Regulation of ceramide biosynthesis by TOR complex 2
- PMID: 18249174
- PMCID: PMC3882310
- DOI: 10.1016/j.cmet.2007.11.015
Regulation of ceramide biosynthesis by TOR complex 2
Abstract
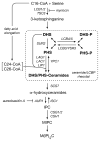
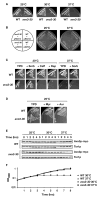
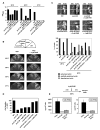
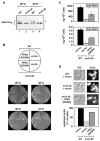
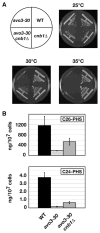
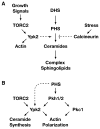
Ceramides and sphingoid long-chain bases (LCBs) are precursors to more complex sphingolipids and play distinct signaling roles crucial for cell growth and survival. Conserved reactions within the sphingolipid biosynthetic pathway are responsible for the formation of these intermediates. Components of target of rapamycin complex 2 (TORC2) have been implicated in the biosynthesis of sphingolipids in S. cerevisiae; however, the precise step regulated by this complex remains unknown. Here we demonstrate that yeast cells deficient in TORC2 activity are impaired for de novo ceramide biosynthesis both in vivo and in vitro. We find that TORC2 regulates this step in part by activating the AGC kinase Ypk2 and that this step is antagonized by the Ca2+/calmodulin-dependent phosphatase calcineurin. Because Ypk2 is activated independently by LCBs, the direct precursors to ceramides, our data suggest a model wherein TORC2 signaling is coupled with LCB levels to control Ypk2 activity and, ultimately, regulate ceramide formation.
Figures

Comment in
-
More chores for TOR: de novo ceramide synthesis.Cell Metab. 2008 Feb;7(2):99-100. doi: 10.1016/j.cmet.2008.01.002. Cell Metab. 2008. PMID: 18249168
Similar articles
-
Target of rapamycin complex 2 signals to downstream effector yeast protein kinase 2 (Ypk2) through adheres-voraciously-to-target-of-rapamycin-2 protein 1 (Avo1) in Saccharomyces cerevisiae.J Biol Chem. 2012 Feb 24;287(9):6089-99. doi: 10.1074/jbc.M111.303701. Epub 2011 Dec 28. J Biol Chem. 2012. PMID: 22207764 Free PMC article.
-
More chores for TOR: de novo ceramide synthesis.Cell Metab. 2008 Feb;7(2):99-100. doi: 10.1016/j.cmet.2008.01.002. Cell Metab. 2008. PMID: 18249168
-
Tor2 directly phosphorylates the AGC kinase Ypk2 to regulate actin polarization.Mol Cell Biol. 2005 Aug;25(16):7239-48. doi: 10.1128/MCB.25.16.7239-7248.2005. Mol Cell Biol. 2005. PMID: 16055732 Free PMC article.
-
Thematic review series: sphingolipids. New insights into sphingolipid metabolism and function in budding yeast.J Lipid Res. 2008 May;49(5):909-21. doi: 10.1194/jlr.R800003-JLR200. Epub 2008 Feb 23. J Lipid Res. 2008. PMID: 18296751 Free PMC article. Review.
-
Unraveling the role of the Target of Rapamycin signaling in sphingolipid metabolism.Prog Lipid Res. 2016 Jan;61:109-33. doi: 10.1016/j.plipres.2015.11.001. Epub 2015 Dec 17. Prog Lipid Res. 2016. PMID: 26703187 Review.
Cited by
-
mTORC2 Deficiency Alters the Metabolic Profile of Conventional Dendritic Cells.Front Immunol. 2019 Jul 2;10:1451. doi: 10.3389/fimmu.2019.01451. eCollection 2019. Front Immunol. 2019. PMID: 31338091 Free PMC article.
-
Decrease in plasma membrane tension triggers PtdIns(4,5)P2 phase separation to inactivate TORC2.Nat Cell Biol. 2018 Sep;20(9):1043-1051. doi: 10.1038/s41556-018-0150-z. Epub 2018 Aug 27. Nat Cell Biol. 2018. PMID: 30154550 Free PMC article.
-
Rictor/TORC2 regulates Caenorhabditis elegans fat storage, body size, and development through sgk-1.PLoS Biol. 2009 Mar 3;7(3):e60. doi: 10.1371/journal.pbio.1000060. PLoS Biol. 2009. PMID: 19260765 Free PMC article.
-
Cell Size and Growth Rate Are Modulated by TORC2-Dependent Signals.Curr Biol. 2018 Jan 22;28(2):196-210.e4. doi: 10.1016/j.cub.2017.11.069. Epub 2017 Dec 28. Curr Biol. 2018. PMID: 29290562 Free PMC article.
-
Experimental Approaches in Delineating mTOR Signaling.Genes (Basel). 2020 Jul 2;11(7):738. doi: 10.3390/genes11070738. Genes (Basel). 2020. PMID: 32630768 Free PMC article. Review.
References
-
- Beeler T, Bacikova D, Gable K, Hopkins L, Johnson C, Slife H, Dunn T. The Saccharomyces cerevisiae TSC10/YBR265w gene encoding 3-ketosphinganine reductase is identified in a screen for temperature-sensitive suppressors of the Ca2+-sensitive csg2Delta mutant. J Biol Chem. 1998;273:30688–30694. - PubMed
-
- Canton DA, Litchfield DW. The shape of things to come: an emerging role for protein kinase CK2 in the regulation of cell morphology and the cytoskeleton. Cell Signal. 2006;18:267–275. - PubMed
-
- Clarke CJ, Hannun YA. Neutral sphingomyelinases and nSMase2: bridging the gaps. Biochim Biophys Acta. 2006;1758:1893–1901. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

