Inhibition of type 1 immunity with tofacitinib is associated with marked improvement in longstanding sarcoidosis
- PMID: 35668129
- PMCID: PMC9170782
- DOI: 10.1038/s41467-022-30615-x
Inhibition of type 1 immunity with tofacitinib is associated with marked improvement in longstanding sarcoidosis
Abstract
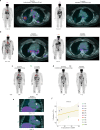
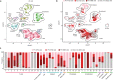
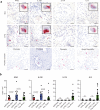
Sarcoidosis is an idiopathic inflammatory disorder that is commonly treated with glucocorticoids. An imprecise understanding of the immunologic changes underlying sarcoidosis has limited therapeutic progress. Here in this open-label trial (NCT03910543), 10 patients with cutaneous sarcoidosis are treated with tofacitinib, a Janus kinase inhibitor. The primary outcome is the change in the cutaneous sarcoidosis activity and morphology instrument (CSAMI) activity score after 6 months of treatment. Secondary outcomes included change in internal organ involvement, molecular parameters, and safety. All patients experience improvement in their skin with 6 patients showing a complete response. Improvement in internal organ involvement is also observed. CD4+ T cell-derived IFN-γ is identified as a central cytokine mediator of macrophage activation in sarcoidosis. Additional type 1 cytokines produced by distinct cell types, including IL-6, IL-12, IL-15 and GM-CSF, also associate with pathogenesis. Suppression of the activity of these cytokines, especially IFN-γ, correlates with clinical improvement. Our results thus show that tofacitinib treatment is associated with improved sarcoidosis symptoms, and predominantly acts by inhibiting type 1 immunity.
© 2022. The Author(s).
Conflict of interest statement
W.D. has research funding from Pfizer and Advanced Cell Diagnostics/Bio-techne, serves as a consultant for Eli Lilly, Pfizer, Incyte, and Twi Biotechnology, and receives licensing fees from EMD/Millipore/Sigma. B.D.Y receives research funding from Pfizer. E.J.M. receives research funding from Alnylam, Pfizer, and Eidos and serves as a consultant for Alnylam, Pfizer, and Eidos. D.P. receives consulting fees from Telix Pharmaceuticals and Cohere Health. M.B. is a consultant for Eli Lilly and receives licensing fees from EMD/Millipore//Sigma. R.A.F. is a consultant for Glaxo Smith Kline and Zai labs. B.K. is a consultant to and/or has served on advisory boards for Aclaris Therapeutics, Arena Pharmaceuticals, Bristol-Meyers Squibb, Concert Pharmaceuticals Inc, Dermavant Sciences, Eli Lilly and Company, Pfizer, and VielaBio; he is on speaker’s bureau for Pfizer, Regeneron and Sanofi Genzyme. A.W., D.J.K., K.S., M.J.M., J.D., A.C., R.A., C.R., M.K.M., I.D.O., R.F.C., R.H. and M.G. have no disclosures.
Figures






Comment in
-
Sarcoidosis: can tofacitinib slay the dragon?Nat Rev Rheumatol. 2022 Oct;18(10):557-558. doi: 10.1038/s41584-022-00832-1. Nat Rev Rheumatol. 2022. PMID: 35999390 No abstract available.
-
Tofacitinib for sarcoidosis, a new potential treatment.Int J Rheum Dis. 2022 Nov;25(11):1217-1219. doi: 10.1111/1756-185X.14441. Int J Rheum Dis. 2022. PMID: 36320145 No abstract available.
Similar articles
-
Treatment of granuloma annulare and suppression of proinflammatory cytokine activity with tofacitinib.J Allergy Clin Immunol. 2021 May;147(5):1795-1809. doi: 10.1016/j.jaci.2020.10.012. Epub 2020 Dec 11. J Allergy Clin Immunol. 2021. PMID: 33317858 Clinical Trial.
-
Janus kinase inhibition induces disease remission in cutaneous sarcoidosis and granuloma annulare.J Am Acad Dermatol. 2020 Mar;82(3):612-621. doi: 10.1016/j.jaad.2019.05.098. Epub 2019 Jun 8. J Am Acad Dermatol. 2020. PMID: 31185230 Free PMC article.
-
Tofacitinib for cutaneous and pulmonary sarcoidosis: A case series.J Am Acad Dermatol. 2021 Feb;84(2):581-583. doi: 10.1016/j.jaad.2020.10.016. Epub 2020 Oct 16. J Am Acad Dermatol. 2021. PMID: 33075414 No abstract available.
-
Successful management with Janus kinase inhibitor tofacitinib in refractory juvenile dermatomyositis: a pilot study and literature review.Rheumatology (Oxford). 2021 Apr 6;60(4):1700-1707. doi: 10.1093/rheumatology/keaa558. Rheumatology (Oxford). 2021. PMID: 33024992 Review.
-
Tofacitinib for the treatment of moderate-to-severe psoriasis.Expert Rev Clin Immunol. 2015 Apr;11(4):443-55. doi: 10.1586/1744666X.2015.1013534. Epub 2015 Feb 10. Expert Rev Clin Immunol. 2015. PMID: 25666451 Review.
Cited by
-
[Position paper of the Austrian Society for Rheumatology and the Austrian Society for Pneumology on the diagnosis and treatment of sarcoidosis 2024].Wien Klin Wochenschr. 2024 Oct;136(Suppl 17):669-687. doi: 10.1007/s00508-024-02444-z. Epub 2024 Oct 9. Wien Klin Wochenschr. 2024. PMID: 39382646 Free PMC article. German.
-
Recruitment of CXCR4+ type 1 innate lymphoid cells distinguishes sarcoidosis from other skin granulomatous diseases.J Clin Invest. 2024 Sep 3;134(17):e178711. doi: 10.1172/JCI178711. J Clin Invest. 2024. PMID: 39225100 Free PMC article.
-
Salvage use of tofacitinib in sarcoidosis patient with refractory multiorgan involvement.Sarcoidosis Vasc Diffuse Lung Dis. 2024 Jun 28;41(2):e2024019. doi: 10.36141/svdld.v41i2.15655. Sarcoidosis Vasc Diffuse Lung Dis. 2024. PMID: 38940706 Free PMC article. No abstract available.
-
Autoimmune comorbidities associated with sarcoidosis: a case-control study in the All of Us research program.Rheumatol Adv Pract. 2023 Apr 12;7(2):rkad030. doi: 10.1093/rap/rkad030. eCollection 2023. Rheumatol Adv Pract. 2023. PMID: 38606002 Free PMC article.
-
Treatment of Granulomatous Inflammation in Pulmonary Sarcoidosis.J Clin Med. 2024 Jan 27;13(3):738. doi: 10.3390/jcm13030738. J Clin Med. 2024. PMID: 38337432 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

