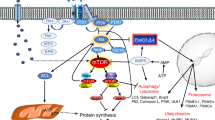
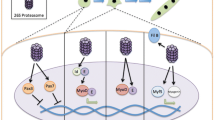
Abstract
Muscle atrophy typically is a direct effect of protein degradation induced by a diversity of pathophysiologic states such as disuse, immobilization, denervation, aging, sepsis, cachexia, glucocorticoid treatment, hereditary muscular disorders, cancer, diabetes and obesity, kidney and heart failure, and others. Muscle atrophy is defined by changes in the muscles, consisting in shrinkage of myofibers, changes in the types of fiber and myosin isoforms, and a net loss of cytoplasm, organelles and overall a protein loss. Although in the literature there are extensive studies in a range of animal models, the paucity of human data is a reality. This chapter is focused on various aspects of muscle wasting and describes the transitions of myofiber types during the progression of muscle atrophy in several pathological states. Clinical conditions associated with muscle atrophy have been grouped based on the fast-to-slow or slow-to-fast fiber-type shifts. We have also summarized the ultrastructural and histochemical features characteristic for muscle atrophy in clinical and experimental models for aging, cancer, diabetes and obesity, and heart failure and arrhythmia.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Simon L, Jolley SE, Molina PE (2017) Alcoholic myopathy: pathophysiologic mechanisms and clinical implications. Alcohol Res 38:207–217
Kneppers AEM, Langen RCJ, Gosker HR, Verdijk LB, Cebron LN, Leermakers PA, Kelders MCJM, de Theije CC, Omersa D, Lainscak M, Schols AMWJ (2017) Increased myogenic and protein turnover signaling in skeletal muscle of chronic obstructive pulmonary disease patients with sarcopenia. J Am Med Dir Assoc 18:637
de Oliveira SA, Dutra MT, de Moraes WMAM, Funghetto SS, de FD L, Dos Santos PHF, Vieira DCL, Nascimento DDC, Orsano VSM, Schoenfeld BJ, Prestes J (2018) Resistance training-induced gains in muscle strength, body composition, and functional capacity are attenuated in elderly women with sarcopenic obesity. Clin Interv Aging 13:411–417
Vellas B, Fielding R, Bhasin S, Cerreta F, Goodpaster B, Guralnik JM, Kritchevsky S, Legrand V, Forkin C, Magaziner J, Morley JE, Rodriguez-Manas L, Roubenoff R, Studenski S, Villareal DT, Cesari M (2016) Sarcopenia trials in specific diseases: report by the international conference on Frailty and Sarcopenia research task force. J Frailty Aging 5:194–200
Sedano MJ, Canga A, de PC, Polo JM, Berciano J (2013) Muscle MRI in severe Guillain-Barre syndrome with motor nerve inexcitability. J Neurol 260:1624–1630
Bowerman M, Murray LM, Scamps F, Schneider BL, Kothary R, Raoul C (2017) Pathogenic commonalities between spinal muscular atrophy and amyotrophic lateral sclerosis: converging roads to therapeutic development. Eur J Med Genet. https://doi.org/10.1016/j.ejmg.2017.12.001
Tosolini AP, Sleigh JN (2017) Motor neuron gene therapy: lessons from spinal muscular atrophy for amyotrophic lateral sclerosis. Front Mol Neurosci 10:405
Berridge BR, Van Vleet JF, Herman E (2013) Cardiac, vascular, and skeletal muscle systems. In: Haschek WM, Rousseaux CG, Wallig MA, Bolon B, Ochoa R (eds) Haschek and Rousseaux’s handbook of toxicologic pathology. Elsevier, Amsterdam
Dubowitz D, Sewry CA, Oldfors A (2013) Muscle biopsy: a practical approach. Elsevier, Amsterdam
Pernus F, Erzen I (1994) Fibre size, atrophy, and hypertrophy factors in vastus lateralis muscle from 18- to 29-year-old men. J Neurol Sci 121:194–202
Tajsharghi H, Hilton-Jones D, Raheem O, Saukkonen AM, Oldfors A, Udd B (2010) Human disease caused by loss of fast IIa myosin heavy chain due to recessive MYH2 mutations. Brain 133:1451–1459
Mastaglia FL, Walton JN (1971) Histological and histochemical changes in skeletal muscle from cases of chronic juvenile and early adult spinal muscular atrophy (the Kugelberg-Welander syndrome). J Neurol Sci 12:15–44
Yoshihara K, Shirai Y, Nakayama Y, Uesaka S (2001) Histochemical changes in the multifidus muscle in patients with lumbar intervertebral disc herniation. Spine (Phila Pa 1976) 26:622–626
Snow LM, McLoon LK, Thompson LV (2005) Adult and developmental myosin heavy chain isoforms in soleus muscle of aging Fischer Brown Norway rat. Anat Rec A Discov Mol Cell Evol Biol 286:866–873
Vihola A, Bassez G, Meola G, Zhang S, Haapasalo H, Paetau A, Mancinelli E, Rouche A, Hogrel JY, Laforet P, Maisonobe T, Pellissier JF, Krahe R, Eymard B, Udd B (2003) Histopathological differences of myotonic dystrophy type 1 (DM1) and PROMM/DM2. Neurology 60:1854–1857
van WT, de HA, van der Laarse WJ, Jaspers RT (2010) The muscle fiber type-fiber size paradox: hypertrophy or oxidative metabolism? Eur J Appl Physiol 110:665–694
Ciciliot S, Rossi AC, Dyar KA, Blaauw B, Schiaffino S (2013) Muscle type and fiber type specificity in muscle wasting. Int J Biochem Cell Biol 45:2191–2199
Grimby G, Broberg C, Krotkiewska I, Krotkiewski M (1976) Muscle fiber composition in patients with traumatic cord lesion. Scand J Rehabil Med 8:37–42
Herbison GJ, Jaweed MM, Ditunno JF (1979) Muscle atrophy in rats following denervation, casting, inflammation, and tenotomy. Arch Phys Med Rehabil 60:401–404
Ohira Y, Jiang B, Roy RR, Oganov V, Ilyina-Kakueva E, Marini JF, Edgerton VR (1992) Rat soleus muscle fiber responses to 14 days of spaceflight and hindlimb suspension. J Appl Physiol (1985) 73:51S–57S
Campione M, Ausoni S, Guezennec CY, Schiaffino S (1993) Myosin and troponin changes in rat soleus muscle after hindlimb suspension. J Appl Physiol (1985) 74:1156–1160
Sullivan MJ, Duscha BD, Klitgaard H, Kraus WE, Cobb FR, Saltin B (1997) Altered expression of myosin heavy chain in human skeletal muscle in chronic heart failure. Med Sci Sports Exerc 29:860–866
Satta A, Migliori GB, Spanevello A, Neri M, Bottinelli R, Canepari M, Pellegrino MA, Reggiani C (1997) Fibre types in skeletal muscles of chronic obstructive pulmonary disease patients related to respiratory function and exercise tolerance. Eur Respir J 10:2853–2860
Goldberg AL, Goodman HM (1969) Relationship between cortisone and muscle work in determining muscle size. J Physiol 200:667–675
Mendell JR, Engel WK (1971) The fine structure of type II muscle fiber atrophy. Neurology 21:358–365
Armstrong RB, Gollnick PD, Ianuzzo CD (1975) Histochemical properties of skeletal muscle fibers in streptozotocin-diabetic rats. Cell Tissue Res 162:387–394
Li JB, Goldberg AL (1976) Effects of food deprivation on protein synthesis and degradation in rat skeletal muscles. Am J Phys 231:441–448
Holloszy JO, Chen M, Cartee GD, Young JC (1991) Skeletal muscle atrophy in old rats: differential changes in the three fiber types. Mech Ageing Dev 60:199–213
Larsson L, Biral D, Campione M, Schiaffino S (1993) An age-related type IIB to IIX myosin heavy chain switching in rat skeletal muscle. Acta Physiol Scand 147:227–234
Lexell J (1995) Human aging, muscle mass, and fiber type composition. J Gerontol A Biol Sci Med Sci 50:11–16
Tiao G, Lieberman M, Fischer JE, Hasselgren PO (1997) Intracellular regulation of protein degradation during sepsis is different in fast- and slow-twitch muscle. Am J Phys 272:R849–R856
Levine S, Kaiser L, Leferovich J, Tikunov B (1997) Cellular adaptations in the diaphragm in chronic obstructive pulmonary disease. N Engl J Med 337:1799–1806
Tikunov B, Levine S, Mancini D (1997) Chronic congestive heart failure elicits adaptations of endurance exercise in diaphragmatic muscle. Circulation 95:910–916
Mercadier JJ, Schwartz K, Schiaffino S, Wisnewsky C, Ausoni S, Heimburger M, Marrash R, Pariente R, Aubier M (1998) Myosin heavy chain gene expression changes in the diaphragm of patients with chronic lung hyperinflation. Am J Phys 274:L527–L534
Acharyya S, Butchbach ME, Sahenk Z, Wang H, Saji M, Carathers M, Ringel MD, Skipworth RJ, Fearon KC, Hollingsworth MA, Muscarella P, Burghes AH, Rafael-Fortney JA, Guttridge DC (2005) Dystrophin glycoprotein complex dysfunction: a regulatory link between muscular dystrophy and cancer cachexia. Cancer Cell 8:421–432
Serrano AL, Jardi M, Suelves M, Klotman PE, Munoz-Canoves P (2008) HIV-1 transgenic expression in mice induces selective atrophy of fast-glycolytic skeletal muscle fibers. Front Biosci 13:2797–2805
Gannon J, Doran P, Kirwan A, Ohlendieck K (2009) Drastic increase of myosin light chain MLC-2 in senescent skeletal muscle indicates fast-to-slow fibre transition in sarcopenia of old age. Eur J Cell Biol 88:685–700
Hering T, Braubach P, Landwehrmeyer GB, Lindenberg KS, Melzer W (2016) Fast-to-slow transition of skeletal muscle contractile function and corresponding changes in Myosin heavy and light chain formation in the R6/2 mouse model of Huntington’s disease. PLoS One 11:e0166106
Wang Y, Pessin JE (2013) Mechanisms for fiber-type specificity of skeletal muscle atrophy. Curr Opin Clin Nutr Metab Care 16:243–250
Sandona D, Desaphy JF, Camerino GM, Bianchini E, Ciciliot S, Danieli-Betto D, Dobrowolny G, Furlan S, Germinario E, Goto K, Gutsmann M, Kawano F, Nakai N, Ohira T, Ohno Y, Picard A, Salanova M, Schiffl G, Blottner D, Musaro A, Ohira Y, Betto R, Conte D, Schiaffino S (2012) Adaptation of mouse skeletal muscle to long-term microgravity in the MDS mission. PLoS One 7:e33232
Furuno K, Goodman MN, Goldberg AL (1990) Role of different proteolytic systems in the degradation of muscle proteins during denervation atrophy. J Biol Chem 265:8550–8557
Wing SS, Goldberg AL (1993) Glucocorticoids activate the ATP-ubiquitin-dependent proteolytic system in skeletal muscle during fasting. Am J Phys 264:E668–E676
Temparis S, Asensi M, Taillandier D, Aurousseau E, Larbaud D, Obled A, Bechet D, Ferrara M, Estrela JM, Attaix D (1994) Increased ATP-ubiquitin-dependent proteolysis in skeletal muscles of tumor-bearing rats. Cancer Res 54:5568–5573
Wing SS, Banville D (1994) 14-kDa ubiquitin-conjugating enzyme: structure of the rat gene and regulation upon fasting and by insulin. Am J Phys 267:E39–E48
Chrysis D, Underwood LE (1999) Regulation of components of the ubiquitin system by insulin-like growth factor I and growth hormone in skeletal muscle of rats made catabolic with dexamethasone. Endocrinology 140:5635–5641
Lecker SH, Solomon V, Price SR, Kwon YT, Mitch WE, Goldberg AL (1999) Ubiquitin conjugation by the N-end rule pathway and mRNAs for its components increase in muscles of diabetic rats. J Clin Invest 104:1411–1420
Fischer D, Sun X, Gang G, Pritts T, Hasselgren PO (2000) The gene expression of ubiquitin ligase E3alpha is upregulated in skeletal muscle during sepsis in rats-potential role of glucocorticoids. Biochem Biophys Res Commun 267:504–508
Lorite MJ, Smith HJ, Arnold JA, Morris A, Thompson MG, Tisdale MJ (2001) Activation of ATP-ubiquitin-dependent proteolysis in skeletal muscle in vivo and murine myoblasts in vitro by a proteolysis-inducing factor (PIF). Br J Cancer 85:297–302
Bodine SC, Latres E, Baumhueter S, Lai VK, Nunez L, Clarke BA, Poueymirou WT, Panaro FJ, Na E, Dharmarajan K, Pan ZQ, Valenzuela DM, DeChiara TM, Stitt TN, Yancopoulos GD, Glass DJ (2001) Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 294:1704–1708
Gomes MD, Lecker SH, Jagoe RT, Navon A, Goldberg AL (2001) Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy. Proc Natl Acad Sci U S A 98:14440–14445
Kwon YT, Xia Z, Davydov IV, Lecker SH, Varshavsky A (2001) Construction and analysis of mouse strains lacking the ubiquitin ligase UBR1 (E3alpha) of the N-end rule pathway. Mol Cell Biol 21:8007–8021
Li YP, Lecker SH, Chen Y, Waddell ID, Goldberg AL, Reid MB (2003) TNF-alpha increases ubiquitin-conjugating activity in skeletal muscle by up-regulating UbcH2/E220k. FASEB J 17:1048–1057
Kwak KS, Zhou X, Solomon V, Baracos VE, Davis J, Bannon AW, Boyle WJ, Lacey DL, Han HQ (2004) Regulation of protein catabolism by muscle-specific and cytokine-inducible ubiquitin ligase E3alpha-II during cancer cachexia. Cancer Res 64:8193–8198
Lecker SH, Jagoe RT, Gilbert A, Gomes M, Baracos V, Bailey J, Price SR, Mitch WE, Goldberg AL (2004) Multiple types of skeletal muscle atrophy involve a common program of changes in gene expression. FASEB J 18:39–51
Lecker SH, Goldberg AL, Mitch WE (2006) Protein degradation by the ubiquitin-proteasome pathway in normal and disease states. J Am Soc Nephrol 17:1807–1819
Sacheck JM, Hyatt JP, Raffaello A, Jagoe RT, Roy RR, Edgerton VR, Lecker SH, Goldberg AL (2007) Rapid disuse and denervation atrophy involve transcriptional changes similar to those of muscle wasting during systemic diseases. FASEB J 21:140–155
Nakao R, Hirasaka K, Goto J, Ishidoh K, Yamada C, Ohno A, Okumura Y, Nonaka I, Yasutomo K, Baldwin KM, Kominami E, Higashibata A, Nagano K, Tanaka K, Yasui N, Mills EM, Takeda S, Nikawa T (2009) Ubiquitin ligase Cbl-b is a negative regulator for insulin-like growth factor 1 signaling during muscle atrophy caused by unloading. Mol Cell Biol 9:4798–4811
Paul PK, Gupta SK, Bhatnagar S, Panguluri SK, Darnay BG, Choi Y, Kumar A (2010) Targeted ablation of TRAF6 inhibits skeletal muscle wasting in mice. J Cell Biol 191:1395–1411
Baehr LM, Furlow JD, Bodine SC (2011) Muscle sparing in muscle RING finger 1 null mice: response to synthetic glucocorticoids. J Physiol 589:4759–4776
Raben N, Hill V, Shea L, Takikita S, Baum R, Mizushima N, Ralston E, Plotz P (2008) Suppression of autophagy in skeletal muscle uncovers the accumulation of ubiquitinated proteins and their potential role in muscle damage in Pompe disease. Hum Mol Genet 17:3897–3908
Moresi V, Williams AH, Meadows E, Flynn JM, Potthoff MJ, McAnally J, Shelton JM, Backs J, Klein WH, Richardson JA, Bassel-Duby R, Olson EN (2010) Myogenin and class II HDACs control neurogenic muscle atrophy by inducing E3 ubiquitin ligases. Cell 143:35–45
Moresi V, Carrer M, Grueter CE, Rifki OF, Shelton JM, Richardson JA, Bassel-Duby R, Olson EN (2012) Histone deacetylases 1 and 2 regulate autophagy flux and skeletal muscle homeostasis in mice. Proc Natl Acad Sci U S A 109:1649–1654
Sandri M (2013) Protein breakdown in muscle wasting: role of autophagy-lysosome and ubiquitin-proteasome. Int J Biochem Cell Biol 45:2121–2129
Milan G, Romanello V, Pescatore F, Armani A, Paik JH, Frasson L, Seydel A, Zhao J, Abraham R, Goldberg AL, Blaauw B, DePinho RA, Sandri M (2015) Regulation of autophagy and the ubiquitin-proteasome system by the FoxO transcriptional network during muscle atrophy. Nat Commun 6:6670
Bothe GW, Haspel JA, Smith CL, Wiener HH, Burden SJ (2000) Selective expression of Cre recombinase in skeletal muscle fibers. Genesis 26:165–166
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R, Yokoyama M, Mishima K, Saito I, Okano H, Mizushima N (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885–889
Bonaldo P, Sandri M (2013) Cellular and molecular mechanisms of muscle atrophy. Dis Model Mech 6:25–39
Bae SK, Cha HN, Ju TJ, Kim YW, Kim HS, Kim YD, Dan JM, Kim JY, Kim SD, Park SY (2012) Deficiency of inducible nitric oxide synthase attenuates immobilization-induced skeletal muscle atrophy in mice. J Appl Physiol (1985) 113:114–123
Reed SA, Sandesara PB, Senf SM, Judge AR (2012) Inhibition of FoxO transcriptional activity prevents muscle fiber atrophy during cachexia and induces hypertrophy. FASEB J 26:987–1000
Selsby JT, Morine KJ, Pendrak K, Barton ER, Sweeney HL (2012) Rescue of dystrophic skeletal muscle by PGC-1alpha involves a fast to slow fiber type shift in the mdx mouse. PLoS One 7:e30063
Vogel H, Zamecnik J (2005) Diagnostic immunohistology of muscle diseases. J Neuropathol Exp Neurol 64:181–193
Lee WS, Cheung WH, Qin L, Tang N, Leung KS (2006) Age-associated decrease of type IIA/B human skeletal muscle fibers. Clin Orthop Relat Res 450:231–237
Frontera WR, Hughes VA, Fielding RA, Fiatarone MA, Evans WJ, Roubenoff R (2000) Aging of skeletal muscle: a 12-yr longitudinal study. J Appl Physiol (1985) 88:1321–1326
Visser M, Kritchevsky SB, Goodpaster BH, Newman AB, Nevitt M, Stamm E, Harris TB (2002) Leg muscle mass and composition in relation to lower extremity performance in men and women aged 70 to 79: the health, aging and body composition study. J Am Geriatr Soc 50:897–904
Hvid LG, Suetta C, Nielsen JH, Jensen MM, Frandsen U, Ortenblad N, Kjaer M, Aagaard P (2014) Aging impairs the recovery in mechanical muscle function following 4 days of disuse. Exp Gerontol 52:1–8
Rolland Y, Van Abellan KG, Gillette-Guyonnet S, Vellas B (2011) Cachexia versus sarcopenia. Curr Opin Clin Nutr Metab Care 14:15–21
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinkova E, Vandewoude M, Zamboni M (2010) Sarcopenia: European consensus on definition and diagnosis: report of the European working group on Sarcopenia in older people. Age Ageing 39:412–423
Tinetti ME, Williams CS (1997) Falls, injuries due to falls, and the risk of admission to a nursing home. N Engl J Med 337:1279–1284
Lexell J, Taylor CC, Sjostrom M (1988) What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J Neurol Sci 84:275–294
Volpi E, Sheffield-Moore M, Rasmussen BB, Wolfe RR (2001) Basal muscle amino acid kinetics and protein synthesis in healthy young and older men. JAMA 286:1206–1212
Linnane AW, Marzuki S, Ozawa T, Tanaka M (1989) Mitochondrial DNA mutations as an important contributor to ageing and degenerative diseases. Lancet 1:642–645
Muller-Hocker J (1992) Mitochondria and ageing. Brain Pathol 2:149–158
Fayet G, Jansson M, Sternberg D, Moslemi AR, Blondy P, Lombes A, Fardeau M, Oldfors A (2002) Ageing muscle: clonal expansions of mitochondrial DNA point mutations and deletions cause focal impairment of mitochondrial function. Neuromuscul Disord 12:484–493
Greaves LC, Turnbull DM (2009) Mitochondrial DNA mutations and ageing. Biochim Biophys Acta 1790:1015–1020
Marzetti E, Hwang JC, Lees HA, Wohlgemuth SE, Dupont-Versteegden EE, Carter CS, Bernabei R, Leeuwenburgh C (2010) Mitochondrial death effectors: relevance to sarcopenia and disuse muscle atrophy. Biochim Biophys Acta 1800:235–244
Jackson MJ, McArdle A (2011) Age-related changes in skeletal muscle reactive oxygen species generation and adaptive responses to reactive oxygen species. J Physiol 589:2139–2145
Deschenes MR, Gaertner JR, O’Reilly S (2013) The effects of sarcopenia on muscles with different recruitment patterns and myofiber profiles. Curr Aging Sci 6:266–272
Park SC, Kim WH, Lee MC, Seong SC, Song KY, Choe MA (1994) Modulation of transglutaminase expression in rat skeletal muscle by induction of atrophy and endurance training. J Korean Med Sci 9:490–496
Connor NP, Suzuki T, Lee K, Sewall GK, Heisey DM (2002) Neuromuscular junction changes in aged rat thyroarytenoid muscle. Ann Otol Rhinol Laryngol 111:579–586
Delmonico MJ, Harris TB, Visser M, Park SW, Conroy MB, Velasquez-Mieyer P, Boudreau R, Manini TM, Nevitt M, Newman AB, Goodpaster BH (2009) Longitudinal study of muscle strength, quality, and adipose tissue infiltration. Am J Clin Nutr 90:1579–1585
Wronska A, Kmiec Z (2012) Structural and biochemical characteristics of various white adipose tissue depots. Acta Physiol (Oxford) 205:194–208
Raguso CA, Kyle U, Kossovsky MP, Roynette C, Paoloni-Giacobino A, Hans D, Genton L, Pichard C (2006) A 3-year longitudinal study on body composition changes in the elderly: role of physical exercise. Clin Nutr 25:573–580
Perry BD, Caldow MK, Brennan-Speranza TC, Sbaraglia M, Jerums G, Garnham A, Wong C, Levinger P, Asrar Ul HM, Hare DL, Price SR, Levinger I (2016) Muscle atrophy in patients with type 2 Diabetes Mellitus: roles of inflammatory pathways, physical activity and exercise. Exerc Immunol Rev 22:94–109
Patsouris D, Cao JJ, Vial G, Bravard A, Lefai E, Durand A, Durand C, Chauvin MA, Laugerette F, Debard C, Michalski MC, Laville M, Vidal H, Rieusset J (2014) Insulin resistance is associated with MCP1-mediated macrophage accumulation in skeletal muscle in mice and humans. PLoS One 9:e110653
Mashili F, Chibalin AV, Krook A, Zierath JR (2013) Constitutive STAT3 phosphorylation contributes to skeletal muscle insulin resistance in type 2 diabetes. Diabetes 62:457–465
Zhang L, Pan J, Dong Y, Tweardy DJ, Dong Y, Garibotto G, Mitch WE (2013) Stat3 activation links a C/EBPdelta to myostatin pathway to stimulate loss of muscle mass. Cell Metab 18:368–379
Chiu CY, Yang RS, Sheu ML, Chan DC, Yang TH, Tsai KS, Chiang CK, Liu SH (2016) Advanced glycation end-products induce skeletal muscle atrophy and dysfunction in diabetic mice via a RAGE-mediated, AMPK-down-regulated, Akt pathway. J Pathol 238:470–482
Hittel DS, Hathout Y, Hoffman EP, Houmard JA (2005) Proteome analysis of skeletal muscle from obese and morbidly obese women. Diabetes 54:1283–1288
Wijers SL, Smit E, Saris WH, Mariman EC, van Marken Lichtenbelt WD (2010) Cold- and overfeeding-induced changes in the human skeletal muscle proteome. J Proteome Res 9:2226–2235
Andreassen CS, Jensen JM, Jakobsen J, Ulhoj BP, Andersen H (2014) Striated muscle fiber size, composition, and capillary density in diabetes in relation to neuropathy and muscle strength. J Diabetes 6:462–471
Leenders M, Verdijk LB, van der Hoeven L, Adam JJ, VAN KJ, Nilwik R, van Loon LJ (2013) Patients with type 2 diabetes show a greater decline in muscle mass, muscle strength, and functional capacity with aging. J Am Med Dir Assoc 14:585–592
Jerkovic R, Bosnar A, Jurisic-Erzen D, Azman J, Starcevic-Klasan G, Peharec S, Coklo M (2009) The effects of long-term experimental diabetes mellitus type I on skeletal muscle regeneration capacity. Coll Antropol 33:1115–1119
Xiang J, Zhao Y, Chen J, Zhou J (2014) Expression of basic fibroblast growth factor, protein kinase C and members of the apoptotic pathway in skeletal muscle of streptozotocin-induced diabetic rats. Tissue Cell 46:1–8
Medina-Sanchez M, Rodriguez-Sanchez C, Vega-Alvarez JA, Menedez-Pelaez A, Perez-Casas A (1991) Proximal skeletal muscle alterations in streptozotocin-diabetic rats: a histochemical and morphometric analysis. Am J Anat 191:48–56
Klueber KM, Feczko JD (1994) Ultrastructural, histochemical, and morphometric analysis of skeletal muscle in a murine model of type I diabetes. Anat Rec 239:18–34
Chen GQ, Mou CY, Yang YQ, Wang S, Zhao ZW (2011) Exercise training has beneficial anti-atrophy effects by inhibiting oxidative stress-induced MuRF1 upregulation in rats with diabetes. Life Sci 89:44–49
Johns N, Stephens NA, Fearon KC (2013) Muscle wasting in cancer. Int J Biochem Cell Biol 45:2215–2229
Darnell RB, Posner JB (2003) Paraneoplastic syndromes involving the nervous system. N Engl J Med 349:1543–1554
Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, Jatoi A, Loprinzi C, MacDonald N, Mantovani G, Davis M, Muscaritoli M, Ottery F, Radbruch L, Ravasco P, Walsh D, Wilcock A, Kaasa S, Baracos VE (2011) Definition and classification of cancer cachexia: an international consensus. Lancet Oncol 12:489–495
Weber MA, Krakowski-Roosen H, Schroder L, Kinscherf R, Krix M, Kopp-Schneider A, Essig M, Bachert P, Kauczor HU, Hildebrandt W (2009) Morphology, metabolism, microcirculation, and strength of skeletal muscles in cancer-related cachexia. Acta Oncol 48:116–124
Eley HL, Skipworth RJ, Deans DA, Fearon KC, Tisdale MJ (2008) Increased expression of phosphorylated forms of RNA-dependent protein kinase and eukaryotic initiation factor 2alpha may signal skeletal muscle atrophy in weight-losing cancer patients. Br J Cancer 98:443–449
Zampieri S, Doria A, Adami N, Biral D, Vecchiato M, Savastano S, Corbianco S, Carraro U, Merigliano S (2010) Subclinical myopathy in patients affected with newly diagnosed colorectal cancer at clinical onset of disease: evidence from skeletal muscle biopsies. Neurol Res 32:20–25
So MW, Koo BS, Kim YG, Lee CK, Yoo B (2011) Idiopathic inflammatory myopathy associated with malignancy: a retrospective cohort of 151 Korean patients with dermatomyositis and polymyositis. J Rheumatol 38:2432–2435
Silvestre J, Santos L, Batalha V, Del RA, Lima C, Carvalho A, Martins A, Miranda H, Cabral F, Felix A, Aleixo A (2009) Paraneoplastic necrotizing myopathy in a woman with breast cancer: a case report. J Med Case Rep 3:95
Dalakas MC (2011) Inflammatory myopathies: management of steroid resistance. Curr Opin Neurol 24:457–462
De BJ, Vervaet V, Van den Bergh P (2004) Necrotizing myopathy with microvascular deposition of the complement membrane attack complex. Clin Neuropathol 23:76–79
Penna F, Costamagna D, Pin F, Camperi A, Fanzani A, Chiarpotto EM, Cavallini G, Bonelli G, Baccino FM, Costelli P (2013) Autophagic degradation contributes to muscle wasting in cancer cachexia. Am J Pathol 182:1367–1378
Holecek M (2012) Muscle wasting in animal models of severe illness. Int J Exp Pathol 93:157–171
Zhou X, Wang JL, Lu J, Song Y, Kwak KS, Jiao Q, Rosenfeld R, Chen Q, Boone T, Simonet WS, Lacey DL, Goldberg AL, Han HQ (2010) Reversal of cancer cachexia and muscle wasting by ActRIIB antagonism leads to prolonged survival. Cell 142:531–543
Kumar A, Bhatnagar S, Paul PK (2012) TWEAK and TRAF6 regulate skeletal muscle atrophy. Curr Opin Clin Nutr Metab Care 15:233–239
Paul PK, Kumar A (2011) TRAF6 coordinates the activation of autophagy and ubiquitin-proteasome systems in atrophying skeletal muscle. Autophagy 7:555–556
Kinugawa S, Takada S, Matsushima S, Okita K, Tsutsui H (2015) Skeletal muscle abnormalities in heart failure. Int Heart J 56:475–484
Carvalho RF, Castan EP, Coelho CA, Lopes FS, Almeida FL, Michelin A, de Souza RW, Araujo JP Jr, Cicogna AC, Dal Pai-Silva M (2010) Heart failure increases atrogin-1 and MuRF1 gene expression in skeletal muscle with fiber type-specific atrophy. J Mol Histol 41:81–87
Manfredi LH, Paula-Gomes S, Zanon NM, Kettelhut IC (2017) Myostatin promotes distinct responses on protein metabolism of skeletal and cardiac muscle fibers of rodents. Braz J Med Biol Res 50:e6733
Bowen TS, Rolim NP, Fischer T, Baekkerud FH, Medeiros A, Werner S, Bronstad E, Rognmo O, Mangner N, Linke A, Schuler G, Silva GJ, Wisloff U, Adams V (2015) Heart failure with preserved ejection fraction induces molecular, mitochondrial, histological, and functional alterations in rat respiratory and limb skeletal muscle. Eur J Heart Fail 17:263–272
Damatto RL, Martinez PF, Lima AR, Cezar MD, Campos DH, Oliveira Junior SA, Guizoni DM, Bonomo C, Nakatani BT, Dal Pai SM, Carvalho RF, Okoshi K, Okoshi MP (2013) Heart failure-induced skeletal myopathy in spontaneously hypertensive rats. Int J Cardiol 167:698–703
Vescovo G, Dalla LL (2006) Skeletal muscle apoptosis in experimental heart failure: the only link between inflammation and skeletal muscle wastage? Curr Opin Clin Nutr Metab Care 9:416–422
Deswal A, Petersen NJ, Feldman AM, Young JB, White BG, Mann DL (2001) Cytokines and cytokine receptors in advanced heart failure: an analysis of the cytokine database from the Vesnarinone trial (VEST). Circulation 103:2055–2059
Dalla LL, Vescovo G, Volterrani M (2008) Physiological basis for contractile dysfunction in heart failure. Curr Pharm Des 14:2572–2581
Isner JM, Hawley RJ, Weintraub AM, Engel WK (1979) Cardiac findings in Charcot-Marie-Tooth disease. A prospective study of 68 patients. Arch Intern Med 139:1161–1165
Araki A, Katsuno M, Suzuki K, Banno H, Suga N, Hashizume A, Mano T, Hijikata Y, Nakatsuji H, Watanabe H, Yamamoto M, Makiyama T, Ohno S, Fukuyama M, Morimoto S, Horie M, Sobue G (2014) Brugada syndrome in spinal and bulbar muscular atrophy. Neurology 82:1813–1821
Finsterer J, Stollberger C (2018) Only some patients with bulbar and spinal muscular atrophy may develop cardiac disease. Mol Genet Metab Rep 14:44–46
Sanguinetti MC (2010) HERG1 channelopathies. Pflugers Arch 460:265–276
Molenaar P, Chen L, Parsonage WA (2006) Cardiac implications for the use of beta2-adrenoceptor agonists for the management of muscle wasting. Br J Pharmacol 147:583–586
Acknowledgments
BM Radu is currently funded from Competitiveness Operational Programme 2014–2020 project P_37_675 (contract no. 146/2016), Priority Axis 1, Action 1.1.4, co-financed by the European Funds for Regional Development and Romanian Government funds. The contents of this publication do not necessarily reflect the official position of the European Union or Romanian Government.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Dumitru, A., Radu, B.M., Radu, M., Cretoiu, S.M. (2018). Muscle Changes During Atrophy. In: Xiao, J. (eds) Muscle Atrophy. Advances in Experimental Medicine and Biology, vol 1088. Springer, Singapore. https://doi.org/10.1007/978-981-13-1435-3_4
Download citation
DOI: https://doi.org/10.1007/978-981-13-1435-3_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-1434-6
Online ISBN: 978-981-13-1435-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)