Abstract
Background
Interstitial Cystitis/Bladder Pain Syndrome (IC/BPS) is a condition characterized by pelvic pain and urinary symptoms. Some IC/BPS patients have pain confined to the pelvic region, while others suffer widespread pain. Inflammatory processes have previously been linked to pelvic pain in IC/BPS, but their association with widespread pain in IC/BPS has not been characterized.
Methods
Sixty-six women meeting criteria for IC/BPS completed self-report measures of pain as part of the Multidisciplinary Approach to the Study of Chronic Pelvic Pain (MAPP), collected 3 days of saliva for cortisol assays, and provided blood samples. Peripheral blood mononuclear cells (PBMCs) were stimulated with Toll-Like Receptor (TLR) 2 and 4 agonists and cytokines were measured in supernatant; IL-6 was also measured in plasma. Associations between inflammatory variables and the likelihood of endorsing extra-pelvic pain, or the presence of a comorbid syndrome, were tested by logistic regression and General Linear Models, respectively. A subset of patients (n=32) completed Quantitative Sensory Testing.
Results
A one standard deviation increase in TLR-4 inflammatory response was associated with a 1.59 greater likelihood of endorsing extra-pelvic pain (p = .019). Participants with comorbid syndromes also had higher inflammatory responses to TLR-4 stimulation in PBMCs (p = .016). Lower pressure pain thresholds were marginally associated with higher TLR-4 inflammatory responses (p = .062), and significantly associated with higher IL-6 in plasma (p = .031).
Conclusions
TLR-4 inflammatory responses in PBMCs are a marker of widespread pain in IC/BPS, and should be explored in other conditions characterized by medically unexplained pain.
Keywords: Inflammation, Toll-Like Receptors, Functional Somatic Syndromes, Pain, Negative Affect, Interstitial Cystitis/Bladder Pain Syndrome
1. Introduction
Interstitial Cystitis/Bladder Pain Syndrome (IC/BPS) is a highly prevalent debilitating chronic condition characterized by pelvic/bladder pain and urinary symptoms such as frequency, urgency, and nocturia (Hanno et al., 2011). Additionally, IC/BPS patients have a high prevalence of psychiatric comorbidities including depression and anxiety disorders (Clemens et al., 2008). While some patients present with Hunner's ulcers, inflammatory lesions found on the wall of the bladder, approximately 90% do not (Simon et al, 1997). IC/BPS is therefore a diagnosis of exclusion, and is sometimes considered a cluster of medically unexplained symptoms.
It has been proposed that there may be distinct subtypes of IC/BPS, as some patients appear to experience pain and discomfort in the pelvic/bladder region only (i.e. local pain), while others report extra-pelvic pain consistent with somatic syndromes like fibromyalgia, Irritable Bowel Syndrome (IBS), Chronic Fatigue Syndrome (CFS), or Temporomandibular joint disorder (TMD), suggesting a condition mediated by the central nervous system. A recent investigation found that that comorbid IBS and CFS were present in 39% and 19% of IC/BPS patients, respectively (Nickel et al., 2010). These findings are consistent with the results of many studies finding high degrees of comorbidity between somatic syndromes (Wessely et al., 1999). This suggests that there may be common physiological factors that support global changes in central pain pathways and increased pain perception (Phillips & Clauw, 2011). Furthermore, IC/BPS patients with comorbid somatic syndromes (e.g. CFS) appear to be at risk of developing additional somatic syndromes in the future, suggesting a progressive element of altered pain perception in some patients (Warren et al., 2013).
Identifying markers of pain sensitization in IC/BPS may improve early phenotyping of vulnerable patients and lead to novel therapeutic targets with the potential to prevent disease progression. Furthermore, identifying markers of central sensitization in chronic pain patients may further prevent psychiatric comorbidity as chronic pain has recently been shown to induce dysfunction in the locus coeruleus and subsequent depression and anxiety like behaviors in an animal model (Alba-Delgado et al., 2013). Much research has been devoted to identifying altered mechanisms of pain perception (i.e. sensitized pathways), and whether reliable markers of these alterations can be identified. Candidate markers include changes in pain processing networks identified through functional magnetic resonance imaging (fMRI), and hyperalgesia/allodyina identified by quantitative sensory testing (QST), both of which have identified abnormal responses to stimuli in chronic pain patients, including patients with IC/BPS (Kilpatrick et al., 2014; Ness et al., 2014). Another promising biomarker is the inflammatory response to Toll-Like Receptor (TLR) stimulation in peripheral immune cells, as we have recently found these responses to be associated with heightened pelvic pain in IC/BPS (Schrepf et al., 2014).
TLRs are highly conserved receptors on sentinel immune cells that respond to both Microbe Associated Molecular Patterns (MAMPs) and Damage Associated Molecular Patterns (DAMPs; Hutchinson et al., 2009). We have recently reported that TLR-2 inflammatory responses distinguish IC/BPS patients from healthy controls, and that the magnitude of TLR-4 inflammatory responses in stimulated PBMCs are associated with the extent of painful urinary and pelvic symptoms reported by IC/BPS patients. PBMCs have been hypothesized to mark pain sensitization in humans since it was demonstrated that proliferation of PBMCs incubated with morphine is strongly associated with tolerance for noxious cold stimuli (Hutchinson et al., 2004). Additionally, we found that IC/BPS patients had higher serum levels of Interleukin (IL)-6, a marker of systemic inflammation, and altered diurnal cortisol patterns (Schrepf et al., 2014). These finding echo a recent investigation that found that TLR-2 and TLR-4 inflammatory responses in PBMCs differentiate chronic pain patients from healthy controls (Kwok et al., 2012) and other work identifying altered TLR inflammatory responses as features of other conditions characterized by persistent pain such as Inflammatory Bowel Disease and Rheumatoid arthritis (Kovarik et al., 2011; Kowalski et al., 2008). However, it is unknown if inflammatory responses in PBMCs can differentiate subtypes of painful syndromes such as IC/BPS, particularly those characterized by pain not typically considered part of the IC/BPS syndrome (i.e. widespread, extra-pelvic pain.).
The purpose of the current study was to determine if inflammatory processes, especially TLR-2 and TLR-4 inflammatory responses in PBMCs, are differentially associated with pelvic vs. extra-pelvic pain in IC/BPS. Additionally, we examined relationships between TLR-mediated inflammation, pain intensity/interference with daily life, and pressure pain sensitivity determined by QST. We also examined the relationship between TLR-mediated inflammation and the presence of comorbid pain conditions in IC/BPS patients, as these conditions are characterized by pain outside the pelvic region, and contribute substantially to the difficulty of treating the IC/BPS syndrome (Nickel et al., 2010). In line with the results of our earlier work and results from animal models of chronic pain, we hypothesized that greater TLR inflammatory responses in PBMCs would be associated with pain outside the pelvic region, increased pain sensitivity by QST, and comorbid somatic syndromes.
2. Methods
2.1 MAPP Study and Recruitment
The Multidisciplinary Approach to the Study of Chronic Pelvic Pain (MAPP) is a National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) sponsored research initiative comprising several sites with the objective of characterizing the epidemiology, symptom trajectories, phenotypes and biological correlates of chronic pelvic pain (Clemens et al. 2014). The University of Iowa is a participating institution emphasizing biomarker research. Participants were eligible if they were at least 18 years of age, female, not pregnant, and reported chronic pain/pressure/discomfort associated with the bladder or pelvic region in the preceding three months. Participants had negative urine cultures for uropathogens. Exclusion criteria included conditions which might result in tissue damage to areas relevant to IC/BPS symptomology (e.g. history of urethral stricture, neurological disorder affecting the bladder or bowel). Additional information about the MAPP project, including patient characterization, study aims, and full exclusion criteria is available (Clemens et al., 2014; Landis et al., 2014; Schrepf et al., 2014).
2.2 Demographic and Symptom Information
The sample was composed of an expanded group of participants from a previously reported study (Schrepf et al. 2014) who provided additional information on pain and comorbid syndromes. In addition, a subsample completed QST. Sixty-six women provided demographic information at the time of eligibility screening, including information about income, education, employment, race and ethnicity. Upon study entry, participants had a blood draw, urine collection, physical examination and completed a battery of questionnaires relating to pain and urological symptoms. These included the Brief Pain Inventory (BPI), a measure of pain intensity, interference with daily life, and a body map for selection of painful areas (Cleeland & Ryan, 1994) which has previously been validated in chronic pain populations (Tan et al., 2004). The body map was modified so that patients could select regions where pain was experienced from a standardized form of 45 distinct areas. Participants were also administered self-report screens to assess the presence of comorbid somatic syndromes. These included the Rome III criteria for IBS (Drossman & Dumitrascu, 2006), the American College of Rheumatology diagnostic criteria for Fibromyalgia (Wolfe et al., 2010), International Chronic Fatigue Syndrome Study Group criteria for CFS (Fukuda et al., 1994), an 8 question MAPP specific diagnostic tool for symptoms of vulvodynia (e.g. “experience constant burning or raw feeling at the opening of the vagina,”) and the Research Diagnostic Criteria for TMD (Dworkin et al., 2002). These diagnostic criteria show adequate reliability and validity (Dworkin et al., 2002; Ford et al., 2013; Komaroff et al., 1996; Wolfe et al., 2010;) excepting the criteria for vulvodynia, which is necessarily exploratory. Additionally, participants completed the reliable and validated Positive and Negative Affect Scale (PANAS; Watson et al. 1988). Use of non-steroidal anti-inflammatory drugs (NSAIDs), tricyclic antidepressants, opioids, selective serotonin/norepinephrine reuptake inhibitors (SSRI/SNRI), and pentosan polysulfate, and duration of symptoms in years, were collected by patient self-report.
2.3 Cortisol
Salivary cortisol was collected in salivettes by participants at 3 time points (upon waking: 4-9am, afternoon: 5 pm, and bedtime: 8pm-12am) for three consecutive days prior to the baseline visit. Samples collected outside this time frame were excluded to maintain homogeneity. Individual waking and bed times have been demonstrated to better approximate diurnal cortisol rhythms than scheduled collection times (Kraemer et al., 2006). Participants were instructed not to eat, exercise or consume caffeine for thirty minutes prior to collecting a sample. Participants wrote the time of each collection on the salivette tubes. Self-report of collection time has been demonstrated to be reliable and salivary cortisol is stable at room temperature (Kraemer et al., 2006). Salivettes were analyzed by chemiluminescence immunoassay (IBL, Hamburg, Germany) at the Technical University of Dresden. The lower detection limit is 0.41 nmol/L and inter-assay and intra-assay coefficients of variance are less than 10%.
2.4 Inflammatory Measures
Blood samples were collected between approximately 11:30am and 12:30pm. PBMCs were separated by Ficoll-paque gradient centrifugation within 30 minutes of blood collection and cultured in RPMI 1640 medium containing 10% fetal bovine serum, 100 U/ml penicillin and 100 ug/ml streptomycin for 3 days with TLR agonists at 37°C in a humidified incubator with 5% CO2. TLR-2 and TLR-4 agonists were selected on the basis of the role of these receptors in chronic pain in animal models and our previous findings linking them to symptoms in IC/BPS (Bastos et al., 2013; Hutchinson et al., 2008; Schrepf et al., 2014;). For stimulation of TLR-4, 50 ng/ml of Lipopolysaccharide (LPS) was used; for TLR-2 stimulation, 0.04 ng/ml of Staphylococcus aureus Cowan I (SAC) was used. Conditioned media was then harvested and frozen at −80°C prior to batch ELISA analysis. Each well contained 1×106 cells in 24 well plates, with one well per subject for TLR-4 and TLR-2 stimulation. Cytokines IL-6 and IL-1β were assayed in duplicate by DuoSet ELISAs (R&D Systems) according to instructions included with the kit. Plasma IL-6 was assayed with a high sensitivity Quantikine ELISA (R & D Systems).
2.5 Quantitative Sensory Testing
A subsample of participants completed the MAPP network QST protocol. Pressure pain sensitivity was evaluated at the thumbnail (As-Sanie et al., 2013; Giesecke et al., 2004; Petzke et al., 2001) using the Multimodal Automated Sensory Testing (MAST) system (Harte et al., 2013). The MAST system consists of a control computer that executes testing algorithms and stores testing data, and a touch-screen interface for participant feedback. Computer-controlled pressure stimuli are applied to the thumbnail bed via a 1 cm2 rubber probe housed within a wireless, pistol-grip style handset. Probe movement is driven by a miniature servo-motor. A closed-looped control system measures applied pressures and dynamically self-adjusts motor output to the resistance of the thumb and any movement to ensure accurate and repeatable force delivery.
Participants received scripted instructions, and MAST system familiarization and practice testing prior to data collection. During testing, an ascending series of 5-s duration pressures were delivered at a rate of 4 kg/cm2/s to the dominant thumbnail beginning at 0.50 kg/cm2 and increasing in 0.50 kg/cm2 steps, with a minimum inter-stimulus interval of 20 s. Pain intensity was rated after each stimulus on a 0-100 numerical rating scale (NRS) displayed on the interface screen (0 = no pain; 100 = worst pain imaginable). Testing terminated when the first of three possible stop conditions were met: 1) participant reached her personal pain tolerance (i.e., requested to stop the test), 2) patient reported a pain intensity rating of ≥ 80/100, or 3) the maximum pressure of 10 kg/cm2 was delivered. A modified three-parameter logistic model was used to fit the stimulus-response data obtained from this procedure. The midpoint between the minimum and maximum stimulus intensity was estimated within-person using the SAS NLIN procedure to derive an overall measure of supra-threshold pressure pain sensitivity, referred to as Pain50. Additional outcome variables included pressure pain threshold, defined as the first pressure in a string of at least two consecutive pressures that elicited a NRS pain rating > 0, and pressure pain tolerance, defined as the last pressure recorded in the stimulus response profile.
2.6 Data Analysis
Statistical analyses were performed using SPSS v. 21 and R v. 3.1.1. Cytokine values were log-10 transformed, and cortisol values natural log transformed, to normalize their distribution. The Inflammatory response scores for stimulated cytokines were calculated by summing the z-scores ([individual score- group mean]/ group standard deviation) for the IL-6 and interleukin-1 beta (IL-1β) response in PBMCs following stimulation with LPS (TLR-4) or SAC (TLR-2. This inflammatory response score was then standardized for ease of interpretation. Both IL-6 and IL-1β have been implicated in enhanced pain processing when released by spinal glia, and both are released following TLR-2 and 4 stimulation, in part, by transcription of nuclear factor-kappaB (NFκB; Milligan & Watkins, 2009). The composite score, therefore, is likely more reflective of the inflammatory response to TLR stimulation than either cytokine alone. Distributions of transformed variables were examined for confirmation of normality. Salivary cortisol values at each of the collection points were regressed on the time of collection over the three-day period to calculate cortisol slope, a measure of the average hourly decrease in cortisol over the course of the day as described previously (Kraemer et al., 2006).
To determine if inflammatory variables were associated with a greater likelihood of endorsing extra-pelvic sites as painful, mixed-effects logistic regression models were used. Higher probabilities of endorsing pain outside the pelvic region reflect pain not typically considered part of the IC/BPS syndrome. Thus, endorsement of pain at any of the 44 sites outside the pelvic region was considered indicative of extra-pelvic pain. In these models the dependent variable of interest was the probability of a patient selecting any extra-pelvic site (44 sites) as painful, with inflammatory variables as predictors. Subject and site specific intercepts were tested as random effects, with the maximum random effects structure retained by likelihood testing. Modeling subject specific variance (e.g. if some patients are more likely to endorse any site as painful) and site-specific variance (e.g. if lower back pain is more likely to be endorsed than pain in the hands across subjects) can allow more accurate estimation of fixed effects (Baayen et al., 2008). Random intercept terms were retained for subject and pain site. BMI, age, use of medications, presence of a comorbid condition, and inflammatory variables were used in univariate analyses to determine which, if any variables, were associated with a greater likelihood of endorsing extra-pelvic pain. Significant variables (p < .05) were retained in multivariate analyses. Relationships between pain severity/interference (from the BPI) and inflammation were tested in multivariate General Linear Models with the same set of covariates. To explore the relationships between inflammatory variables (e.g. TLR-4 inflammatory responses and cortisol slope) and duration of symptoms in years Pearson correlations were used.
Group differences between IC/BPS only and IC/BPS comorbid patients (IBS, CFS, fibromyalgia, TMD, vulvodynia) with respect to inflammatory variables were tested with one-way ANOVAs, and between IC/BPS only and IC/BPS comorbid patients with individual conditions (IBS, fibromyalgia, CFS, TMD and vulvodynia). The association between number of comorbid conditions and inflammatory variables was tested by Spearman's Rank correlations. Relationships between inflammatory variables and pain intensity and interference were assessed using General Linear Models controlling for negative affect, comorbid condition status, and use of SSRI/SSNIs, following the results of the univariate analyses. Due to the non-normal distribution of QST Spearman's rank correlation tests were used to test the association with inflammatory variables.
3. Results
3.1 Demographic Characteristics and Covariates
Participants were on average approximately 42 years old (range 20-74), and the vast majority were non-Hispanic and white. See Table 1. Higher TLR-4 inflammatory responses were associated with a greater likelihood of endorsing extra-pelvic pain (p= .006). SSRI/SNRI use was associated with a greater likelihood of endorsing extra-pelvic pain (p < .001). Tricyclic antidepressant use was marginally associated with a lower likelihood of endorsing extra-pelvic pain (p = .081) whereas older age was marginally associated with a greater likelihood of endorsing extra-pelvic pain (p =.071). Greater negative affect and presence of a comorbid condition were both associated with a greater likelihood of endorsing extra-pelvic pain (both p <.001). BMI, opioid use, NSAID use, Pentosan Polysulfate use, cortisol slope, and duration of symptoms in years were not associated with the likelihood of endorsing extra-pelvic pain (all p > .15; univariate analyses for covariate selection not shown). Additionally, the inflammatory response to TLR-2 stimulation was not associated with a greater likelihood of endorsing pain outside the pelvic region (Odds Ratio = 1.03, 95% CI = .72, 1.49, p = .86). Therefore, multivariate analyses included comorbid status, SSRI/SNRI use, and negative affect in addition to the TLR-4 inflammatory response.
Table 1.
Participant Characteristics.
| Participant Characteristics | N=66 |
|---|---|
| Age Mean(SD) | 42.03 (15.12) |
| BMI Mean(SD) | 27.18 (5.65) |
| PANAS negative affect | 22.27 (8.56) |
| Race % (n) | |
| White | 97 (64) |
| Asian | 1 (1) |
| Multi Race | 1 (1) |
| Ethnicity % (n) | |
| Non-Hispanic | 98 (65) |
| Hispanic | 1 (1) |
| Education % (n) | |
| High School or GED | 13 (9) |
| Some College | 27 (18) |
| Graduated College | 32 (21) |
| Graduate Degree | 27 (18) |
| Employment % (n) | |
| Employed | 62 (41) |
| Unemployed | 11 (7) |
| Disabled | 8 (5) |
| Retired | 9 (6) |
| Full Time Homemaker | 9 (6) |
| Not Answered | 1 (1) |
| Annual Income/$ % (n) | |
| <10,000 | 14 (9) |
| <25,000 | 6 (4) |
| <50,000 | 23 (15) |
| <100,000 | 33 (22) |
| >100,000 | 20 (13) |
| Prefer not to answer | 5 (3) |
| Comorbid Conditions % (n) | |
| None | 39 (26) |
| Irritable Bowel Syndrome | 42 (28) |
| Fibromyalgia | 3 (2) |
| Chronic Fatigue Syndrome | 12 (8) |
| TMD | 35 (23) |
| Vulvodynia | 21 (14) |
| Number of Comorbid Conditions % (n) | |
| 0 | 39 (26) |
| 1 | 26 (17) |
| 2 | 23 (15) |
| 3 | 6 (4) |
| 4 | 6 (4) |
| Tricyclic anti-depressants | |
| No | 56 (37) |
| Yes | 44 (29) |
| Opioids | |
| No | 83 (55) |
| Yes | 17 (11) |
| Pentosan Polysulfate | |
| No | 56 (37) |
| Yes | 44 (29) |
| NSAIDs | |
| No | 89 (59) |
| Yes | 11 (7) |
| SSRI/SNRIs | |
| No | 86 (57) |
| Yes | 14 (9) |
IC/BPS=Interstitial Cystitis/Bladder Pain Syndrome. BMI=Body Mass Index. NSAID= non-steroidal anti-inflammatory drug. SSRI/SNRI = selective serotonin reuptake inhibitor/serotonin-norepinephrine reuptake inhibitor. TMD = temporomandibular disorders.
3.2 TLR-4 Inflammatory Response and Pain
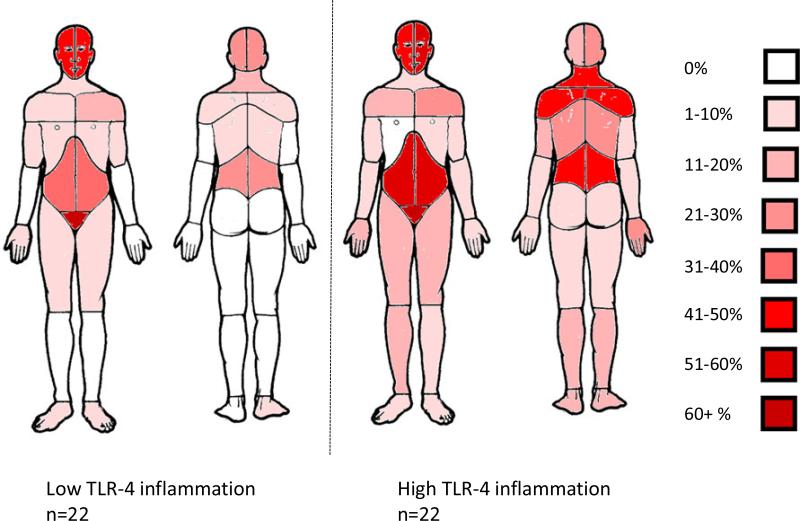
In multivariate analyses, a one standard deviation increase in the TLR-4 inflammatory response was associated with 1.59 greater odds (95% CI = 1.08, 2.33) of endorsing pain outside the pelvic area on the body map (p = .019), controlling for negative affect, comorbid status, and SSRI/SNRI use. This one standard deviation increase in TLR-4 inflammatory response corresponds to a 63% increase in the likelihood of a participant endorsing pain outside the pelvic region at any site on the body map.1 See Table 2. Figure 1 illustrates the higher likelihood of endorsing extra-pelvic pain among those with higher TLR-4 inflammatory response. The TLR-4 inflammatory score was associated with greater self-report of pain severity (p = .013) and pain interference (p = .046) on the BPI, controlling for negative affect, comorbid status, and SSRI/SNRI use. See Table 3. Additionally, the TLR-4 inflammatory response was associated with higher TLR-2 inflammatory responses (r = .304, p = .013) and longer duration of symptoms in years (r = .295, p=.017) but not with cortisol slope (r = .165, p = .23 (n=56) or plasma IL-6 (r = .064, p = .61).
Table 2.
Relationship of TLR-4 inflammatory response and covariates (logistic regression) with extra-pelvic pain.
| Outcome Measure | Predictor | Est. | S.E. | Z value | Odds Ratio 95% CI | p |
|---|---|---|---|---|---|---|
| Extra-Pelvic Pain | ||||||
| TLR-4 inflammation response (one SD above mean) | .46 | .20 | 2.34 | 1.59 1.08, 2.33 |
.019 | |
| Use of SSRI/SNRI | .41 | .57 | .73 | 1.51 .50, 4.59 |
.47 | |
| Comorbid Condition | .39 | .43 | .92 | 1.48 .64, 4.59 |
.36 | |
| PANAS negative affect (one SD above mean) | .66 | .21 | 3.12 | 1.94, 1.28, 2.95 | .002 |
BPI= Brief Pain Inventory. IL-6=Interleukin-6. PANAS=Positive and Negative Affect Scale. TLR=Toll-Like Receptor.
Figure 1.
Prevalence of pain reported in the first tertile (low) of the TLR-4 composite inflammatory score versus the third tertile (high) for each site on the body map.
Table 3.
General Linear Models testing relationship of TLR-4 inflammatory response and covariates with BPI symptom scores.
| Outcome Measure | Predictor | B. | S.E. | F1,65 | 95% CI | p |
|---|---|---|---|---|---|---|
| Pain Severity (BPI) | ||||||
| TLR-4 inflammatory response | .503 | .197 | 6.53 | .109,.896 | .013 | |
| Comorbid condition | .204 | .429 | .23 | −1.063, .655 | .64 | |
| Use of SSRI/SNRI | −.156 | .615 | .06 | −1.075, 1.386 | .97 | |
| PANAS negative affect | .096 | .025 | 14.83 | .046,.145 | <.01 | |
| Pain Interference (BPI) | ||||||
| TLR-4 inflammatory response | .567 | .278 | 4.17 | .012, 1.123 | .046 | |
| comorbid condition | .200 | .607 | .50 | −1.414, 1.014 | .74 | |
| Use of SSRI/SNRI | −.028 | .832 | .01 | −1.635, 1.691 | .97 | |
| PANAS negative affect | .171 | .035 | 23.67 | .101,.241 | <.01 |
BPI= Brief Pain Inventory. IL-6=Interleukin-6. PANAS=Positive and Negative Affect Scale. TLR=Toll-Like Receptor.
3.3 QST
In the subsample of 32 patients (13 IC/BPS only, 19 IC/BPS comorbid) who underwent QST, higherTLR-4 inflammation scores were marginally associated with lower pressure pain thresholds (Spearman's Rho= −.334, p = .062) and increased levels of IL-6 in plasma were significantly associated with lower pressure pain thresholds (Spearman's Rho = −.381, p = .031). In contrast, the TLR-2 inflammation score was not associated with pressure pain thresholds (p =.30), nor was cortisol slope (p = .84). Pain50 and pressure pain tolerance were not associated with inflammatory variables (all p > .19).
3.4 Inflammatory Measures and Comorbid Conditions
The TLR-4 inflammatory response was significantly greater in IC/BPS comorbid patients (p = .016) compared to the IC/BPS patients without comorbid conditions. Additionally, a higher TLR-4 inflammatory response distinguished IC/BPS + IBS patients (n=28, p = .018), IC/BPS + TMD (n=23, p = .007), and IC + vulvodynia (n=14, p = .049), from IC/BPS only participants (n=26). The TLR-4 inflammatory response was higher in IC/BPS + CFS participants (n=8) and IC/BPS + fibromyalgia participants (n=2) but not significantly so (both p > .18), possibly due to small sample sizes. IL-1β measured in supernatant of LPS (TLR-4) stimulated PBMCs was significantly higher in IC/BPS patients with comorbid conditions (p = .008) whereas IL-6 was not significantly elevated (p = .21). IL-1β and IL-6 measured in the supernatant of SAC (TLR-2)-stimulated PBMCs did not differ significantly between groups (both p > .20) nor did the TLR-2 composite inflammatory score (p = .50). IL-6 measured in plasma was marginally higher in IC/BPS comorbid participants (p = .097, while cortisol slopes did not differ between groups (p = .96). See Table 4. Greater numbers of comorbid syndromes were associated with a greater TLR-4 inflammatory response (Spearman's Rho = .311, p = .011). No other inflammatory variable was associated with number of comorbid conditions or presence of individual comorbid conditions (all p >.10).
Table 4.
Means, standard deviations and 95% confidence intervals of biomarkers in participants with IC/BPS only and those with additional comorbid conditions (Fibromyalgia, Irritable Bowel Syndrome, vulvodynia, Chronic Fatigue Syndrome, Temperomandibular Disorder)
| Variable Mean (S.D.) 95% CI Inflammatory Variables | IC Only n=26 | IC Comorbid n=40 | p |
|---|---|---|---|
| TLR-4 inflammatory response | −.36 (1.13) −.82, .09 |
.24 (.85) −.03, .51 |
.016 |
| IL-1β + LPS | 3.11 (.93) 2.73,3.48 |
3.58 (.44) 3.44, 3.72 |
.008 |
| IL-6 + LPS | 4.15 (.40) 3.99, 4.31 |
4.28 (.42) 4.14, 4.41 |
.205 |
| TLR-2 inflammatory response | −.10 (1.06) −.53, .32 |
.07 (.97) −.24, .38 |
.501 |
| IL-1β + SAC | 1.17 (1.19) .69, 1.65 |
1.44 (1.14) 1.08, 1.81 |
.365 |
| IL-6 + SAC | 1.68 (1.37) 1.13, 2.24 |
1.79 (1.22) 1.40, 2.19 |
.719 |
| Cortisol Slope (ln transformed) |
n=22 −.11 (.06) −.14, −.08 |
n=34 −.11 (.07) −.13, −.08 |
.955 |
| IL-6 plasma (log10 transformed) | .33 (.35) −.75, 0.84 |
.46 (.26) .00, .97 |
.097 |
| Duration of Symptoms (years) | 6.91 (7.48) 3.89, 9.93 |
8.3 (8.00) 5.71, 10.90 |
.48 |
IL-6=Interleukin-6. IL-1β=Interleukin-1 beta. LPS= lipopolysaccharide. SAC= Staphylococcus aureus Cowan. TLR = Toll-Like Receptor.
4.1 Discussion
The key finding of this study is that TLR-4-mediated inflammatory responses in PBMCs are associated with extra-pelvic pain in a chronic pelvic pain population. This is demonstrated in the association between LPS evoked inflammation and an increasing likelihood of endorsing pain outside the pelvic region, and by the ability of this inflammatory response to distinguish between patients meeting criteria for IC/BPS only and patients meeting criteria for IC/BPS who also had comorbid syndromes. Use of medications was not associated with measures of pain, while greater negative affect was strongly associated with pain measures independent of TLR-4 inflammation. While exploratory, QST data suggest that TLR-4 inflammation may also be associated with lower pressure pain thresholds measured at a non-symptomatic site remote from the pelvic region (i.e., the thumb), further suggesting a central mechanism of pain hypersensitivity in this population. These findings build on our previous finding that TLR-4 inflammation is associated with pelvic pain in IC/BPS (Schrepf et al., 2014) by demonstrating that TLR-4 inflammation is associated with comorbid pain not typically considered part of the IC/BPS syndrome.
This is the first study to our knowledge which has shown TLR-mediated inflammation to be associated with comorbid pain in a chronic pain population. Further, these differences in TLR-4 mediated inflammation were not associated with a particular comorbid condition, as each condition that was well-represented in our sample (IBS, TMD, and vulvodynia) was characterized by higher TLR-4 mediated inflammation compared to patients with IC/BPS only. This suggests that TLR-4 inflammation may reflect a broad mechanism by which pain signaling is enhanced in IC/BPS. This extends our earlier finding that TLR-4 inflammation predicts non-specific pain severity and frequency of pelvic pain symptoms in IC/BPS but was not associated with pain in particular anatomical regions or during particular activities. A recent investigation in an animal model of visceral pain found that TLR-4 regulates stress-induced pain (Tramullas et al., 2014). This is notable given the high prevalence of IBS in IC/BPS patients, and because stressful events frequently precipitate symptom flares (Rothrock et al., 2001). QST data indicating lower pressure pain thresholds (increased pain sensitivity) in patients with higher TLR-4 inflammatory responses and higher IL-6 in blood suggest that these inflammatory processes are related to global pain sensitivity, not pain associated with IC/BPS only, and are consistent with other findings that higher pro-inflammatory cytokines in blood are associated with altered pain sensitivity on QST in osteoarthritis patients (Lee et al., 2011). While the presence of a comorbid syndrome was associated with an increased likelihood of endorsing extra-pelvic pain in a univariate model, this was no longer true in the multivariate model including TLR-4 inflammation and negative affect. This provides further evidence that TLR-4 inflammation may play a critical role in the painful symptoms associated with comorbid conditions in IC/BPS, though it is not possible to determine from these results what role, if any, TLR-4 inflammation may play in centrally mediated pain sensitization.
The fact that TLR-2 stimulation distinguished patients from comparison participants in our previous study, but did not distinguish between patients with and without comorbid syndromes, is of interest. One possibility is that hypersensitivity to TLR-2 is an early or universal feature of IC/BPS while TLR-4 sensitivity is not. Previous research has demonstrated that higher TLR-2 density on PBMCs, but not TLR-4 density, distinguishes IBS patients from controls (Ohman et al., 2012). Another recent study found that TLR-2 and TLR-4 mRNA were up-regulated in the colonic mucosal tissue of IBS patients with heterogeneous symptom presentation compared to those with symptoms of diarrhea or constipation only (Belmonte et al. 2012). It is unknown if TLR-4 density on PBMCs or other tissue may differ between IC/BPS patients with and without comorbid conditions, or if TLR-4 responses are heightened in patients with comorbid syndromes due to a “priming” effect of prior MAMP or DAMP exposure. Another possibility is that intracellular signaling and subsequent cytokine production may differ significantly following TLR-4 vs. TLR-2 stimulation; a recent investigation of stimulated human whole blood found that both TLR-2 and TLR-4 stimulation resulted in NF-κB family activity, but that a distinct IFN upregulation occurred following TLR-4 stimulation only (Blankley et al., 2014). Another recent investigation used principal component analysis to analyze various cytokine and chemokine responses to both TLR-2 and TLR-4 receptor stimulation in human whole blood; the results indicated that different TLR agonists (including TLR-2 and TLR-4), evoked distinct protein signatures, suggesting divergent intracellular signaling pathways (Duffy et al., 2014). Thus it appears that though TLR2 and 4 ligands may signal through the same receptor, each cytokine is capable of eliciting unique signaling patterns. Another possibility is that unexplored vulnerabilities (e.g. genetic factors) could mask the relationship between TLR-2 mediated inflammation and pain, if such a relationship exists. Clearly, more research is needed to identify relevant cellular and intracellular differences in TLR-4 vs. 2 responses in the context of IC/BPS.
Microglia and astrocytes express TLRs including TLR-2 and TLR-4, and stimulation of TLR-4 on microglia induces release of pro-inflammatory cytokines IL-6, IL-1β- and TNF-alpha in the spinal cord (Milligan & Watkins, 2009). TLR-4-mediated inflammation released by glia cells in the dorsal horn of the spinal cord is thought to be one contributing mechanism for central pain amplification in rodent models of chronic pain (Ellis et al., 2014; Grace et al., 2014; Hutchinson et al., 2008), though this effect may be sex specific, as recent work suggests that LPS promotes hyperalgesia when delivered to the brain or periphery, but not the spinal cord, in female mice (Sorge et al. 2011). Regardless, TLR-4 is essential in LPS induced hyperalgesia as LPS injection fails to promote hyperalgesia in TLR-4 deficient mice (Mattioli et al., 2014). Importantly for this study, the amplification of pain signaling sometimes involves extension of pain from the original site of injury, termed “extra-territorial” pain (Wieseler-Frank et al., 2005). Whether circulating PBMCs reflect neuro-inflammatory processes remains an open question; recent work suggests that TLR-mediated inflammation in PBMCs corresponds to the same TLR-mediated inflammation in the spinal cord, in a rodent model of chronic pain (Kwok et al., 2013). However, relevant animal models of IC/BPS will need to be developed before concordance between inflammatory responses in PBMCs and spinal microglia can be formally tested.
While rodent models that investigate the role of TLRs in pain have typically used neuropathic injury models (e.g. sciatic constriction), studies of human pain populations with little evidence of peripheral tissue damage have also identified differential responses to TLR stimulation in PBMCs (Kowalski et al., 2008; Schrepf et al., 2014). If sensitization of TLR-induced inflammation is a mechanism for pain amplification in IC/BPS, this raises the question of what the initiating events may be in this population, given the generally low proportion of patients with evidence of peripheral tissue damage. One large twin study implicated both genetic factors (approximately one third) and non-shared environmental factors (approximately two thirds) in the risk of IC/BPS (Altman et al., 2011). At least two large studies have identified an increased number of antecedent urogenital infections as a risk factor for IC/BPS in women raising the possibility that recurrent or severe infections might serve as initiating events in IC/BPS (Díaz Mohedo et al., 2014; Li et al. 2010). TLRs including TLR-4 are pattern recognition receptors that respond to PAMPs and DAMPs; they play a critical role in expelling bacteria from the urinary tract and are expressed on both bladder epithelial cells and phagocytic cells that migrate into the bladder during infections (Song & Abraham, 2008). Purified LPS infused directly into the bladder induces pain in the pelvic region in a rodent model of urinary tract infection (Rudick et al., 2010). One possibility, therefore, is that sustained local inflammatory events precipitate TLR sensitization in migrating immune cells that then begin to modulate central pain processing after evidence of local infection is gone. This concept is supported by experiments demonstrating that a single peripheral inflammatory challenge (e.g. formalin) or peripheral trauma (e.g. laparotomy) can prime spinal microglia activation and subsequent LPS induced allodynia for as long as two weeks (Hains et al., 2010). More direct implications for IC/BPS symptoms have been demonstrated in an animal study that found a demyelination injury to the sciatic nerve increased the sensitivity of bladder-associated sensory neurons to chemokines and increased the frequency of micturition (Foster et al., 2011).
4.2 Limitations
This sample of IC/BPS patients was disproportionately non-Hispanic and white compared to the general population. These findings require replication in a more diverse sample. Isolated PBMCs were stimulated at a single dose-level of LPS and SAC. It is possible that characterizing TLR-2 inflammatory responses at a wide range of doses might reveal differences between IC/BPS only patients and those suffering comorbid conditions (Kwok et al., 2013). These analyses are cross-sectional and cannot determine causal directions between pain measures and TLR-mediated inflammation. A large number of statistical tests were performed in exploring these novel hypotheses; these associations require replication in other samples of IC/BPS patients.
4.1 Conclusions and Future Directions
TLR-4 mediated inflammation is a promising biomarker of comorbid pain in IC/BPS patients and may be associated with pain in other somatic syndromes. Putative TLR-4 antagonists that have been shown effective at suppressing pain in animal models may have application in IC/BPS populations. In addition to characterizing TLR-mediated inflammatory responses at a wider range of concentrations, more research is required to delineate the relationship between local inflammatory events and central pain sensitivity. As IC/BPS is often characterized by symptom fluctuation, termed “flares,” future studies should consider TLR-mediated inflammation in relation to these events.
Highlights.
We measured inflammatory responses in PBMCs to TLR stimulation in IC/BPS patients
Greater responses to TLR-4 stimulation were associated with widespread pain
Greater responses to TLR-4 stimulation were associated with comorbid conditions
TLR-4 mediated inflammation may be a therapeutic target in unexplained chronic pain
Acknowledgments
The authors gratefully acknowledge the assistance of Mary Eno in carrying out this project. This research was funded by grant UO1DK082344 to K.K. from the National Institute of Diabetes Digestive and Kidney Diseases and by the Institute for Clinical and Translational Science at the University of Iowa, grant 2 UL1 TR000442-06 . The authors gratefully acknowledge the MAPP research network:
MAPP Network Executive Committee: J. Quentin Clemens, MD, FACS, MSci, Network Chair, 2013-, Philip Hanno, MD, Ziya Kirkali, MD, John W. Kusek, PhD, J. Richard Landis, PhD, M. Scott Lucia, MD, Chris Mullins, PhD, Michel A. Pontari, MD. Northwestern University Discovery Site : David J. Klumpp, PhD, Co-Director, Anthony J. Schaeffer, MD, Co-Director, Apkar (Vania) Apkarian, PhD, David Cella, PhD, Melissa A. Farmer, PhD, Colleen Fitzgerald, MD, Richard Gershon, PhD, James W. Griffith, PhD, Charles J. Heckman II, PhD, Mingchen Jiang, PhD, Laurie Keefer, PhD, Darlene S. Marko, RN, BSN, CCRC, Jean Michniewicz, Todd Parrish, PhD, Frank Tu, MD, MPH. University of California, Los Angeles Discovery Site and PAIN Neuroimaging Core: Emeran A. Mayer, MD, Co-Director, Larissa V. Rodríguez, MD, Co-Director, Jeffry Alger, PhD, Cody P. Ashe-McNalley, Ben Ellingson, PhD, Nuwanthi Heendeniya, Lisa Kilpatrick, PhD, Jason Kutch, PhD, Jennifer S. Labus, PhD, Bruce D. Naliboff, PhD, Fornessa Randal, Suzanne R. Smith, RN, NP. University of Iowa Discovery Site: Karl J. Kreder, MD, MBA, Director, Catherine S. Bradley, MD, MSCE, Mary Eno, RN, RA II, Kris Greiner, BA, Yi Luo, PhD, MD, Susan K. Lutgendorf, PhD, Michael A. O’Donnell, MD, Barbara Ziegler, BA. University of Michigan Discovery Site: Daniel J. Clauw, MD, Co-Director; Network Chair, 2008-2013, J. Quentin Clemens, MD, FACS, MSci, Co-Director; Network Chair, 2013-, Suzie As-Sanie, MD, Sandra Berry, MA, Megan E. Halvorson, BS, CCRP, Richard Harris, PhD, Steve Harte, PhD, Eric Ichesco, BS, Ann Oldendorf, MD, Katherine A. Scott, RN, BSN, David A. Williams, PhD. University of Washington, Seattle Discovery Site: Dedra Buchwald, MD, Director, Niloofar Afari, PhD, Univ. Of California, San Diego, John Krieger, MD, Jane Miller, MD, Stephanie Richey, BS, Susan O. Ross, RN, MN, Roberta Spiro, MS, TJ Sundsvold, MPH, Eric Strachan, PhD, Claire C. Yang, MD. Washington University, St. Louis Discovery Site: Gerald L. Andriole, MD, Co-Director, H. Henry Lai, MD, Co-Director, Rebecca L. Bristol, BA, BS, Coordinator, Graham Colditz, MD, DrPH, Georg Deutsch, PhD, Univ. of Alabama at Birmingham, Vivien C. Gardner, RN, BSN, Coordinator, Robert W. Gereau IV, PhD, Jeffrey P Henderson, MD, PhD, Barry A. Hong, PhD, FAACP, Thomas M. Hooton, MD, Univ of Miami, Timothy J. Ness, MD, PhD, Univ. of Alabama at Birmingham, Carol S. North, MD, MPE, Univ. Texas Southwestern, Theresa M. Spitznagle, PT, DPT, WCS, Siobhan Sutcliffe, PhD, ScM, MHS. University of Pennsylvania Data Coordinating Core (DCC): J. Richard Landis, PhD, Core Director, Ted Barrell, BA, Philip Hanno, MD, Xiaoling Hou, MS, Tamara Howard, MPH, Michel A. Pontari, MD, Nancy Robinson, PhD, Alisa Stephens, PhD, Yanli Wang, MS, University of Colorado Denver Tissue Analysis & Technology Core (TATC): M. Scott Lucia, MD, Core Director, Adrie van Bokhoven, PhD, Andrea A. Osypuk, BS, Robert Dayton, Jr, Karen R. Jonscher, PhD, Holly T. Sullivan, BS, R. Storey Wilson, MS. Drexel University College of Medicine: Garth D.Ehrlich, PhD. Harvard Medical School/Boston Children's Hospital: Marsha A. Moses, PhD, Director, Andrew C. Briscoe, David Briscoe, MD, Adam Curatolo, BA, John Froehlich, PhD, Richard S. Lee, MD, Monisha Sachdev, BS, Keith R. Solomon, PhD, Hanno Steen, PhD. Stanford University : ,Sean Mackey, MD, PhD, Director, Epifanio Bagarinao, PhD, Lauren C. Foster, BA, Emily Hubbard, BA, Kevin A. Johnson, PhD, RN, Katherine T. Martucci, PhD, Rebecca L. McCue, BA, Rachel R. Moericke, MA, Aneesha Nilakantan, BA, Noorulain Noor, BS. Queens University: J. Curtis Nickel, MD, FRCSC, Director. National Institutes of Diabetes and Digestive and Kidney Diseases (NIDDK), National Institutes of Health (NIH): Chris Mullins, PhD, John W. Kusek, PhD, Ziya Kirkali, MD, Tamara G. Bavendam, MD
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Financial Disclosures
Dr. Harte has received consulting fees from Analgesic Solutions, DeCode genetics, and Pfizer, and has received grant support from Forest Laboratories and Cerephex, outside the submitted work; in addition, Dr. Harte has proprietary interest in the Multimodal Automated Sensory Testing system used in this research, including inventorship on United States and International patent applications, a licensing agreement, and the right to receive royalties from product commercialization. Dr. Kreder has received honorariums from Tengion and Medtronic, and served as a consultant for Tengion, Medtronic, and Symptelligence, which he has an equity interest in, outside the submitted work. Mr. Schrepf and Drs. Bradley, O'Donnell, Luo and Lutgendorf report no financial disclosures or potential conflicts of interest.
As a conservative measure, an additional analysis was conducted including tricyclic antidepressant use and patient age in addition to the above mentioned covariates. Neither tricyclic antidepressant use nor patient age were significantly associated with extra-pelvic pain (both p > .08) and their inclusion did not attenuate the association between TLR-4 mediated inflammation and extra-pelvic pain (odds ratio: 1.63, p = .013).
References
- Alba-Delgado C, Llorca-Torralba M, Horrillo I, Ortega JE, Mico JA, Sánchez-Blázquez P, Meana JJ, Berrocoso E. Chronic pain leads to concomitant noradrenergic impairment and mood disorders. Biol Psychiatry. 2013;73:54–62. doi: 10.1016/j.biopsych.2012.06.033. [DOI] [PubMed] [Google Scholar]
- Altman D, Lundholm C, Milsom I, Peeker R, Fall M, Iliadou AN, Pedersen NL. The genetic and environmental contribution to the occurrence of bladder pain syndrome: an empirical approach in a nationwide population sample. Eur Urol. 2011;59:280–285. doi: 10.1016/j.eururo.2010.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- As-Sanie S, Harris RE, Harte SE, Tu FF, Neshewat G, Clauw DJ. Increased pressure pain sensitivity in women with chronic pelvic pain. Obstet Gynecol. 2013;122:1047–1055. doi: 10.1097/AOG.0b013e3182a7e1f5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baayen RH, Davidson DJ, Bates DM. Mixed-effects modeling with crossed random effects for subjects and items. J Mem Lang. 2008;59:390–412. [Google Scholar]
- Bastos LF, Godin AM, Zhang Y, Jarussophon S, Ferreira BC, Machado RR, Maier SF, Konishi Y, de Freitas RP, Fiebich BL, Watkins LR, Coelho MM, Moraes MF. A minocycline derivative reduces nerve injury-induced allodynia, LPS-induced prostaglandin E2 microglial production and signaling via toll-like receptors 2 and 4. Neurosci Lett. 2013;543:157–162. doi: 10.1016/j.neulet.2013.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belmonte L, Beutheu Youmba S, Bertiaux-Vandaële N, Antonietti M, Lecleire S, Zalar A, Gourcerol G, Leroi AM, Déchelotte P, Coëffier M, Ducrotté P. Role of toll like receptors in irritable bowel syndrome: differential mucosal immune activation according to the disease subtype. PLoS One. 2012;7:e42777. doi: 10.1371/journal.pone.0042777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blankley S, Graham CM, Howes A, Bloom CI, Berry MP, Chaussabel D, Pascual V, Banchereau J, Lipman M, O'Garra A. Identification of the key differential transcriptional responses of human whole blood following TLR2 or TLR4 ligation in-vitro. PLoS One. 2014;9:e97702. doi: 10.1371/journal.pone.0097702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cleeland CS, Ryan KM. Pain assessment: global use of the Brief Pain Inventory. Ann Acad Med Singapore. 1994;23:129–138. [PubMed] [Google Scholar]
- Clemens JQ, Meenan RT, O'Keeffe Rosetti MC, Kimes TA, Calhoun EA. Case-control study of medical comorbidities in women with interstitial cystitis. J Urol. 2008;179:2222–2225. doi: 10.1016/j.juro.2008.01.172. [DOI] [PubMed] [Google Scholar]
- Clemens JQ, Mullins C, Kusek JW, Kirkali Z, Mayer EA, Rodríguez LV, Klumpp DJ, Schaeffer AJ, Kreder KJ, Buchwald D, Andriole GL, Lucia MS, Landis JR, Clauw DJ. The MAPP research network: a novel study of urologic chronic pelvic pain syndromes. BMC Urol. 2014;14:57. doi: 10.1186/1471-2490-14-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Díaz Mohedo E, Wärnberg J, Barón López FJ, Mera Velasco S, Cabello Burgos A. Chronic pelvic pain in Spanish women: prevalence and associated risk factors. A cross-sectional study. Clin Exp Obstet Gynecol. 2014;41:243–248. [PubMed] [Google Scholar]
- Drossman DA, Dumitrascu DL. Rome III: New standard for functional gastrointestinal disorders. J Gastrointestin Liver Dis. 2006;15:237–241. [PubMed] [Google Scholar]
- Duffy D, Rouilly V, Libri V, Hasan M, Beitz B, David M, Urrutia A, Bisiaux A, Labrie ST, Dubois A, Boneca IG, Delval C, Thomas S, Rogge L, Schmolz M, Quintana-Murci L, Albert ML, Milieu Intérieur Consortium Functional analysis via standardized whole-blood stimulation systems defines the boundaries of a healthy immune response to complex stimuli. Immunity. 2014;40:436–50. doi: 10.1016/j.immuni.2014.03.002. doi:10.1016/j.immuni.2014.03.002. [DOI] [PubMed] [Google Scholar]
- Dworkin SF, Sherman J, Mancl L, Ohrbach R, LeResche L, Truelove E. Reliability, validity, and clinical utility of the research diagnostic criteria for Temporomandibular Disorders Axis II Scales: depression, non-specific physical symptoms, and graded chronic pain. J Orofac Pain. 2002;16:207–220. [PubMed] [Google Scholar]
- Ellis A, Wieseler J, Favret J, Johnson KW, Rice KC, Maier SF, Falci S, Watkins LR. Systemic administration of propentofylline, ibudilast, and (+)-naltrexone each reverses mechanical allodynia in a novel rat model of central neuropathic pain. J Pain. 2014;15:407–421. doi: 10.1016/j.jpain.2013.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ford AC, Bercik P, Morgan DG, Bolino C, Pintos-Sanchez MI, Moayyedi P. Validation of the Rome III criteria for the diagnosis of irritable bowel syndrome in secondary care. Gastroenterology. 2013;145:1262–1270. e1. doi: 10.1053/j.gastro.2013.08.048. doi: 10.1053/j.gastro.2013.08.048. [DOI] [PubMed] [Google Scholar]
- Foster R, Jung J, Farooq A, McClung C, Ripsch MS, Fitzgerald MP, White FA. Sciatic nerve injury induces functional pro-nociceptive chemokine receptors in bladder-associated primary afferent neurons in the rat. Neuroscience. 2011;183:230–237. doi: 10.1016/j.neuroscience.2011.03.035. doi: 10.1016/j.neuroscience.2011.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda K, Straus SE, Hickie I, Sharpe MC, Dobbins JG, Komaroff A. The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med. 1994;121:953–959. doi: 10.7326/0003-4819-121-12-199412150-00009. [DOI] [PubMed] [Google Scholar]
- Giesecke J, Reed BD, Haefner HK, Giesecke T, Clauw DJ, Gracely RH. Quantitative Sensory Testing in Vulvodynia Patients and Increased Peripheral Pressure Pain Sensitivity. Obstet.Gynecol. 2004;104:126–133. doi: 10.1097/01.AOG.0000129238.49397.4e. [DOI] [PubMed] [Google Scholar]
- Grace PM, Hutchinson MR, Maier SF, Watkins LR. Pathological pain and the neuroimmune interface. Nat Rev Immunol. 2014;14:217–31. doi: 10.1038/nri3621. doi: 10.1038/nri3621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hains LE, Loram LC, Weiseler JL, Frank MG, Bloss EB, Sholar P, Taylor FR, Harrison JA, Martin TJ, Eisenach JC, Maier SF, Watkins LR. Pain intensity and duration can be enhanced by prior challenge: initial evidence suggestive of a role of microglial priming. J Pain. 2010;11:1004–1014. doi: 10.1016/j.jpain.2010.01.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanno PM, Burks DA, Clemens JQ, Dmochowski RR, Erickson D, Fitzgerald MP, Forrest JB, Gordon B, Gray M, Mayer RD, Newman D, Nyberg L, Jr, Payne CK, Wesselmann U, Faraday MM. AUA guideline for the diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J Urol. 2011;185:2162–2170. doi: 10.1016/j.juro.2011.03.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harte SE, Mitra M, Ichesco EA, Halvorson ME, Clauw DJ, Shih AJ, Kruger GH. Development and validation of a pressure-type automated quantitative sensory testing system for point-of-care pain assessment. Med Biol Eng Comput. 2013;51:633–644. doi: 10.1007/s11517-013-1033-x. [DOI] [PubMed] [Google Scholar]
- Hutchinson MR, La Vincente SF, Somogyi AA. In vitro opioid induced proliferation of peripheral blood immune cells correlates with in vivo cold pressor pain tolerance in humans: a biological marker of pain tolerance. Pain. 2004;110:751–755. doi: 10.1016/j.pain.2004.05.017. [DOI] [PubMed] [Google Scholar]
- Hutchinson MR, Zhang Y, Brown K, Coats BD, Shridhar M, Sholar PW, Patel SJ, Crysdale NY, Harrison JA, Maier SF, Rice KC, Watkins LR. Non-stereoselective reversal of neuropathic pain by naloxone and naltrexone: involvement of toll-like receptor 4 (TLR4). Eur J Neurosci. 2008;28:20–29. doi: 10.1111/j.1460-9568.2008.06321.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutchinson MR, Ramos KM, Loram LC, Wieseler J, Sholar PW, Kearney JJ. Evidence for a role of heat shock protein-90 in toll like receptor 4 mediated pain enhancement in rats. Neuroscience. 2009;164:1821–1832. doi: 10.1016/j.neuroscience.2009.09.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilpatrick LA, Kutch JJ, Tillisch K, Naliboff BD, Labus JS, Jiang Z. Alterations in resting state oscillations and connectivity in sensory and motor networks in women with interstitial cystitis/painful bladder syndrome. J Urol. 2014;192:947–955. doi: 10.1016/j.juro.2014.03.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komaroff AL, Fagioli LR, Geiger AM, Doolittle TH, Lee J, Kornish RJ, Gleit MA, Guerriero RT. An examination of the working case definition of chronic fatigue syndrome. Am J Med. 1996;100:56–64. doi: 10.1016/s0002-9343(96)90012-1. [DOI] [PubMed] [Google Scholar]
- Kovarik JJ, Tillinger W, Hofer J, Hölzl MA, Heinzl H, Saemann MD, Zlabinger GJ. Impaired anti-inflammatory efficacy of n-butyrate in patients with IBD. Eur J Clin Invest. 2011;41:291–298. [Google Scholar]
- Kowalski ML, Wolska A, Grzegorczyk J, Hilt J, Jarzebska M, Drobniewski M, Synder M, Kurowski M. Increased responsiveness to toll-like receptor 4 stimulation in peripheral blood mononuclear cells from patients with recent onset rheumatoid arthritis. Mediators Inflamm. 2008;2008:132732. doi: 10.1155/2008/132732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraemer HC, Giese-Davis J, Yutsis M, O'Hara R, Neri E, Gallagher-Thompson D, Taylor CB, Spiegel D. Design decisions to optimize reliability of daytime cortisol slopes in an older population. Am J Geriatr Psychiatry. 2006;14:325–333. doi: 10.1097/01.JGP.0000201816.26786.5b. [DOI] [PubMed] [Google Scholar]
- Kwok YH, Hutchinson MR, Gentgall MG, Rolan PE. Increased responsiveness of peripheral blood mononuclear cells to in vitro TLR 2, 4 and 7 ligand in chronic pain patients. PLoS One. 2012;7:e44232. doi: 10.1371/journal.pone.0044232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwok YH, Tuke J, Nicotra LL, Grace PM, Rolan PE, Hutchinson MR. TLR 2 and 4 responsiveness from isolated peripheral blood mononuclear cells from rats and humans as potential chronic pain biomarkers. PLoS One. 2013;8:e77799. doi: 10.1371/journal.pone.0077799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landis JR, Williams DA, Lucia MS, Clauw DJ, Naliboff BD, Robinson NA, van Bokhoven A, Sutcliffe S, Schaeffer AJ, Rodriguez LV, Mayer EA, Lai HH, Krieger JN, Kreder KJ, Afari N, Andriole GL, Bradley CS, Griffith JW, Klumpp DJ, Hong BA, Lutgendorf SK, Buchwald D, Yang CC, Mackey S, Pontari MA, Hanno P, Kusek JW, Mullins C, Clemens JQ. The MAPP research network: design, patient characterization and operations. BMC Urol. 2014;14:58. doi: 10.1186/1471-2490-14-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee YC, Lu B, Bathon JM, Haythornthwaite JA, Smith MT, Page GG, Edwards RR. Pain sensitivity and pain reactivity in osteoarthritis. Arthritis Care Res. 2011;63:320–327. doi: 10.1002/acr.20373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li GZ, Zhang N, Du P, Yang Y, Wu SL, Xiao YX, Jin R, Liu L, Shen H, Dai Y. Risk factors for interstitial cystitis/painful bladder syndrome in patients with lower urinary tract symptoms: a Chinese multi-center study. Chin Med J. 2010;23:2842–2846. [PubMed] [Google Scholar]
- Mattioli TA, Leduc-Pessah H, Skelhorne-Gross G, Nicol CJ, Milne B, Trang T, Cahill CM. Toll-like receptor 4 mutant and null mice retain morphine-induced tolerance, hyperalgesia, and physical dependence. PLoS One. 2014;9:e97361. doi: 10.1371/journal.pone.0097361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan ED, Watkins LR. Pathological and protective roles of glia in chronic pain. Nat Rev Neurosci. 2009;10:23–36. doi: 10.1038/nrn2533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ness TJ, Lloyd LK, Fillingim RB. An endogenous pain control system is altered in subjects with interstitial cystitis. J Urol. 2014;191:364–370. doi: 10.1016/j.juro.2013.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nickel JC, Tripp DA, Pontari M, Moldwin R, Mayer R, Carr LK. Interstitial cystitis/painful bladder syndrome and associated medical conditions with an emphasis on irritable bowel syndrome, fibromyalgia and chronic fatigue syndrome. J Urol. 2010;184:1358–1363. doi: 10.1016/j.juro.2010.06.005. [DOI] [PubMed] [Google Scholar]
- Ohman L, Lindmark AC, Isaksson S, Posserud I, Strid H, Sjövall H, Simrén M. Increased TLR2 expression on blood monocytes in irritable bowel syndrome patients. Eur J Gastroenterol Hepatol. 2012;24:398–405. doi: 10.1097/MEG.0b013e3283503f39. [DOI] [PubMed] [Google Scholar]
- Petzke F, Khine A, Williams D, Groner K, Clauw DJ, Gracely RH. Dolorimetry performed at 3 paired tender points highly predicts overall tenderness. J Rheumatol. 2001;28:2568–2569. [PubMed] [Google Scholar]
- Phillips K, Clauw DJ. Central pain mechanisms in chronic pain states--maybe it is all in their head. Best Pract Res Clin Rheumatol. 2011;25:141–154. doi: 10.1016/j.berh.2011.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothrock NE, Lutgendorf SK, Kreder KJ, Ratliff T, Zimmerman B. Stress and symptoms in patients with interstitial cystitis: a life stress model. Urology. 2001;57:422–427. doi: 10.1016/s0090-4295(00)00988-2. [DOI] [PubMed] [Google Scholar]
- Rudick CN, Billips BK, Pavlov VI, Yaggie RE, Schaeffer AJ, Klumpp DJ. Host-pathogen interactions mediating pain of urinary tract infection. J Infect Dis. 2010;201:1240–9. doi: 10.1086/651275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrepf A, O'Donnell M, Luo Y, Bradley CS, Kreder K, Lutgendorf S, Multidisciplinary Approach to the Study of Chronic Pelvic Pain (MAPP) Research Network Inflammation and inflammatory control in interstitial cystitis/bladder pain syndrome: Associations with painful symptoms. Pain. 2014;155:1755–1761. doi: 10.1016/j.pain.2014.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon LJ, Landis JR, Erickson DR, Nyberg LM. The Interstitial Cystitis Data Base Study: concepts and preliminary baseline descriptive statistics. Urology. 1997;49:64–75. doi: 10.1016/s0090-4295(99)80334-3. [DOI] [PubMed] [Google Scholar]
- Song J, Abraham SN. TLR-mediated immune responses in the urinary tract. Curr Opin Microbiol. 2008;11:66–73. doi: 10.1016/j.mib.2007.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorge RE, LaCroix-Fralish ML, Tuttle AH, Sotocinal SG, Austin JS, Ritchie J, Chanda ML, Graham AC, Topham L, Beggs S, Salter MW, Mogil JS. Spinal cord Toll-like receptor 4 mediates inflammatory and neuropathic hypersensitivity in male but not female mice. J Neurosci. 2011;31:15450–15454. doi: 10.1523/JNEUROSCI.3859-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan G, Jensen MP, Thornby JI, Shanti BF. Validation of the Brief Pain Inventory for chronic nonmalignant pain. J Pain. 2004;5:133–137. doi: 10.1016/j.jpain.2003.12.005. [DOI] [PubMed] [Google Scholar]
- Tramullas M, Finger BC, Moloney RD, Golubeva AV, Moloney G, Dinan TG, Cryan JF. Toll-like receptor 4 regulates chronic stress-induced visceral pain in mice. Biol Psychiatry. 2014;76:340–8. doi: 10.1016/j.biopsych.2013.11.004. [DOI] [PubMed] [Google Scholar]
- Warren JW, Langenberg P, Clauw DJ. The number of existing functional somatic syndromes (FSSs) is an important risk factor for new, different FSSs. J Psychosom Res. 2013;74:12–17. doi: 10.1016/j.jpsychores.2012.09.002. [DOI] [PubMed] [Google Scholar]
- Watson D, Clark LA, Tellegen A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol. 1988;54:1063–1070. doi: 10.1037//0022-3514.54.6.1063. [DOI] [PubMed] [Google Scholar]
- Wessely S, Nimnuan C, Sharpe M. Functional somatic syndromes: one or many? Lancet. 1999;354:936–939. doi: 10.1016/S0140-6736(98)08320-2. [DOI] [PubMed] [Google Scholar]
- Wieseler-Frank J, Maier SF, Watkins LR. Central proinflammatory cytokines and pain enhancement. Neurosignals. 2005;14:166–174. doi: 10.1159/000087655. [DOI] [PubMed] [Google Scholar]
- Wolfe F, Clauw DJ, Fitzcharles MA, Goldenberg DL, Katz RS, Mease P, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res. 2010;62:600–610. doi: 10.1002/acr.20140. [DOI] [PubMed] [Google Scholar]