fibroblastoid colony-forming cells and granulocyte/macrophage precursor cells in patients with acute leukemia. Acta Haematol 1986; 76:185–191.
Nagao T, Yamauchi K, Komatsuda M. Serial in vitro bone marrow fibroblast culture in human leukemia. Blood 1983; 61:589–592.
Nara N, Jinnai I, Imai Y, et al. Reduction of granulocyte-macrophage progenitor cells (CFU-C) and fibroblastoid colony-forming units (CFU-F) by leukemic cells in human and murine leukemia. Acta Haematol 1984; 72:171–180.
Nagao T, Yamauchi K, Komatsuda M, et al. Inhibition of human bone marrow fibroblast colony formation by leukemic cells. Blood 1983; 62:1261–1265.
Wu LP, Chen FX, Lu HM. Biological characteristics of bone marrow-derived mesenchymal stem cells in children with acute leukemia. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2009; 17:734–738.
Yin WJ, Yang PD, Huang YZ, et al. Comparison of biological characteristics of bone marrow mesenchymal stem cells in acute myeloid leukemia with those from non-leukemia. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2009; 17:395–399.
Tanikawa S, Sakamaki H, Mori S, et al. Relationship between the presence of side-holes in bone marrow aspiration needle and the number of harvested bone marrow mononuclear cells. Rinsho Ketsueki 1997; 38:1249–1253.
Lannert H, Able T, Becker S, et al. Optimizing BM harvesting from normal adult donors. Bone Marrow Transplant 2008; 42:443–447.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Wong, W.HS., Chan, S. et al. Factors affecting mesenchymal stromal cells yield from bone marrow aspiration. Chin. J. Cancer Res. 23, 43–48 (2011). https://doi.org/10.1007/s11670-011-0043-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11670-011-0043-1