Abstract
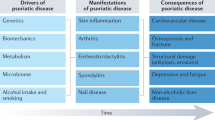
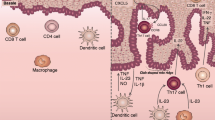
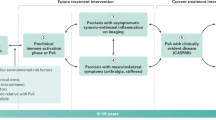
The clinical picture of psoriatic arthritis (PsA) is heterogeneous, potentially involving numerous organs and tissues, such as skin and joint. From a clinical point of view, discrete tissue PsA features develop and respond to treatments apparently independently. The pathogenic events occurring in the various tissues are only partially understood. Although the vast majority of known genetic predisposing factors are shared between patients with skin psoriasis (PSO) and those affected by PsA, some tissue-specific variants have been identified. Furthermore, current data suggest that the TNF pathway and IL-23/Th17 pathways may be differentially activated in distinct tissue sites. In this review, we briefly describe current knowledge on the pathogenesis of PsA in terms of genetic predisposition, environmental factors and immunology, advancing our hypothesis to explain why a common immunologic process can express itself with significant differences in various tissues.

Similar content being viewed by others
References
Ritchlin CT, Colbert RA, Gladman DD (2017) Psoriatic arthritis. N Engl J Med 376(10):957–970. https://doi.org/10.1056/NEJMra1505557
Ogdie A, Weiss P (2015) The epidemiology psoriatic arthritis. Rheum Dis Clin N Am 41(4):545–568. https://doi.org/10.1016/j.rdc.2015.07.001
Lin VW, Ringold S, Devine EB (2012) Comparison of ustekinumab with other biological agents for the treatment of moderate to severe plaque psoriasis: a Bayesian network meta-analysis. Arch Dermatol 148(12):1403–1410. https://doi.org/10.1001/2013.jamadermatol.238
Grjibovski AM, Olsen AO, Magnus P, Harris JR (2007) Psoriasis in Norwegian twins: contribution of genetic and environmental effects. J Eur Acad Dermatol Venereol JEADV 21(10):1337–1134. https://doi.org/10.1111/j.1468-3083.2007.02268.x
Lønnberg AS, Skov L, Skytthe A, Kyvik KO, Pedersen OB, Thomsen SF (2013) Heritability of psoriasis in a large twin sample. Br J Dermatol 169(2):412–441. https://doi.org/10.1111/bjd.12375
O’Rielly DD, Rahman P (2014) Genetics of psoriatic arthritis. Best Pract Res Clin Rheumatol 28(5):673–685. https://doi.org/10.1016/j.berh.2014.10.010
Prinz JC (2017) Autoimmune aspects of psoriasis: heritability and autoantigens. Autoimmun Rev 16(9):970–979. https://doi.org/10.1016/j.autrev.2017.07.011
Stuart PE, Nair RP, Tsoi LC, Tejasvi T, Das S, Kang HM, Ellinghaus E, Chandran V, Callis-Duffin K, Ike R, Li Y, Wen X, Enerbäck C, Gudjonsson JE, Kõks S, Kingo K, Esko T, Mrowietz U, Reis A, Wichmann HE, Gieger C, Hoffmann P, Nöthen MM, Winkelmann J, Kunz M, Moreta EG, Mease PJ, Ritchlin CT, Bowcock AM, Krueger GG, Lim HW, Weidinger S, Weichenthal M, Voorhees JJ, Rahman P, Gregersen PK, Franke A, Gladman DD, Abecasis GR, Elder JT (2015) Genome-wide association analysis of psoriatic arthritis and cutaneous psoriasis reveals differences in their genetic architecture. Am J Hum Genet 97(6):816–836. https://doi.org/10.1016/j.ajhg.2015.10.019
Zhang Z, Yuan J, Tian Z, Xu J, Lu Z (2017) Investigation of 36 non-HLA (human leucocyte antigen) psoriasis susceptibility loci in a psoriatic arthritis cohort. Arch Dermatol Res 309(2):71–77. https://doi.org/10.1007/s00403-016-1706-z
Winchester R, Minevich G, Steshenko V, Kirby B, Kane D, Greenberg DA, FitzGerald O (2012) HLA associations reveal genetic heterogeneity in psoriatic arthritis and in the psoriasis phenotype. Arthritis Rheum 64(4):1134–1144. https://doi.org/10.1002/art.33415
Balendran N, Clough RL, Arguello JR et al (1999) Characterization of the major susceptibility region for psoriasis at chromosome 6p21.3. J Invest Dermatol 113(3):322–328. https://doi.org/10.1046/j.1523-1747.1999.00710.x
FitzGerald O, Haroon M, Giles JT, Winchester R (2015) Concepts of pathogenesis in psoriatic arthritis: genotype determines clinical phenotype. Arthritis Res Ther 17(1):115. https://doi.org/10.1186/s13075-015-0640-3
Gorman JA, Hundhausen C, Errett JS, Stone AE, Allenspach EJ, Ge Y, Arkatkar T, Clough C, Dai X, Khim S, Pestal K, Liggitt D, Cerosaletti K, Stetson DB, James RG, Oukka M, Concannon P, Gale M, Buckner JH, Rawlings DJ (2017) The A946T variant of the RNA sensor IFIH1 mediates an interferon program that limits viral infection but increases the risk for autoimmunity. Nat Immunol 18(7):744–752. https://doi.org/10.1038/ni.3766
Velazquez L, Fellous M, Stark GR, Pellegrini S (1992) A protein tyrosine kinase in the interferon alpha/beta signaling pathway. Cell 70(2):313–322. https://doi.org/10.1016/0092-8674(92)90105-L
Vereecke L, Beyaert R, van Loo G (2009) The ubiquitin-editing enzyme A20 (TNFAIP3) is a central regulator of immunopathology. Trends Immunol 30(8):383–391. https://doi.org/10.1016/j.it.2009.05.007
Ramirez VP, Gurevich I, Aneskievich BJ (2012) Emerging roles for TNIP1 in regulating post-receptor signaling. Cytokine Growth Factor Rev 23(3):109–118. https://doi.org/10.1016/j.cytogfr.2012.04.002
Doyle MS, Collins ES, FitzGerald OM, Pennington SR (2012) New insight into the functions of the interleukin-17 receptor adaptor protein Act1 in psoriatic arthritis. Arthritis Res Ther 14(5):226. https://doi.org/10.1186/ar4071
Boisson B, Puel A, Picard C, Casanova J-L (2017) Human IκBα gain of function: a severe and syndromic immunodeficiency. J Clin Immunol 37(5):397–412. https://doi.org/10.1007/s10875-017-0400-z
Ruben SM, Klement JF, Coleman TA, Maher M, Chen CH, Rosen CA (1992) I-Rel: a novel rel-related protein that inhibits NF-kappa B transcriptional activity. Genes Dev 6(5):745–760. https://doi.org/10.1101/gad.6.5.745
Shan Q, Zeng Z, Xing S, Li F, Hartwig SM, Gullicksrud JA, Kurup SP, van Braeckel-Budimir N, Su Y, Martin MD, Varga SM, Taniuchi I, Harty JT, Peng W, Badovinac VP, Xue HH (2017) The transcription factor Runx3 guards cytotoxic CD8(+) effector T cells against deviation towards follicular helper T cell lineage. Nat Immunol 18(8):931–939. https://doi.org/10.1038/ni.3773
Bowes J, Budu-Aggrey A, Huffmeier U, Uebe S, Steel K, Hebert HL, Wallace C, Massey J, Bruce IN, Bluett J, Feletar M, Morgan AW, Marzo-Ortega H, Donohoe G, Morris DW, Helliwell P, Ryan AW, Kane D, Warren RB, Korendowych E, Alenius GM, Giardina E, Packham J, McManus R, FitzGerald O, McHugh N, Brown MA, Ho P, Behrens F, Burkhardt H, Reis A, Barton A (2015) Dense genotyping of immune-related susceptibility loci reveals new insights into the genetics of psoriatic arthritis. Nat Commun 6(1):6046. https://doi.org/10.1038/ncomms7046
Gill T, Asquith M, Rosenbaum JT, Colbert RA (2015) The intestinal microbiome in spondyloarthritis. Curr Opin Rheumatol 27(4):319–325. https://doi.org/10.1097/BOR.0000000000000187
Scher JU, Littman DR, Abramson SB (2016) Microbiome in inflammatory arthritis and human rheumatic diseases. Arthritis Rheumatol Hoboken NJ 68(1):35–45. https://doi.org/10.1002/art.39259
Yan D, Issa N, Afifi L, Jeon C, Chang HW, Liao W (2017) The role of the skin and gut microbiome in psoriatic disease. Curr Dermatol Rep 6(2):94–103. https://doi.org/10.1007/s13671-017-0178-5
Perera GK, Di Meglio P, Nestle FO (2012) Psoriasis. Annu Rev Pathol 7(1):385–422. https://doi.org/10.1146/annurev-pathol-011811-132448
Boehncke W-H, Schön MP (2015) Psoriasis. Lancet Lond Engl 386(9997):983–994. https://doi.org/10.1016/S0140-6736(14)61909-7
Grozdev I, Korman N, Tsankov N (2014) Psoriasis as a systemic disease. Clin Dermatol 32(3):343–350. https://doi.org/10.1016/j.clindermatol.2013.11.001
Gunes AT, Fetil E, Akarsu S, Ozbagcivan O, Babayeva L (2015) Possible triggering effect of influenza vaccination on psoriasis. J Immunol Res 2015:258430. https://doi.org/10.1155/2015/258430
Gilliet M, Cao W, Liu Y-J (2008) Plasmacytoid dendritic cells: sensing nucleic acids in viral infection and autoimmune diseases. Nat Rev Immunol 8(8):594–606. https://doi.org/10.1038/nri2358
Albanesi C, De Pità O, Girolomoni G (2007) Resident skin cells in psoriasis: a special look at the pathogenetic functions of keratinocytes. Clin Dermatol 25(6):581–588. https://doi.org/10.1016/j.clindermatol.2007.08.013
Pène J, Chevalier S, Preisser L et al (2008) Chronically inflamed human tissues are infiltrated by highly differentiated Th17 lymphocytes. J Immunol Baltim Md 1950 180:7423–7430
Zaba LC, Cardinale I, Gilleaudeau P, Sullivan-Whalen M, Suárez-Fariñas M, Fuentes-Duculan J, Novitskaya I, Khatcherian A, Bluth MJ, Lowes MA, Krueger JG (2007) Amelioration of epidermal hyperplasia by TNF inhibition is associated with reduced Th17 responses. J Exp Med 204(13):3183–3194. https://doi.org/10.1084/jem.20071094
Zaba LC, Fuentes-Duculan J, Eungdamrong NJ, Abello MV, Novitskaya I, Pierson KC, Gonzalez J, Krueger JG, Lowes MA (2009) Psoriasis is characterized by accumulation of immunostimulatory and Th1/Th17 cell-polarizing myeloid dendritic cells. J Invest Dermatol 129(1):79–88. https://doi.org/10.1038/jid.2008.194
McGeachy MJ, Chen Y, Tato CM et al (2009) The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol 10(3):314–324. https://doi.org/10.1038/ni.1698
Duhen T, Geiger R, Jarrossay D, Lanzavecchia A, Sallusto F (2009) Production of interleukin 22 but not interleukin 17 by a subset of human skin-homing memory T cells. Nat Immunol 10(8):857–863. https://doi.org/10.1038/ni.1767
Mashiko S, Bouguermouh S, Rubio M, Baba N, Bissonnette R, Sarfati M (2015) Human mast cells are major IL-22 producers in patients with psoriasis and atopic dermatitis. J Allergy Clin Immunol 136(2):351–359.e1. https://doi.org/10.1016/j.jaci.2015.01.033
Nograles KE, Zaba LC, Guttman-Yassky E, Fuentes-Duculan J, Suárez-Fariñas M, Cardinale I, Khatcherian A, Gonzalez J, Pierson KC, White TR, Pensabene C, Coats I, Novitskaya I, Lowes MA, Krueger JG (2008) Th17 cytokines interleukin (IL)-17 and IL-22 modulate distinct inflammatory and keratinocyte-response pathways. Br J Dermatol 159:1092–1102. https://doi.org/10.1111/j.1365-2133.2008.08769.x
Witowski J, Pawlaczyk K, Breborowicz A et al (2000) IL-17 stimulates intraperitoneal neutrophil infiltration through the release of GRO alpha chemokine from mesothelial cells. J Immunol Baltim Md 1950 165:5814–5821
Cai Y, Shen X, Ding C, Qi C, Li K, Li X, Jala VR, Zhang HG, Wang T, Zheng J, Yan J (2011) Pivotal role of dermal IL-17-producing γδ T cells in skin inflammation. Immunity 35(4):596–610. https://doi.org/10.1016/j.immuni.2011.08.001
Teunissen MBM, Yeremenko NG, Baeten DLP, Chielie S, Spuls PI, de Rie MA, Lantz O, Res PCM (2014) The IL-17A-producing CD8+ T-cell population in psoriatic lesional skin comprises mucosa-associated invariant T cells and conventional T cells. J Invest Dermatol 134(12):2898–2907. https://doi.org/10.1038/jid.2014.261
Boniface K, Bernard F-X, Garcia M et al (2005) IL-22 inhibits epidermal differentiation and induces proinflammatory gene expression and migration of human keratinocytes. J Immunol Baltim Md 1950 174:3695–3702
Belasco J, Louie JS, Gulati N, Wei N, Nograles K, Fuentes-Duculan J, Mitsui H, Suárez-Fariñas M, Krueger JG (2015) Comparative genomic profiling of synovium versus skin lesions in psoriatic arthritis. Arthritis Rheumatol Hoboken NJ 67(4):934–944. https://doi.org/10.1002/art.38995
Costello PJ, Winchester RJ, Curran SA, et al (2001) Psoriatic arthritis joint fluids are characterized by CD8 and CD4 T cell clonal expansions appear antigen driven. J Immunol Baltim Md 1950 166:2878–2886
Kruithof E, Baeten D, De Rycke L et al (2005) Synovial histopathology of psoriatic arthritis, both oligo- and polyarticular, resembles spondyloarthropathy more than it does rheumatoid arthritis. Arthritis Res Ther 7(3):R569–R580. https://doi.org/10.1186/ar1698
van Baarsen LG, Lebre MC, van der Coelen D et al (2014) Heterogeneous expression pattern of interleukin 17A (IL-17A), IL-17F and their receptors in synovium of rheumatoid arthritis, psoriatic arthritis and osteoarthritis: possible explanation for nonresponse to anti-IL-17 therapy? Arthritis Res Ther 16(4):426. https://doi.org/10.1186/s13075-014-0426-z
Leijten EFA, van Kempen TS, Boes M, Michels-van Amelsfort JMR, Hijnen D, Hartgring SAY, van Roon JAG, Wenink MH, Radstake TRDJ (2015) Brief report: enrichment of activated group 3 innate lymphoid cells in psoriatic arthritis synovial fluid. Arthritis Rheumatol Hoboken NJ 67(10):2673–2678. https://doi.org/10.1002/art.39261
Noordenbos T, Blijdorp I, Chen S, Stap J, Mul E, Cañete JD, Lubberts E, Yeremenko N, Baeten D (2016) Human mast cells capture, store, and release bioactive, exogenous IL-17A. J Leukoc Biol 100(3):453–462. https://doi.org/10.1189/jlb.3HI1215-542R
Zhang LY, Ogdie AR, Schumacher HR (2012) Light and electron microscopic features of synovium in patients with psoriatic arthritis. Ultrastruct Pathol 36(4):207–218. https://doi.org/10.3109/01913123.2011.651523
Thorarensen SM, Lu N, Ogdie A, Gelfand JM, Choi HK, Love TJ (2017) Physical trauma recorded in primary care is associated with the onset of psoriatic arthritis among patients with psoriasis. Ann Rheum Dis 76(3):521–525. https://doi.org/10.1136/annrheumdis-2016-209334
Benjamin M, McGonagle D (2009) The enthesis organ concept and its relevance to the spondyloarthropathies. Adv Exp Med Biol 649:57–70. https://doi.org/10.1007/978-1-4419-0298-6_4
McGonagle D, Lories RJU, Tan AL, Benjamin M (2007) The concept of a “synovio-entheseal complex” and its implications for understanding joint inflammation and damage in psoriatic arthritis and beyond. Arthritis Rheum 56(8):2482–2491. https://doi.org/10.1002/art.22758
McGonagle D, Benjamin M, Tan AL (2009) The pathogenesis of psoriatic arthritis and associated nail disease: not autoimmune after all? Curr Opin Rheumatol 21(4):340–347. https://doi.org/10.1097/BOR.0b013e32832c6ab9
Tan AL, Benjamin M, Toumi H, Grainger AJ, Tanner SF, Emery P, McGonagle D (2007) The relationship between the extensor tendon enthesis and the nail in distal interphalangeal joint disease in psoriatic arthritis—a high-resolution MRI and histological study. Rheumatol Oxf Engl 46(2):253–256. https://doi.org/10.1093/rheumatology/kel214
Coates LC, Kavanaugh A, Mease PJ, Soriano ER, Laura Acosta-Felquer M, Armstrong AW, Bautista-Molano W, Boehncke WH, Campbell W, Cauli A, Espinoza LR, FitzGerald O, Gladman DD, Gottlieb A, Helliwell PS, Husni ME, Love TJ, Lubrano E, McHugh N, Nash P, Ogdie A, Orbai AM, Parkinson A, O’Sullivan D, Rosen CF, Schwartzman S, Siegel EL, Toloza S, Tuong W, Ritchlin CT (2016) Group for research and assessment of psoriasis and psoriatic arthritis 2015 treatment recommendations for psoriatic arthritis. Arthritis Rheumatol Hoboken NJ 68:1060–1071. https://doi.org/10.1002/art.39573
Gossec L, Coates LC, de Wit M, Kavanaugh A, Ramiro S, Mease PJ, Ritchlin CT, van der Heijde D, Smolen JS (2016) Management of psoriatic arthritis in 2016: a comparison of EULAR and GRAPPA recommendations. Nat Rev Rheumatol 12(12):743–750. https://doi.org/10.1038/nrrheum.2016.183
Alunno A, Carubbi F, Cafaro G, Pucci G, Battista F, Bartoloni E, Giacomelli R, Schillaci G, Gerli R (2015) Targeting the IL-23/IL-17 axis for the treatment of psoriasis and psoriatic arthritis. Expert Opin Biol Ther 15(12):1727–1737. https://doi.org/10.1517/14712598.2015.1084284
Acosta Felquer ML, Coates LC, Soriano ER, Ranza R, Espinoza LR, Helliwell PS, FitzGerald O, McHugh N, Roussou E, Mease PJ (2014) Drug therapies for peripheral joint disease in psoriatic arthritis: a systematic review. J Rheumatol 41(11):2277–2285. https://doi.org/10.3899/jrheum.140876
Langley RG, Elewski BE, Lebwohl M, Reich K, Griffiths CE, Papp K, Puig L, Nakagawa H, Spelman L, Sigurgeirsson B, Rivas E, Tsai TF, Wasel N, Tyring S, Salko T, Hampele I, Notter M, Karpov A, Helou S, Papavassilis C, ERASURE Study Group, FIXTURE Study Group (2014) Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med 371(4):326–338. https://doi.org/10.1056/NEJMoa1314258
Pariser D, Frankel E, Schlessinger J, Poulin Y, Vender R, Langley RG, Meng X, Guana A, Nyirady J (2017) Efficacy of secukinumab in the treatment of moderate to severe plaque psoriasis in the north American subgroup of patients: pooled analysis of four phase 3 studies. Dermatol Ther. https://doi.org/10.1007/s13555-017-0211-4
Paul C, Lacour J-P, Tedremets L, Kreutzer K, Jazayeri S, Adams S, Guindon C, You R, Papavassilis C, the JUNCTURE study group (2015) Efficacy, safety and usability of secukinumab administration by autoinjector/pen in psoriasis: a randomized, controlled trial (JUNCTURE). J Eur Acad Dermatol Venereol JEADV 29(6):1082–1090. https://doi.org/10.1111/jdv.12751
Griffiths CEM (2010) Comparing biological therapies in psoriasis: implications for clinical practice. J Eur Acad Dermatol Venereol JEADV 24(Suppl 6):10–14. https://doi.org/10.1111/j.1468-3083.2010.03831.x
Gottlieb A, Menter A, Mendelsohn A, Shen YK, Li S, Guzzo C, Fretzin S, Kunynetz R, Kavanaugh A (2009) Ustekinumab, a human interleukin 12/23 monoclonal antibody, for psoriatic arthritis: randomised, double-blind, placebo-controlled, crossover trial. Lancet Lond Engl 373(9664):633–640. https://doi.org/10.1016/S0140-6736(09)60140-9
Reich K, Pinter A, Lacour JP, Ferrandiz C, Micali G, French LE, Lomaga M, Dutronc Y, Henneges C, Wilhelm S, Hartz S, Paul C, on behalf of the IXORA-S investigators (2017) Comparison of ixekizumab with ustekinumab in moderate-to-severe psoriasis: 24-week results from IXORA-S, a phase III study. Br J Dermatol 177(4):1014–1023. https://doi.org/10.1111/bjd.15666
McInnes IB, Mease PJ, Kirkham B et al (2015) Secukinumab, a human anti-interleukin-17A monoclonal antibody, in patients with psoriatic arthritis (FUTURE 2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Lond Engl 386(9999):1137–1146. https://doi.org/10.1016/S0140-6736(15)61134-5
McInnes IB, Mease PJ, Ritchlin CT et al (2017) Secukinumab sustains improvement in signs and symptoms of psoriatic arthritis: 2 year results from the phase 3 FUTURE 2 study. Rheumatol Oxf Engl 56(11):1993–2003. https://doi.org/10.1093/rheumatology/kex301
Khmaladze I, Kelkka T, Guerard S, Wing K, Pizzolla A, Saxena A, Lundqvist K, Holmdahl M, Nandakumar KS, Holmdahl R (2014) Mannan induces ROS-regulated, IL-17A-dependent psoriasis arthritis-like disease in mice. Proc Natl Acad Sci U S A 111(35):E3669–E3678. https://doi.org/10.1073/pnas.1405798111
Sherlock JP, Joyce-Shaikh B, Turner SP, Chao CC, Sathe M, Grein J, Gorman DM, Bowman EP, McClanahan TK, Yearley JH, Eberl G, Buckley CD, Kastelein RA, Pierce RH, LaFace DM, Cua DJ (2012) IL-23 induces spondyloarthropathy by acting on ROR-γt+ CD3+CD4-CD8- entheseal resident T cells. Nat Med 18(7):1069–1076. https://doi.org/10.1038/nm.2817
Ruutu M, Thomas G, Steck R, Degli-Esposti MA, Zinkernagel MS, Alexander K, Velasco J, Strutton G, Tran A, Benham H, Rehaume L, Wilson RJ, Kikly K, Davies J, Pettit AR, Brown MA, McGuckin MA, Thomas R (2012) β-glucan triggers spondylarthritis and Crohn’s disease-like ileitis in SKG mice. Arthritis Rheum 64(7):2211–2222. https://doi.org/10.1002/art.34423
van der Fits L, Mourits S, Voerman JSA, Kant M, Boon L, Laman JD, Cornelissen F, Mus AM, Florencia E, Prens EP, Lubberts E (2009) Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol Baltim Md 1950 182(9):5836–5845. https://doi.org/10.4049/jimmunol.0802999
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The manuscript does not contain clinical studies or patient data.
Conflict of interest
IBM has received honoraria or research funding from Novartis, Lilly, Celgene, Janssen, Abbvie, BMS, UCB and Pfizer all of whom are developing or have medicines involved in the treatment of psoriatic arthritis.
Rights and permissions
About this article
Cite this article
Cafaro, G., McInnes, I.B. Psoriatic arthritis: tissue-directed inflammation?. Clin Rheumatol 37, 859–868 (2018). https://doi.org/10.1007/s10067-018-4012-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-018-4012-7