Abstract
The development of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines and therapeutics will depend on understanding viral immunity. We studied T cell memory in 42 patients following recovery from COVID-19 (28 with mild disease and 14 with severe disease) and 16 unexposed donors, using interferon-γ-based assays with peptides spanning SARS-CoV-2 except ORF1. The breadth and magnitude of T cell responses were significantly higher in severe compared with mild cases. Total and spike-specific T cell responses correlated with spike-specific antibody responses. We identified 41 peptides containing CD4+ and/or CD8+ epitopes, including six immunodominant regions. Six optimized CD8+ epitopes were defined, with peptide–MHC pentamer-positive cells displaying the central and effector memory phenotype. In mild cases, higher proportions of SARS-CoV-2-specific CD8+ T cells were observed. The identification of T cell responses associated with milder disease will support an understanding of protective immunity and highlights the potential of including non-spike proteins within future COVID-19 vaccine design.
COVID-19 is caused by the recently emerged severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). While the majority of COVID-19 infections are relatively mild, with recovery typically within 2–3 weeks[1, 2], a significant number of patients develop severe illness, which is postulated to be related to both an overactive immune response and viral-induced pathology[3, 4]. The role of T cell immune responses in disease pathogenesis and longer-term protective immunity is currently poorly defined, but essential to understand in order to inform therapeutic interventions and vaccine design.
Currently, there are many ongoing vaccine trials, but it is unknown whether they will provide long-lasting protective immunity. Most vaccines are designed to induce antibodies to the SARS-CoV-2 spike protein, but it is not yet known if this will be sufficient to induce full protective immunity to SARS-CoV-2 (refs. [5, 6, 7, 8]). Studying natural immunity to the virus, including the role of SARS-CoV-2-specific T cells, is critical to fill the current knowledge gaps for improved vaccine design.
For many primary virus infections, it typically takes 7-10 d to prime and expand adaptive T cell immune responses in order to control the virus[9]. This coincides with the typical time it takes for patients with COVID-19 to either recover or develop severe illness. There is an incubation time of 4-7 d before symptom onset and a further 7-10 d before individuals progress to severe disease[10]. Such a pattern of progression raises the possibility that a poor T cell response contributes to SARS-CoV-2 viral persistence and COVID-19 mortality, whereas strong T cell responses are protective in the majority of individuals.
Evidence supporting a role for T cells in COVID-19 protection and pathogenesis is currently incomplete and sometimes conflicting[3, 11, 12, 13, 14]. To date, there have been few studies analyzing SARS-CoV-2-specific T cell responses and their role in disease progression[15], although virus-specific T cells have been shown to be protective in human influenza infection[16]. In a study of CD4+ and CD8+ T cell responses to SARS-CoV-2 in non-hospitalized convalescent individuals, Grifoni et al.[17] found that all recovered patients established CD4+ responses and 70% established CD8+ memory responses to SARS-CoV-2. SARS-CoV-2-specific CD4+ T cell responses were also frequently observed in unexposed participants in their study, suggesting the possibility of pre-existing cross-reactive immune memory to seasonal coronaviruses. In Singapore, Le Bert et al.[18] found long-lasting T cell immunity to the original SARS coronavirus nucleoprotein (NP) in those who were infected in 2003. These T cells cross-reacted with SARS-CoV-2 NP, and T cells cross-reactive with non-structural proteins 7 and 13 of other coronaviruses were also present in those uninfected with either of the SARS coronaviruses[18].
In the present study, the overall and immunodominant SARS-CoV-2-specific memory T cell responses in patients who had recovered from COVID-19 were evaluated ex vivo using peptides spanning the full proteome of SARS-CoV-2, except ORF1. Epitopes were identified using two-dimensional matrix peptide pools, and CD4+ and CD8+ T cell responses were distinguished. The epitope specificity and human leukocyte antigen (HLA) restriction of the dominant CD8+ T cell responses were defined in ex vivo assays and using in vitro-cultured short-term T cell lines. The ex vivo functions of SARS-CoV-2-specific T cells specific for dominant epitopes were evaluated by their intracellular cytokine production profiles. Broad, and frequently strong, SARS-CoV-2-specific CD4+ and CD8+ T cell responses were seen in the majority of convalescent patients, with significantly larger overall T cell responses in those who had severe compared with mild disease. However, there was a greater proportion of CD8+ T cell compared with CD4+ T cell responses in mild cases, with higher frequencies of multi-cytokine production by matrix (M)- and NP- specific CD8+ T cells.
Results
Study participants
A total of 42 individuals were recruited following recovery from COVID-19, including 28 mild cases and 14 severe cases. In addition, 16 control individuals sampled in 2017–2019, before COVID-19 appeared, were studied in parallel. Supplementary Fig. 1 shows the participant characteristics. No significant differences in gender or age were noted between mild and severe groups. The percentage of oxyhemoglobin saturation in arterial blood (SaO2)/fraction of inspired O2 (FiO2) ratio in severe cases ranged from 4.3 (where 4.5 would be the estimate for an individual with mild disease breathing ambient air) to 1.6, with the patients with critical disease having an estimate of 0.8 (median in severe group = 3.8).
Ex vivo assessment of memory T cell responses specific to SARS-CoV-2
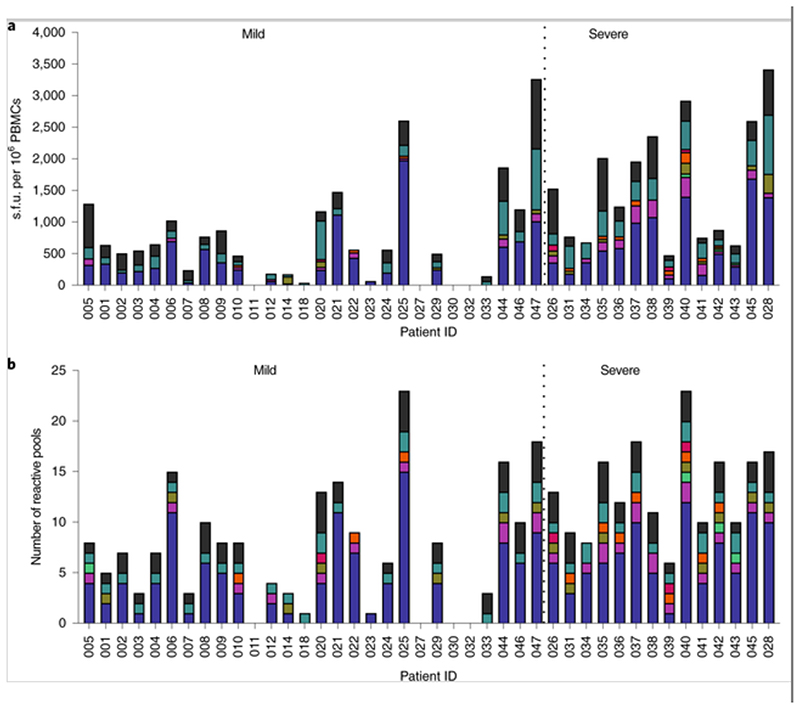
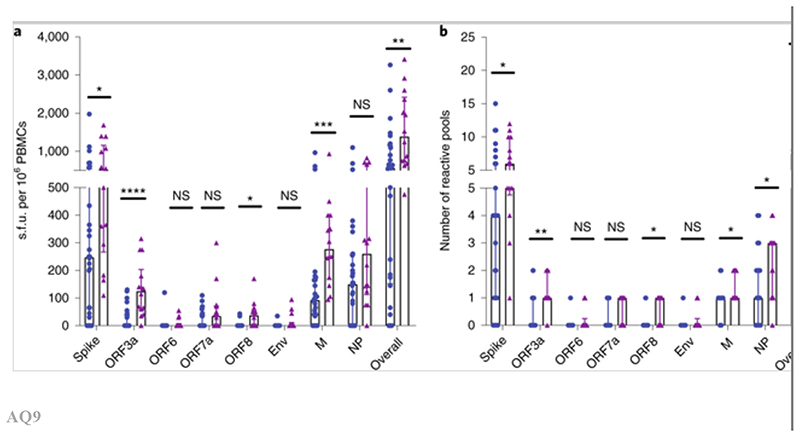
Peripheral blood mononuclear cells (PBMCs) were tested for responses to a panel of 423 overlapping peptides spanning the SARS-CoV-2 proteome except ORF1, using ex vivo interferon-γ (IFN-γ) enzyme-linked immunospot (ELISpot) assays. All overlapping peptides were placed into two twodimensional peptide matrices. A total of 61 peptide pools were tested, with 29 peptides in the first-dimension pools, as described in Supplementary Table 1. The majority of the participants exhibited SARS-CoV-2 memory T cell responses to at least one of the peptides. The overall distribution, magnitude and breadth of the IFN-γ responses against all SARS-CoV-2 virus peptides are shown in Fig. 1. There was no correlation between the T cell responses and the time that had elapsed from symptom development (Supplementary Fig. 2). No ex vivo IFN-γ-producing SARS-CoV-2-specific T cell responses were observed in healthy volunteers, who were all sampled before any chance of exposure, but in those with appropriate HLA types, T cell responses were observed to influenza virus, Epstein–Barr virus and cytomegalovirus (CMV) using pools of known T cell epitopes, as well as phytohemagglutinin, as positive controls (Supplementary Fig. 3). The breadth and magnitude of the T cell responses varied considerably between individuals. T cell responses were detected against epitopes distributed across a wide variety of virus proteins. Significantly higher-magnitude (P = 0.002) and broader (P = 0.002) overall T cell responses were observed in severe cases compared with mild cases, in particular for responses to spike (P = 0.021 for magnitude; P = 0.016 for breadth), membrane (P = 0.0003 for magnitude; P = 0.033 for breadth), ORF3 (P < 0.0001 for magnitude; P < 0.001 for breadth) and ORF8 proteins (P = 0.011 for magnitude; P = 0.014 for breadth) (Fig. 2). Overall, we found that strong and broad T cell memory responses were induced after recovery from COVID-19, and the breadth and magnitude of T cell responses were significantly higher in severe compared with mild cases.
Fig. 1. Memory T cell responses specific to SARS-CoV-2 virus proteins in 42 convalescent patients infected with SARS-CoV-2.
Of the 42 patients studied, 28 had mild symptoms while 14 showed severe symptoms. PBMCs were isolated and IFN-γ production was detected by ELISpot after incubation with SARS-CoV-2 peptides. a, Magnitude of IFN-γ T cell responses for each individual. Each bar shows the total T cell responses of each individual specific to all of the SARS-CoV-2 protein peptides tested. Each colored segment represents the source protein corresponding to peptide pools eliciting IFN-γ T cell responses. b, Breadth of T cell responses for each individual. The breadth of T cell responses was calculated by the number of peptide pools in the first-dimension (n = 29) cells that responded to spot-forming units. The experiments were repeated in 35 participants where sample availability permitted. Env: envelope protein
Fig. 2. Comparison of the magnitude and breadth of T cell responses specific to each viral protein between convalescent patients with mild symptoms and those with severe symptoms.
PBMCs were isolated and IFN-γ production was detected by ELISpot after incubation with SARS-CoV-2 peptides. a,b, Magnitude (a) and breadth (b) of T cell responses against each viral protein between those with mild symptoms (n = 28) and those with severe symptoms (n= 14). P values were as follows: overall: P = 0.002 for magnitude; P = 0.002 for breadth; spike: P = 0.021 for magnitude; P = 0.016 for breadth; membrane: P = 0.0003 for magnitude; P = 0.033 for breadth; ORF3a: P < 0.0001 for magnitude; P = 0.001 for breadth; ORF8: P = 0.011 for magnitude; P = 0.014 for breadth. Data are presented as medians with interquartile ranges. The Mann–Whitney U-test was used for the analysis and two-tailed P values were calculated. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.001. NS, not significant.
Correlation with spike-specific antibody responses
The relationships between overall and spike-specific T cell responses and spike-specific, receptor-binding domain (RBD)-specific and NP-specific antibody end-point titers (EPTs) were assessed (Fig. 3 and supplementary figure 4). There were significant correlations between: (1) spike-specific antibody titers and both overall T cell responses (P = 0.0004; R = 0.5185) and spike-specific T cell responses (P = 0.0006; R = 0.505); (2) RBD-specific antibody titers and both overall T cell responses (P = 0.0004; R = 0.5198) and spike-specific T cell responses (P = 0.0004; R = 0.5189); and (3) NP-specific antibody titers and both overall T cell responses (P = 0.0015; R = 0.4738) and spike-specific T cell responses (P = 0.007; R = 0.412). However, there was no significant association between NP-specific antibody titers and NP-specific T cell responses (P = 0.067; R = 0.286) (Supplementary Fig. 4). Moreover, significantly higher levels of spike, RBD and NP EPTs were observed in severe cases compared with mild cases (Fig. 3d). It was noted that some individuals had low RBD-specific antibodies (Fig. 3b), yet had detectable spike-specific antibodies (Fig. 3a), suggesting that antibodies were able to target non-RBD regions of spike. This is under further investigation. Thus, total and spike-specific T cell responses were found to be correlated with spike-specific antibody responses.
Fig. 3. Correlation of T cell responses against SARS-CoV-2 with spike-, RBD- and NP-specific antibody responses.
a-c, Spike (a), RBD (b) and NP EPTs (c) in association with overall T cell responses. Red and black data points represent patients with severe and mild symptoms, respectively. n = 42. Spearman’s rank correlation coefficients (R) are shown. d, Comparison of spike EPTs (P < 0.0001), RBD EPTs (P < 0.0001) and NP EPTs (P = 0.0004) for patients with mild symptoms (n = 28) versus severe symptoms (n = 14). Data are presented as medians with interquartile ranges and a Mann-Whitney U-test was used for comparison. Two-tailed P values were calculated. ***P < 0.001; ****P < 0.0001.
Distribution of SARS-CoV-2-specific CD4+ and CD8+ memory T cell responses
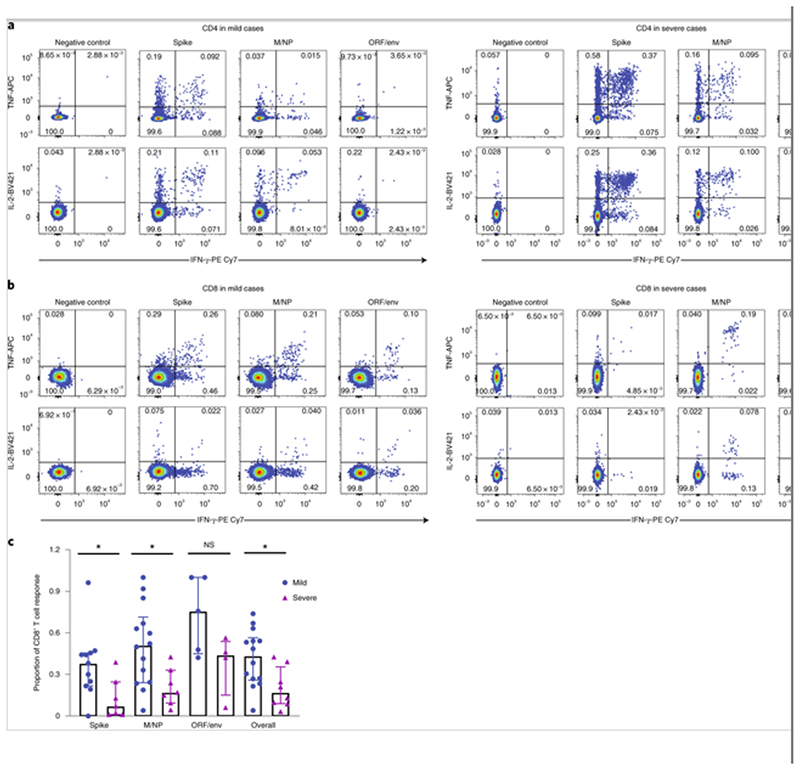
Having identified overall T cell responses to SARS-CoV-2 peptides, the responses detected against positive peptide pools were characterized by flow cytometry for peptide recognition by CD4+ or CD8+ T cell subsets and for intracellular production of IFN-γ, tumor necrosis factor-α (TNF-α) and interleukin-2 (IL-2) after stimulation (Fig. 4a,b and Supplementary Fig. 5). A greater proportion of the T cell responses to spike (P = 0.0268) and M/NP (P = 0.02) were contributed to by CD8+ T cells in those with mild disease compared with those with severe disease (Fig. 4c and Supplementary Fig. 6a). Differential subsets of SARS-CoV-2-specific T cells therefore associate with clinical outcome.
Fig. 4. Distribution of SARS-CoV-2-specific CD4+ and CD8+ memory T cell responses.
Cytokine-producing T cells were detected by ICS after incubation with SARS-CoV-2 peptides. a,b, Flow cytometric plots representing CD4+ T cells (a) and CD8+ T cells (b) expressing IFN-γ (x axis), TNF (y axis) and/or IL-2 (y axis) upon stimulation with the respective SARS-CoV-2 peptide pools in examples of mild (left) and severe cases (right). c, Comparison of the relative proportion of SARS-CoV-2 peptide pool-reactive CD8+ T cells between mild (spike, n = 11; M/NP, n = 14; ORF/Env, n = 5; overall: n = 14) and severe cases (spike, n = 7; M/NP, n = 7; ORF/Env, n = 4; overall, n = 8). P values were as follows: P = 0.0268 (spike); P = 0.02 (M/NP); P = 0.0159 (overall). The SARS-CoV-2 peptide pool-reactive CD4+ or CD8+ T cells were identified with at least one of the three cytokines (IFN-γ, TNF and IL-2) detected. Data are shown as medians with interquartile ranges. The Mann-Whitney U-test was used for the analysis. Two-tailed P values were calculated. *P < 0.05.
Evaluation of the polyfunctionality of T cells responding to SARS-CoV-2 peptides
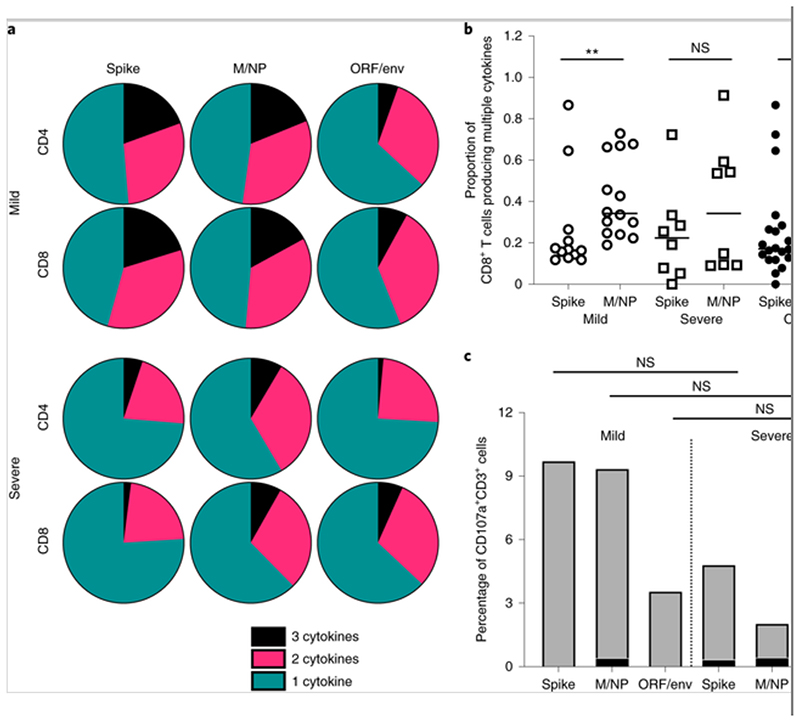
Multi-cytokine analysis revealed patterns of IFN-γ, TNF and IL-2 production by CD4+ and CD8+ T cells in both mild and severe cases (Fig. 5a). For 22 individuals tested, both CD4+ and CD8+ antigen-specific T cells produced at least one of these three cytokines and others in combination. CD8+ but not CD4+ T cells targeting different virus proteins showed different cytokine profiles, with the M/NP-specific CD8+ T cells showing wider functionality than T cells targeting spike protein (P = 0.0231; Fig. 5b and Supplementary Fig. 6b). Furthermore, there were a greater proportion of multifunctional M/NP-specific CD8+ T cells compared with spike-specific T cells in those who had mild disease (P = 0.0037), but not in those who had severe disease (P = 0.3823). In contrast with observations seen in influenza virus infection[19], we did not observe significant differences in the cytotoxic potential (as indicated by expression of the degranulation marker CD107a) in patients with mild and severe disease (Fig. 5c) and we observed very few CD107a+CD4+ T cells overall, suggesting that cytotoxic CD4+ T cells might not be a major contributor to virus clearance.
Fig. 5. Cytokine profile of SARS-Cov-2-specific T cells.
The cytokine production of SARS-Cov-2-specific T cells was assessed by ICS after incubation with SARS-CoV-2 peptides. a, Pie charts representing the relative proportions of spike-, M/NP- and ORF/env-specific CD4+ and CD8+ T cells producing one (green), two (pink) or three cytokines (black) (out of IFN-γ, TNF and IL-2). b, Comparison of the frequency of multifunctional CD8+ T cells targeting spike and M/NP. The open circles and squares represent T cell responses in mild cases and severe cases, respectively. P values are as follows: P = 0.0037 (mild); P = 0.3823 (severe); P = 0.0231 (overall). c, Relative frequencies of CD4+ and CD8+ T cells expressing CD107a after antigen stimulation. The data shown are from 14 patients with mild symptoms and eight patients with severe symptoms. The Mann–Whitney U-test was used for the analysis. Two-tailed P values were calculated. *P < 0.05; **P < 0.01.
Identification of SARS-CoV-2-specific T cell peptides containing epitopes
IFN-γ ELISpot assays were performed with candidate peptides identified from the two-dimensional matrix analysis in 34 participants. A total of 41 peptides containing SARS-CoV-2 T cell epitope regions were recognized by convalescent individuals who had COVID-19: 18 from spike, ten from NP, six from membrane and seven from ORF proteins. Strikingly, six dominant 18-mer peptides were recognized by six or more of the 34 participants tested (Table 1). NP-16 was recognized by 12 out of 34 (35%) participants tested and contained at least two epitopes that were recognized by either CD4+ T or CD8+ T cells.
Table 1. Peptides containing T cell epitopes.
| Peptide | Position | Amino acid sequence | CD4+/CD8+ T cell response | Number of participants | |
|---|---|---|---|---|---|
| Spike(n = 18) | S-34a | 166-180 | CTFEYVSQPFLMDLE | 4/8 | 10 |
| S-39 | 191-205 | EFVFKNIDGYFKIYS | NA | 1 | |
| S-42 | 206-230 | KHTPINLVRDLPQGF | NA | 1 | |
| S-43 | 211-225 | NLVRDLPQGFSALEP | NA | 1 | |
| S-71 | 351-365 | YAWNRKRISNCVADY | 4 | 1 | |
| S-77 | 381-395 | GVSPTKLNDLCFTNV | 4 | 1 | |
| S-90 | 446-460 | GGNYNYLYRLFRKSN | NA | 1 | |
| S-91 | 451-465 | YLYRLFRKSNLKPFE | NA | 1 | |
| S-103 | 506-520 | VVLSFELLHAPATVC | 4 | 1 | |
| S-106 | 526-540 | GPKKSTNLVKNKCVN | 8 | 1 | |
| S-145 | 721-735 | SVTTEILPVSMTKTS | NA | 1 | |
| S-150 | 746-760 | STECSNLLLQYGSFC | NA | 1 | |
| S-151a | 751-765 | NLLLQYGSFCTQLNR | 4 | 8 | |
| S-161 | 801-815 | NFSQILPDPSKPSKR | 4 | 2 | |
| S-174a | 866-880 | TDEMIAQYTSALLAG | 4 | 6 | |
| S-235 | 1,171-1,185 | GINASVVNIQKEIDR | NA | 1 | |
| S-240 | 1,196-1,210 | LIDLQELGKYEQYI | NA | 1 | |
| S-242 | 1,206-1,220 | YEQYIKWPWYIWLGF | NA | 1 | |
| NP (n =10) | NP-1 | 1-17 | MSDNGPQNQRNAPRITF | 8 | 3 |
| NP-2 | 8-25 | NQRNAPRITFGGPSDSTG | 8 | 3 | |
| NP-12 | 82-95 | DQIGYYRRATRRIR | NA | 1 | |
| NP-15 | 101-113 | MKDLSPRWYFYYL | NA | 1 | |
| NP-16a | 104-121 | LSPRWYFYYLGTGPEAGL | 4/8 | 12 | |
| NP-46 | 313-330 | AFFGMSRIGMEVTPSGTW | NA | 1 | |
| NP-47 | 321-338 | GMEVTPSGTWLTYTGAIK | NA | 1 | |
| NP-48 | 329-346 | TWLTYTGAIKLDDKDPNF | 4 | 2 | |
| NP-50 | 344-361 | PNFKDQVILLNKHIDAYK | 4 | 1 | |
| NP-51 | 352-369 | LLNKHIDAYKTFPPTEPK | 8 | 3 | |
| M19 | 133-150 | LLESELVIGAVILRGHLR | NA | 3 | |
| M (n= 6) | M-20a | 141-158 | GAVILRGHLRIAGHHLGR | 4 | 11 |
| M-21 | 149-166 | LRIAGHHLGRCDIKDLPK | NA | 3 | |
| M-23 | 165-181 | PKEITVATSRTLSYYKL | NA | 3 | |
| M-24a | 172-188 | TSRTLSYYKLGASQRVA | 4 | 16 | |
| M-28 | 201-218 | IGNYKLNTDHSSSSDNIA | NA | 1 | |
| ORFs(n =7) | ORF3a-20 | 145-160 | YFLCWHTNCYDYCIPY | NA | 1 |
| ORF3a-27 | 198-215 | KDCVVLHSYFTSDYYQLY | NA | 3 | |
| ORF3a-28 | 206-225 | YFTSDYYQLYSTQLSTDTGV | 8 | 4 | |
| ORF3a-30 | 224-243 | GVEHVTFFIYNKIVDEPEEH | NA | 1 | |
| ORF7a-2 | 9-25 | LITLATCELYHYQECVR | NA | 3 | |
| ORF7a-7 | 46-63 | FHPLADNKFALTCFSTQF | NA | 1 | |
| ORF7a-10 | 69-86 | DGVKHVYQLRARSVSPKL | 4 | 1 |
Overlaps of two adjacent peptides recognized by the same participants are shown in
Immunodominant peptides
not available.
M-24 was recognized by 16 out of 34 participants (47%) tested and contained one or more CD4+ T cell epitopes. Peptide M-20 was recognized by 11 out of 34 participants tested (32%) and contained one or more CD4+ T cell epitopes. Three dominant spike peptides were also identified, with S-34 recognized by ten out of 34 participants (29%) containing both CD4+ and CD8+ T cell epitopes, and a further two spike peptides (S-151 and S-174) were recognized by eight and six out of 34 participants, respectively (24 and 18%), both containing CD4+ T cell epitopes.
Those dominant responses were further confirmed by ex vivo assays and using cultured short-term T cell lines. Supplementary Fig. 7 illustrates examples of fluorescence-activated cell sorting plots from intracellular cytokine staining (ICS) when short-term T cell lines were stimulated with single peptides containing epitopes. CD4+ T cells elicited strong responses against dominant spike peptides and M peptides, whereas cells targeting two NP-dominant peptides were CD8+ T cells. The optimum epitopes within the long peptides recognized by dominant CD8+ T cells, and their HLA restriction matched to the donor’s HLA type, were predicted using the Immune Epitope Database analysis resource (http://tools.iedb.org/mhci). The best-predicted epitope sequences are shown in Supplementary Table 2.
A set of previously defined SARS epitopes[20] with identical sequences to SARS-CoV-2 were also tested by ELISpot assay (Supplementary Table 3). Most of those peptides did not elicit any positive responses in 42 individuals who had recovered from COVID-19, apart from two NP epitope peptides (N-E-3 (MEVTPSGTWL) and N-E-11 (LLNKHIDAYKTFPPTEPK)) and one spike epitope peptide (S-E-19; QLIRAAEIRASANLAATK). N-E-11, which is identical to peptide NP-51, shares the sequence with two other known HLA-A*0201-restricted SARS epitopes (N-E-1 (ILLNKHID) and N-E-5 (ILLNKHIDA)). Interestingly, one of the responders to this peptide did not carry the HLA-A*0201 allele (Table 1), indicating that this peptide may contain a different SARS-CoV-2 epitope presented by a different HLA molecule. Whereas these NP epitopes are targeted by CD8+ T cells, we also detected a CD4+ T cell response targeting SARS spike epitope S-E-19, which spans between the overlapping peptides of S-203 and S-204. This peptide is known to be presented by HLA-DRB1*0401 in SARS infection.
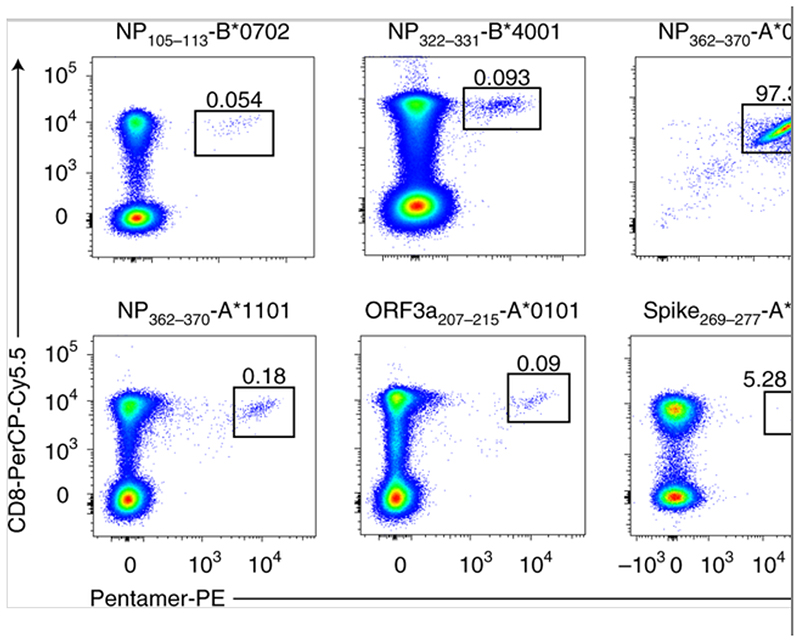
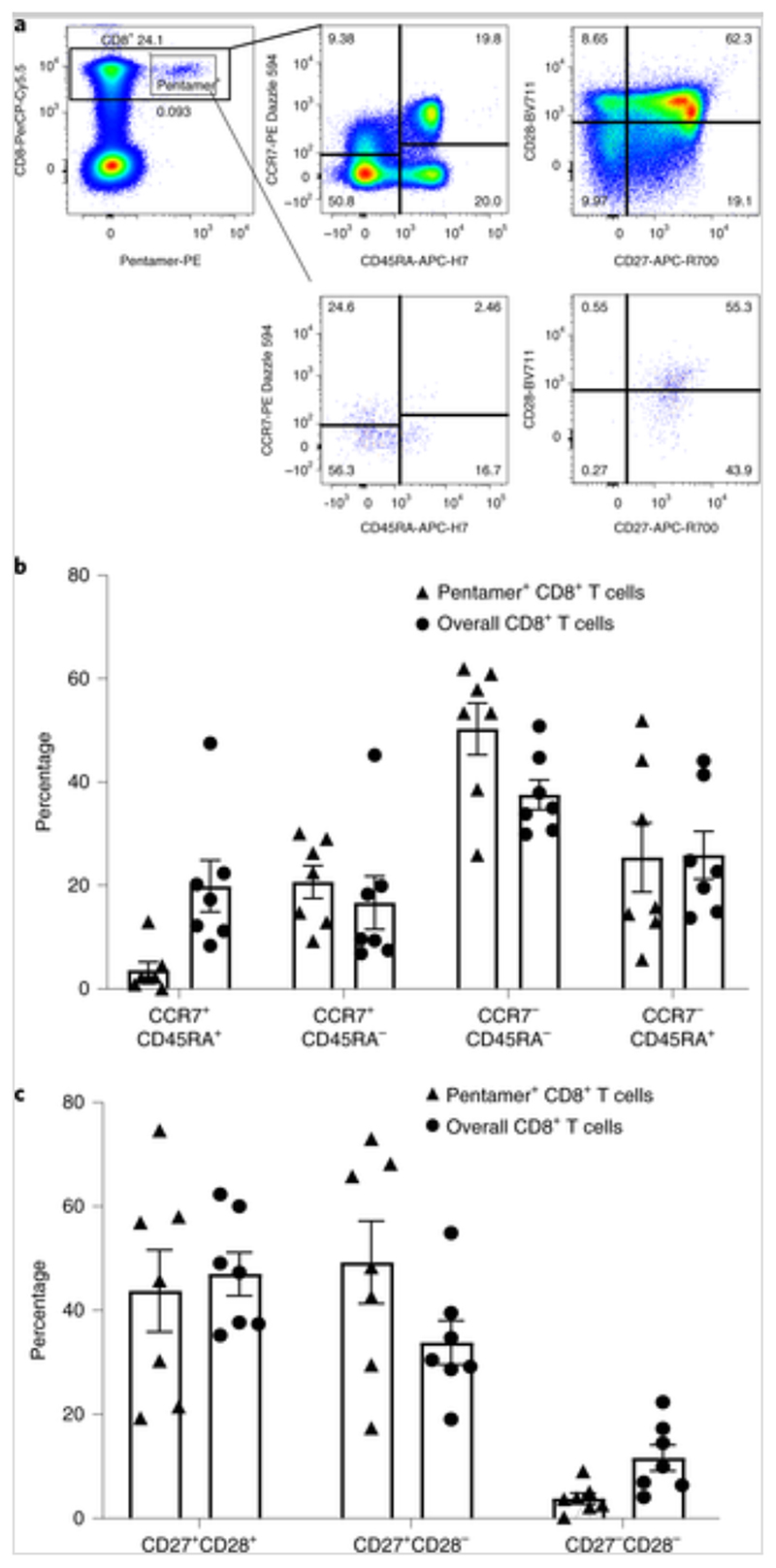
The optimum peptide sequences and their HLA restrictions were confirmed by generating short-term T cell lines and clones, which were tested in ELISpot assays by co-culturing with peptide-loaded HLA-matched and -unmatched immortalized B lymphoblastoid cell lines, as previously described[21]. In total, six CD8+ T cell epitopes restricted by HLA-A*0101, -A*0301, -A*1101, -B*0702, -B*4001 and -B*2705 were confirmed (Table 2). HLA-peptide pentamers were synthesized comprising five peptides bound to the appropriate HLA class I molecules. T cell staining was verified by flow cytometry (Fig. 6) and their phenotypes were determined (Fig. 7). A pentameric HLA-A*0201 with the spike epitope reported by Shomuradova et al.[22], was synthesized. Only one out of six HLA-A*0201-positive donors showed detectable staining, but at a very low frequency. The majority of pentamer-stained SARS-CoV-2-specific CD8+ T cells exhibited central memory (20.7 ± 8.4%) or effector memory phenotypes (50.3 ± 13.3%) (Fig. 7) and early (CD27+CD28+; 43.8 ± 20.9%) or intermediate (CD27+CD28-; 49.3 ± 21.0%) differentiation phenotypes. Overall, multiple peptides containing epitopes and immunodominant regions were defined from 42 individuals who had recovered from COVID-19. The regions were located in the majority of SARS-CoV-2 structural and non-structural proteins, including spike, M, NP and ORF proteins, with CD8+ T cells exhibiting central memory and effector memory phenotypes.
Table 2. Location, sequence and HLA restriction of six identified SARS-CoV2 CD8 optimum epitopes.
| Protein | Position | Epitope sequence | HLA restriction |
|---|---|---|---|
| NP | 9-17 | QRNAPRITF | B*2705 |
| 105-113 | SPRWYFYYL | B*0702 | |
| 322-331 | MEVTPSGTWL | B*4001 | |
| 362-370 | KTFPPTEPK | A*0301 | |
| 362-370 | KTFPPTEPK | A*1101 | |
| ORF3a | 207-215 | FTSDYYQLY | A*0101 |
Fig. 6. Defined SARS-CoV-2-specific CD8 epitopes.
Examples of peptide–MHC class I pentamer staining ex vivo, with PBMCs (HLA-B*0702, -B*4001, -A*1101, -A*0101 and -A*0201) or with cultured cell lines (HLA-A*0301). Eleven donors were tested with positive pentamer staining.
Fig. 7. Memory phenotype and differentiation status of SARS-CoV-2-specific CD8+ T cells.
PBMCs were isolated and stained with peptide–MHC class I pentameric complexes and markers of T cell memory and differentiation. a, Representative fluorescence-activated cell sorting plots of gating for different cell subsets. b,c, Expression of memory markers (CCR7 and CD45RA) (b) and differentiation markers (CD27 and CD28) (c) on CD8+ pentamer+ T cells. n = 7 donors. Data are presented as means ± s.e.m.
Discussion
This study demonstrates the presence of robust memory T cell responses specific for SARS-CoV-2 in the blood of donors who have recovered from COVID-19. The broader and stronger SARS-CoV-2-specific T cell responses in patients who had severe disease may be the result of higher viral loads and may reflect a poorly functioning early T cell response that failed to control the virus, in addition to other factors such as direct virus-induced pathology associated with larger viral inoculums or poorer innate immunity. Alternatively, it is possible that the T cell response was itself harmful and contributes to disease severity. Consistent with recent reports from Grifoni et al.[17] and Sekine et al. [23], a particularly high frequency of spike protein-specific CD4+ T cell responses was observed in patients who had recovered from COVID-19. This is very similar to influenza virus infection, where viral surface hemagglutinin elicited mostly CD4+ T cell responses, whereas the majority of CD8+ T cell responses were specific to viral internal proteins[24]. Understanding the roles of different subsets of T cells in protection or pathogenesis is crucial for the prevention and treatment of COVID-19. The timing and strength of the first T cell responses could be critical in determining this balance at an early stage of the infection.
Among the 41 peptides containing T cell epitopes that were identified in this study, six immunodominant epitope groups (peptides) were frequently targeted by T cells in many donors, including three in spike protein (29, 24 and 18%), two in membrane protein (32 and 47%) and one in NP (35%). The immunodominant peptide regions identified here may include multiple epitopes restricted by different HLAs (both class I and II, such as S-34 and NP-16), with immunodominance preferences imposed by the antigen-processing pathways. Whether or not these dominant responses play a role in immune protection merits further investigation in larger prospective cohorts.
A higher proportion of CD8+ T cell responses was observed in mild disease, suggesting a potential protective role of CD8+ T cell responses in mild disease or a pathogenic role of CD4+ T cell responses in severe disease, which merits further investigation.
The majority of pentamer-binding CD8+ T cells were effector memory and central memory with early and intermediate differentiation phenotypes, with functional potential on antigen re-exposure. Because the number of donors studied was limited and they would probably show diverse T cell receptors, peptide–major histocompatibility complex (MHC) affinities and antigen sensitivities for the different epitopes, it was not possible to make a detailed analysis comparing mild and severe cases. However, the groundwork, including epitope identification, was laid for future studies that can address this important issue.
Multiple strong dominant T cell responses were seen in study participants that were specific for the M and NP proteins. Dominant epitope regions within NP (NP-16) were detected in 35% of study participants, and dominant epitope regions within matrix (M-20 and M-24) were detected in 32 and 47%, respectively. In addition, a higher proportion of multi-cytokine-producing M/NP-specific T cells compared with spike-specific CD8+ T cells were observed in individuals who had recovered from mild disease. A similar trend was also observed in severe cases, although this was not significant, possibly due to fewer cases. These data strongly suggest that NP and M have the potential for inclusion within future vaccines so as to stimulate strong effector T cell responses. Furthermore, T cells responding to these antigens may be more cross-reactive[18].
IFN-γ-producing SARS-CoV-2-specific T cell responses were not observed in 16 healthy unexposed volunteers, in contrast with recently published reports by Grifoni et al.[17] and Braun et al.[25], both of which used peptide-stimulated activation-induced marker (AIM) assays. In contrast, in a recent immunogenicity study of a recombinant adenovirus type-5-vectored COVID-19 vaccine human phase I trial in 108 volunteers without pre-exposure to COVID-19, spike-specific T cell responses, measured IFN-γ ELISpots and ICS assays were not found before vaccination[6]. These differences could result from differences in the sensitivity of the detection methods.
AIM versus IFN-γ production assays
IFN-γ ELISpot and ICS are well-established methods for evaluating antigen-specific T cells, used in different virus infections and vaccine studies, that have direct functional relevance[24, 26, 27, 28]. The AIM assay is a more recently developed assay, capable of detecting early-responding T cells, that is independent of cytokine production. Both methods are valid but differ in sensitivity and possible functional relevance. However, it is also possible that different circulating coronaviruses have been previously present in the different geographical populations studied, giving cross-reactive responses in some regions but not others, as suggested by Le Bert et al.[18]. These T cell cross-reacting viruses could include not only SARS-CoV-1 and human common cold coronaviruses, but also other unknown coronaviruses of animal origin. It is also known that very sensitive assays can detect not only pre-existing naive antigen-specific CD4+ T cells but also memory CD4+ T cells. The latter are potentially primed by other microbes that cross-react with viruses as diverse as CMV, human immunodeficiency virus type 1 and Ebola virus in most unexposed humans[29, 30]. Therefore, similar findings with SARS-CoV-2 peptides do not necessarily mean the T cells were primed by previous infecting coronaviruses. Indeed, the implications of pre-existing cross-reactivity to seasonal coronavirus and other viruses for COVID-19 immunity merit further detailed investigation, as highlighted by Sette and Crotty[31].
This study focuses on T cell responses in PBMCs. There remains a lack of understanding of memory T cells at the site of infection, which probably provide the most potent protection, as observed in influenza virus infection[32]. It is possible that the hierarchy of immunodominant circulating blood memory T cell pools may not exactly reflect that of memory T cells in the lungs[17, 33, 34]. Therefore, understanding the features of tissue-resident memory T cells and their association with disease severity will be critical and also merits further investigation.
Taken together, this study has demonstrated strong and broad SARS-CoV-2-specific CD4+ and CD8+ T cell responses in the majority of humans who had recovered from COVID-19. The immunodominant epitope regions and peptides containing T cell epitopes identified in this study will provide critical tools with which to study the contribution of SARS-CoV-19-specific T cells in protection and immune pathology. The identification of non-spike dominant CD8+ T cell epitopes suggests the potential importance of including non-spike proteins such as NP, M and ORFs in future vaccine designs.
Methods
Ethics
Patients were recruited from the John Radcliffe Hospital in Oxford, United Kingdom, between March and May 2020 by the identification of patients hospitalized during the SARS-COV-2 pandemic and recruited into the Sepsis Immunomics and International Severe Acute Respiratory and Emerging Infection Consortium World Health Organization Clinical Characterisation Protocol UK (IRAS260007 and IRAS126600). Patients were sampled at least 28 d from the start of their symptoms. Unexposed healthy adult donor samples were used from unrelated studies undertaken between 2017 and early 2019. Written informed consent was obtained from all patients. Ethical approval was given by the South Central–Oxford C Research Ethics Committee in England (reference: 13/SC/0149), Scotland A Research Ethics Committee (reference: 20/SS/0028) and World Health Organization Ethics Review Committee (RPC571 and RPC572l; 25 April 2013).
Clinical definitions
All patients were confirmed to have a test positive for SARS-CoV-2 using PCR with reverse transcription from an upper respiratory tract (nose and throat) swab tested at an accredited laboratory. The degree of severity was identified as mild, severe or critical infection, according to recommendations from the World Health Organization. Patients were classified as having mild symptoms if they did not require oxygen (that is, their oxygen saturation was greater than 93% on ambient air) or if their symptoms were managed at home. A large proportion of our mild cases were admitted to hospital for public health reasons during the early phase of the pandemic even though they had no medical reason to be admitted to hospital. Severe infection was defined as one of the following conditions in a patient confirmed as having COVID-19: respiratory distress with a respiratory rate of >30 breaths per minute; blood oxygen saturation of <93%; or arterial oxygen partial pressure/FiO2 < 300 mmHg. Critical infection was defined as: respiratory failure requiring mechanical ventilation or shock; or other organ failures requiring admission to an intensive care unit. Since the severe classification could potentially include individuals spanning a wide spectrum of disease severity, ranging from patients receiving oxygen through a nasal cannula through to those receiving non-invasive ventilation, we also calculated the SaO2/FiO2 ratio at the height of patient illness as a quantitative marker of lung damage. This was calculated by dividing the oxygen saturation (as determined using a bedside pulse oximeter) by the fraction of inspired oxygen (21% for ambient air; 24% for nasal cannulae; 28% for simple face masks; 28, 35, 40 or 60% for Venturi face masks; or precise measurements for non-invasive or invasive ventilation settings). Patients not requiring oxygen who had oxygen saturations (if measured) greater than 93% on ambient air or managed at home were classified as having mild disease. Viral swab Ct values were not available for all patients. In addition, we standardized all of our analyses to the days since symptom onset.
Synthetic peptides
A total of 423 15- to 18-mer peptides overlapping by ten amino acid residues and spanning the full proteome of SARS-CoV-2 except ORF1 (Supplementary Table 1) were designed using the software PeptGen (http://www.hiv.lanl.gov/content/sequence/PEPTGEN/peptgen.html) and synthesized (purity: >75%; ProImmune).
A total of 27 previously defined SARS epitopes[20] were also synthesized (Supplementary Table 2). Pools of CMV, Epstein-Barr virus and influenza virus-specific epitope peptides and the human immunodeficiency virus Gag protein were also used as positive and negative controls.
Two-dimensional peptide matrix system
The overlapping peptides spanning SARS-CoV-2 were assigned to a two-dimensional matrix system in which each peptide was represented in two different peptide pools. Each peptide pool contained no more than 16 individual peptides. The first dimension of the peptide matrix system was designed so that peptides from different source proteins were separated into different pools. (Supplementary Table 1).
Ex vivo ELISpot assay
IFN-γ ELISpot assays were performed using either freshly isolated or cryopreserved PBMCs, as described previously. No significant difference was observed between responses generated by fresh and cryopreserved PBMCs, as described previously[24, 35].
Overlapping peptides were pooled and then added to 200,000 PBMCs per test at a final concentration of 2 μg ml-1 for 16-18 h. The positive responses were confirmed by repeat ELISpot assays. To quantify antigen-specific responses, mean spots of the control wells were subtracted from the positive wells, and the results were expressed as spot-forming units (s.f.u.) per 106 PBMCs. Responses were considered positive if the results were at least three times the mean of the negative control wells and >25 s.f.u. per 106 PBMCs. If negative control wells had >30 s.f.u. per 106 PBMCs or positive control wells (phytohemagglutinin stimulation) were negative, the results were excluded from further analysis.
Determination of plasma binding to trimeric spike, RBD and NP by enzyme-linked immunosorbent assay
MaxiSorp immunoplates (442404; NUNC) were coated with 0.125 μg StrepMAB-Classic (2-1507-001; IBA), blocked with 2% skimmed milk in phosphate-buffered saline (PBS) for 1 h and then incubated with 50 μl of 5 μg ml-1 soluble trimeric spike and 2 μg ml-1 of 2% skimmed milk in PBS. After 1 h, 50 μl of serial twofold dilutions of plasma, from 1:50 to 1:51,200 in PBS containing 2% skimmed milk, were added followed by Alkaline phosphatase-conjugated anti-human IgG (A9544; Sigma-Aldrich) at 1:10,000 dilution. The reaction was developed by the addition of para-nitrophenyl phosphate substrate and stopped with NaOH. The absorbance was measured at 405 nm. EPTs were defined as reciprocal plasma dilutions that corresponded to two times the average optical density values obtained with mock. To determine EPTs to RBD and NP, immunoplates were coated with 0.125 μg Tetra-His antibody (34670; Qiagen) followed by 2 and 5 μg ml-1 of soluble RBD and NP, respectively.
ICS
ICS was performed as described previously[36, 37]. Briefly, overnight-rested PBMCs were stimulated with pooled or individual peptides at a final concentration of 10 μg ml-1 for 1 h in the presence of 2 μg ml-1 monoclonal antibodies CD28 and CD49d, and then for an additional 5 h with GolgiPlug, GolgiStop, and surface stained with PE-anti-CD107a. Dead cells were labeled using LIVE/DEAD Fixable Aqua dye from Invitrogen. Surface markers, including BUV395-anti-CD3, BUV737-anti-CD4, PerCP-Cy5.5-anti-CD8, BV510-anti-CD14, BV510-anti-CD16 and BV510-anti-CD19 (BioLegend) were stained. Cells were then washed, fixed with Cytofix/Cytoperm and stained with PE-Cy7-anti-IFNγ (eBioscience), APC-anti-TNFα (eBioscience) or BV421-anti-IL-2 (BioLegend). Negative controls without peptide stimulation were run for each sample. All reagents were from BD Biosciences unless otherwise stated. All samples were acquired on a BD LSRFortessa (BD Biosciences) flow cytometer and analyzed using FlowJo version 10 software. Peptide pool-reactive CD4+ or CD8+ T cells with a frequency lower than 0.05% of CD4+ or CD8+ T cells, respectively, were excluded from analysis. Cytokine responses were background subtracted individually before further analysis. To determine the frequency of different response patterns based on all possible combinations, Boolean gates were created using IFN-γ, TNF-α and IL-2. Cytokine responses were background subtracted individually before further analysis.
Pentamer phenotyping
Cryopreserved PBMCs were thawed, as described above. A total of 1 × 106 live PBMCs were labeled with peptide-MHC class I Pentamer-PE (ProImmune) and incubated for 15 min at 37 °C. Dead cells were first labeled with LIVE/DEAD Fixable Aqua dye (Invitrogen) and then with the surface markers CD3-BUV395, CD8-PerCP.Cy5.5, CD14-BV510, CD16-BV510, CD19-BV510, CD28-BV711, CD27-APC-R700, CD45RA-APC-H7 and CCR7-PE-Dazzel 594 (BioLegend). All reagents were from BD Biosciences unless otherwise stated. All samples were acquired on a BD LSRFortessa (BD Biosciences) flow cytometer and analyzed using FlowJo version 10 software.
Generation of short-term T cell lines
Short-term SARS-CoV-2-specific T cell lines were established as previously described[35]. Briefly, 3 × 106 to 5 × 106 PBMCs were pulsed as a pellet for 1 h at 37 °C with 10 μM of peptides containing T cell epitope regions and cultured in R10 at 2 × 106 cells per well in a 24-well Costar plate. IL-2 was added to a final concentration of 100 U ml-1 on day 3 and cultured for further 10-14 d.
Statistical analysis
Statistical analysis was performed with IBM SPSS Statistics 25 and the figures were made with GraphPad Prism 8. Chi-squared tests were used to compare ratio differences between two groups. After testing for normality using the Kolmogorov–Smirnov test, the independent-samples t-test or Mann–Whitney U- test was employed to compare variables between two groups. Correlations were performed via Spearman’s rank correlation. Statistical significance was set at *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001. All of the tests were two tailed.
Reporting Summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Supplementary Material
Editor’s Summary.
Questions have arisen as to whether patients with severe COVID-19 disease can generate a T cell response against SARS-CoV-2. Tao Dong and colleagues report that convalescent patients with COVID-19 harbor functional memory CD4+ and CD8+ T cells that recognize multiple epitopes that span the viral proteome. CD4+ T cells predominated the memory response in patients with severe disease, whereas higher proportions of CD8+ T cells were found in patients with mild disease.
Query Details.
-
We reserve ‘significant’ and its derivatives to mean statistically significant; for all instances, please reword (e.g., ‘important’, ‘notable’, ‘substantial’) or supply a statistical measure such as P value. See the sentence beginning ‘The breadth and magnitude of T cell responses...’ for the first use.
checked
-
Please check your article carefully, coordinate with any co-authors and enter all final edits clearly in the eproof, remembering to save frequently. Once corrections are submitted, we cannot routinely make further changes to the article.
checked
-
Note that the eproof should be amended in only one browser window at any one time; otherwise changes will be overwritten.
OK
-
Author surnames have been highlighted. Please check these carefully and adjust if the first name or surname is marked up incorrectly. Note that changes here will affect indexing of your article in public repositories such as PubMed. Also, carefully check the spelling and numbering of all author names and affiliations, and the corresponding email address(es).
checked
-
You cannot alter accepted Supplementary Information files except for critical changes to scientific content. If you do resupply any files, please also provide a brief (but complete) list of changes. If these are not considered scientific changes, any altered Supplementary files will not be used, only the originally accepted version will be published.
OK
-
Your paper has been copy edited. Please review every sentence to ensure that it conveys your intended meaning; if changes are required, please provide further clarification rather than reverting to the original text. Please note that formatting (including hyphenation, Latin words, and any reference citations that might be mistaken for exponents) has been made consistent with our house style.
checked
-
The present title uses dehumanizing language (i.e., ‘COVID-19 patients’). Would the following title be ok? ‘Broad and strong memory CD4+ and CD8+ T cells induced by SARS-CoV-2 in UK convalescent COVID-19 patients’
We agree, thank you. Your suggestion is the same as the original, and so we have made a new suggestion
-
In the sentence ‘The percentage of oxyhemoglobin saturation in arterial blood...’ please check that the definitions of ‘SaO2’ and ‘FiO2’ are correct as added.
checked
-
‘PHA’ has been expanded to ‘phytohemagglutinin’ twice in the paper. Please check this is correct.
correct
-
At the end of the Fig. 1 caption, please define ‘Env’.
done
-
Please check that the edits to the sentence ‘The relationships between overall T cell response...’ retain the intended meaning. This sentence mentions ‘spike-specific T cell responses’ and then cites ‘Fig. 3’, but Fig. 3 only seems to show overall T cell responses. Please check. Is there a supplementary figure that should be cited here, or can we add ‘(data not shown for spike-specific T cell responses)’ for clarity?
checked and Supplementary Fig. 4 added
-
You say ‘However, there was no significant association between NP-specific antibody titers and NP-specific T cell responses (P = 0.067; R = 0.286) (Supplementary Fig. 4)’ but Fig. 3a-c doesn’t show ‘NP-specific T cell responses’. It only shows ‘Overall T cell responses’, so should the citation to ‘Fig. 3a-c’ be removed from this sentence?
checked and fig.3a-c removed
-
Please check that the edits to the Fig. 3d caption and corresponding figure panel axis labels retain the intended meaning. Note that ‘EPT’ (or a label that represents this) should be on the y axis rather than the x axis as this is what was measured for each group (x axis).
OK
-
In Table 1, per journal style, the superscript ‘a’ has been used to highlight the immunodominant peptides and the bolding for multiple donor responders has been removed as the number is clear for each in the last column. Bolding instead of red has been used to show overlapping amino acid residues, since colour is not permitted in tables. Some additional overlapping sequences were identified. Please check they are all highlighted correctly.
checked
-
Please check that the edits to the sentence ‘Understanding the roles of different subsets of T cells in protection or pathogenesis...’ retain the intended meaning.
checked, and added modification
-
Please check that the edits to the sentence ‘Dominant epitope regions within NP (NP-16)...’ retain the intended meaning.
checked
-
Please check that addition of a level 2 heading ‘AIM versus IFN-γ production assays’ after the sentence ‘These differences could result from...’ is correct.
checked
-
In the sentence ‘Severe infection was defined as one of the following conditions...’ please check that ‘arterial oxygen partial pressure’ is correct (as opposed to ‘SaO2’, which is used elsewhere in the paper.
checked
-
In the sentence ‘After 1 h, 50 μl of serial twofold dilutions of plasma...’ please expand ‘ALP’.
done
-
Please check that the edits to the sentence ‘This work uses data provided by patients and collected...’ retain the intended meaning.
checked
-
Please check that all funders have been appropriately acknowledged and that all grant numbers are correct.
checked
-
Where you refer to ‘Y.Z.’ in the acknowledgements and author contributions, please differentiate between the authors ‘Yonghong Zhang’ and ‘Yuguang Zhao’ using ‘Y. Zhang’ and ‘Y. Zhao’, respectively. Also, please differentiate between ‘Peter Simmonds’ and ‘Paul Sopp’ using ‘P. Simmonds’ and ‘P. Sopp’, respectively.
checked
-
The acknowledgements statement doesn’t include the contributions of Andrew McMichael and Jeremy W. Fry. Please add this information.
checked
-
Please check the wording of the Data availability statement is acceptable as edited.
OK
-
If applicable, please ensure accession codes are scheduled for release on or before this article’s scheduled publication date, and update the database record with publication details from this article once available.
NA
-
Please check that the Competing Interests declaration is correct as stated. If you declare competing interests, please check the full text of the declaration for accuracy and completeness.
OK
-
For ref. 1, please check the author list, page range and year are correct as edited/added.
checked
-
For refs. 4, 5, 8, 11, 15, 17, 20, 31 and 32, please check the volume and page range are correct as added/edited.
checked
-
If refs. 7, 12, 22, 23 and 25 (preprints) have now been published in final peer-reviewed form, please update the reference details if appropriate.
checked and updated
-
For ref. 18, please check the URL is correct as added.
checked
Acknowledgements
We are grateful to all of the participants for donating their samples and data for these analyses, and to the research teams involved in the consenting, recruitment and sampling of these participants. We acknowledge the support of the Oxford Immunology Network COVID-19 Response T Cell Consortium (Supplementary Table 4) and ISARIC4C Investigators (Supplementary Table 5). This work is supported by the UK Medical Research Council (to T.D., G.O., Y.P., M.S., G.N. and Y.-L.C.), Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Sciences (CIFMS), China (grant number 2018-I2M-2-002 to T.D., Y.P., X.Y., G.L., D.D., D.I.S., J.M. and G.R.S.), National Institutes of Health, National Key R&D Program of China (2020YFE0202400 to T.D., Y.Zhang. and R.J.), China Scholarship Council (Z.Y., G.L. and C.L.), National Institute for Health Research (NIHR) (award CO-CIN-01 to M.G.S.), Medical Research Council (grant MC_PC_19059 to M.G.S.), Wellcome Trust and Department for International Development (215091/Z/18/Z to M.G.S.) and Bill and Melinda Gates Foundation (OPP1209135 to M.G.S.). The study is also funded by the NIHR Oxford Biomedical Research Centre (to L.-P.H., G.O., P.K., E.B. and G.R.S.), Senior Investigator Award (to G.O.), Clinical Research Network (to G.O.), Schmidt Futures (G.R.S.), Health Protection Research Unit in Respiratory Infections (NIHR200927/WHRG_P82523 to P.J.M.O.), NIHR Senior Investigator Award (NIHR201385/WHRR P84026 to P.J.M.O.), Imperial College Biomedical Research Centre (IS-BRC-1215-20013 to P.J.M.O.) and National Institute of Allergy and Infectious Diseases (Consortium for HIV/AIDS Vaccine Development UM1 AI 144371 to P.B. and A.M. and R01 AI 118549 to P.B.). L.T., P.K. and P.Simmonds. are supported by the NIHR Health Protection Research Unit (HPRU) in Emerging and Zoonotic Infections (NIHR200907) at the University of Liverpool in partnership with Public Health England (PHE), in collaboration with the Liverpool School of Tropical Medicine and University of Oxford. L.T. is based at the University of Liverpool. P.K. and P. Simmonds. are based at the University of Oxford. L.T., T.d.S. and A.A.A. are supported by the Wellcome Trust (grant numbers 205228/Z/16/Z to L.T., 110058/Z/15/Z to T.d.S. and 206377 to A.A.A.). G.R.S. is supported as a Wellcome Trust Senior Investigator (grant 095541/A/11/Z). P.B. and A.M. are Jenner Institute investigators. This work uses data provided by patients and collected by the NHS as part of their care, and supports the Data Saves Lives initiative. The views expressed are those of the authors and not necessarily those of the Department of Health and Social Care, DID, NIHR, MRC, Wellcome Trust or PHE.
Footnotes
Author contributions
T.D. and G.O. conceptualized the project. T.D. and Y.P. designed and supervised the T cell experiments. J.M. and G.R.S. designed the antibody experiments. Y.P., G.L., X.Y., Z.Y. and D.D. performed all of the T cell experiments. W.D., J.M., P.Supasa., C.L., C.L.-C., J.S.-c., Y.Zhao., D.I.S., G.C.P., J.M.G., A.A.A., O.W.B., D.E.D.P.H., B.W. and D.-S.K. performed the spike, RBD and NP EPT experiments. T.R. performed the HLA typing. J.C.K., A.J.M., T.L., R.L., P.K., T.d.S., M.G.S., C.P.C., S.C.M., J.K.B. and P.J.M.O. established the clinical cohorts and collected the clinical samples and data. K.S., P.T., P.Z., C.D., J.R., P. Simmonds., P.Sopp., D.W., U.R., Y.-L.C., W.P., P.B., J.W.F., N.F.S., M.A.A., S.D., M.S., G.N., E.B., G.K., P.G., Y.Zhang., R.J. and L.-P.H. provided the critical reagents and technical assistance. Y.P., G.L., X.Y., Z.Y., D.D., W.D., P.Z. and J.M. analyzed the data. T.D. wrote the original draft. G.O., J.C.K., A.M., A.J.M., P.B., P.K., P.J.M.O., L.T., G.R.S., R.J.C., P.S., M.G.S., B.M.K. and C.P.C. reviewed and edited the manuscript and figures.
Competing interests The authors declare no competing interests.
Data availability
Data relating to the findings of this study are available from the corresponding author upon request. Source data are provided with this paper.
References
- 1.Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. doi: 10.1007/978-1-4939-2438-7_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Perlman S, Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat Rev Microbiol. 2009;7:439–450. doi: 10.1038/nrmicro2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xu Z, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guan WJ, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yu J, et al. DNA vaccine protection against SARS-CoV-2 in rhesus macaques. Science. 2020;369:806–811. doi: 10.1126/science.abc6284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhu FC, et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, openlabel, non-randomised, first-in-human trial. Lancet. 2020;395:1845–1854. doi: 10.1016/S0140-6736(20)31208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Van Doremalen N, et al. ChAdOx1 nCoV-19 vaccination prevents SARS-CoV-2 pneumonia in rhesus macaques. Nature. 2020 doi: 10.1038/s41586-020-2608-y. [DOI] [PubMed] [Google Scholar]
- 8.Folegatti PM, et al. Safety and immunogenicity of the ChAdOx1 nCoV- 19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, singleblind, randomised controlled trial. Lancet. 2020;396:P467–P467. doi: 10.1016/S0140-6736(20)31604-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.John AL, Rathore APS. Adaptive immune responses to primary and secondary dengue virus infections. Nat Rev Immunol. 2019;19:218–230. doi: 10.1038/s41577-019-0123-x. [DOI] [PubMed] [Google Scholar]
- 10.Huang C, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liao M, et al. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020;26:842–844. doi: 10.1038/s41591-020-0901-9. [DOI] [PubMed] [Google Scholar]
- 12.Chen Y, et al. The novel severe acute respiratory syndrome coronavirus 2(SARS-CoV-2) directly decimates human spleens and lymph nodes. medRxiv. 2020 doi: 10.1101/2020.03.27.20045427. Preprint at. [DOI] [Google Scholar]
- 13.Diao B, et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19) Front Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pereira BI, et al. Sestrins induce natural killer function in senescentlike CD8+T cells. Nat Immunol. 2020;21:684–694. doi: 10.1038/s41590-020-0643-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ni L, et al. Detection of SARS-CoV-2-specific humoral and cellular immunity in COVID-19 convalescent individuals. Immunity. 2020;52:971–977.:e3. doi: 10.1016/j.immuni.2020.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hayward AC, et al. Natural T cell-mediated protection against seasonal and pandemic influenza. Results of the Flu Watch cohort study. Am J Respir Crit Care Med. 2015;191:1422–1431. doi: 10.1164/rccm.201411-1988OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grifoni A, et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell. 2020;181:1489–1501.:e15. doi: 10.1016/j.cell.2020.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Le Bert N, et al. SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature. 2020 doi: 10.1038/s41586-020-2550-z. [DOI] [PubMed] [Google Scholar]
- 19.Wilkinson TM, et al. Preexisting influenza-specific CD4+T cells correlate with disease protection against influenza challenge in humans. Nat Med. 2012;18:274–280. doi: 10.1038/nm.2612. [DOI] [PubMed] [Google Scholar]
- 20.Ahmed SF, Quadeer AA, McKay MR. Preliminary identification of potential vaccine targets for the COVID-19 coronavirus (SARS-CoV-2) based on SARS-CoV immunological studies. Viruses. 2020;12:254. doi: 10.3390/v12030254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ogg GS, et al. Four novel cytotoxic T-lymphocyte epitopes in the highly conserved major homology region of HIV-1 Gag, restricted through B*4402, B*1801, A*2601, B*70 (B*1509) AIDS. 1998;12:1561–1563. doi: 10.1097/00002030-199812000-00026. [DOI] [PubMed] [Google Scholar]
- 22.Shomuradova AS, et al. SARS-CoV-2 epitopes are recognized by a public and diverse repertoire of human T-cell receptors. medRxiv. 2020 doi: 10.1101/2020.05.20.20107813. Preprint at. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sekine T, et al. Robust T cell immunity in convalescent individuals with asymptomatic or mild COVID-19. Cell. 2020 doi: 10.1016/Jcell.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee LY, et al. Memory T cells established by seasonal human influenza A infection cross-react with avian influenza A (H5N1) in healthy individuals. J CliN Invest. 2008;118:3478–3490. doi: 10.1172/JCI32460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Braun J, et al. Presence of SARS-CoV-2 reactive T cells in COVID-19 patients and healthy donors. Nature. 2020 doi: 10.1038/s41586-020-2598-9. [DOI] [PubMed] [Google Scholar]
- 26.Li CK, et al. T cell responses to whole SARS coronavirus in humans. J Immunol. 2008;181:5490–5500. doi: 10.4049/jimmunol.181.8.5490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Powell TJ, et al. Identification of H5N1-specific T-cell responses in a high-risk cohort in Vietnam indicates the existence of potential asymptomatic infections. J Infect Dis. 2012;205:20–27. doi: 10.1093/infdis/jir689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dong T, et al. Extensive HLA-driven viral diversity following a narrow-source HIV-1 outbreak in rural China. Blood. 2011;118:98–106. doi: 10.1182/blood-2010-06-291963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Su LF, Davis MM. Antiviral memory phenotype T cells in unexposed adults. Immunol Rev. 2013;255:95–109. doi: 10.1111/imr.12095. [DOI] [PubMed] [Google Scholar]
- 30.Campion SL, et al. Proteome-wide analysis of HIV-specific naive and memory CD4+T cells in unexposed blood donors. J Exp Med. 2014;211:1273–1280. doi: 10.1084/jem.20130555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sette A, Crotty S. Pre-existing immunity to SARS-CoV-2: the knowns and unknowns. Nat Rev Immunol. 2020;20:457–458. doi: 10.1038/s41577-020-0389-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pizzolla A, et al. Resident memory CD8+T cells in the upper respiratory tract prevent pulmonary influenza virus infection. Sci Immunol. 2017;2:eaam6970. doi: 10.1126/sciimmunol.aam6970. [DOI] [PubMed] [Google Scholar]
- 33.Turner DL, et al. Lung niches for the generation and maintenance of tissue-resident memory T cells. Mucosal Immunol. 2014;7:501–510. doi: 10.1038/mi.2013.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yoshizawa A, et al. TCR-pMHC encounter differentially regulates transcriptomes of tissue-resident CD8 T cells. Eur J Immunol. 2018;48:128–150. doi: 10.1002/eji.201747174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peng Y, et al. Boosted influenza-specific T cell responses after H5N1 pandemic live attenuated influenza virus vaccination. Front Immunol. 2015;6:287. doi: 10.3389/fimmu.2015.00287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lillie PJ, et al. Preliminary assessment of the efficacy of a T-cellbased influenza vaccine, MVA-NP+M1, in humans. CliN Infect Dis. 2012;55:19–25. doi: 10.1093/cid/cis327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.De Silva TI, et al. Correlates of T-cell-mediated viral control and phenotype of CD8+T cells in HIV-2, a naturally contained human retroviral infection. Blood. 2013;121:4330–4339. doi: 10.1182/blood-2012-12-472787. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data relating to the findings of this study are available from the corresponding author upon request. Source data are provided with this paper.