Key Points
-
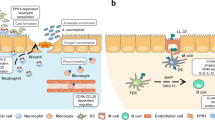
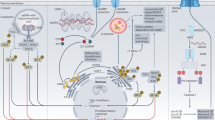
Peptidoglycan in bacterial cell walls is sensed by multiple pattern-recognition receptors, including nucleotide-binding oligomerization domain-containing protein 1 (NOD1), NOD2, NOD-, LRR- and pyrin domain-containing 3 (NLRP3) and peptidoglycan recognition protein 1 (PGLYRP1), that trigger inflammatory responses in immune and nonimmune cells throughout the body.
-
As an important structural component of bacterial cell walls, the sensitivity to degradation of peptidoglycan plays a vital role in determining the overall inflammatory response during infection. Peptidoglycan degradation is regulated by bacterial cell wall modifications that vary substantially among bacterial species and can be altered by exposure to antibiotics.
-
Peptidoglycan fragments are detected throughout the body in the absence of obvious infection. Some circulating peptidoglycan fragments can be traced to the gut microbiota.
-
These circulating peptidoglycan fragments are necessary for proper immune cell development and homeostasis. For example, circulating peptidoglycan fragments induce NOD1 signalling in phagocytes, altering their maturation and antimicrobial function.
-
Circulating peptidoglycan fragments have been shown to affect neuronal cell development in the brain and in the developing fetus.
Abstract
The innate immune system recognizes microbial products using germline-encoded receptors that initiate inflammatory responses to infection. The bacterial cell wall component peptidoglycan is a prime example of a conserved pathogen-associated molecular pattern (PAMP) for which the innate immune system has evolved sensing mechanisms. Peptidoglycan is a direct target for innate immune receptors and also regulates the accessibility of other PAMPs to additional innate immune receptors. Subtle structural modifications to peptidoglycan can influence the ability of the innate immune system to detect bacteria and can allow bacteria to evade or alter host defences. This Review focuses on the mechanisms of peptidoglycan recognition that are used by mammalian cells and discusses new insights into the role of peptidoglycan recognition in inflammation, metabolism, immune homeostasis and disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kawai, T. & Akira, S. The roles of TLRs, RLRs and NLRs in pathogen recognition. Int. Immunol. 21, 317–337 (2009).
Janeway, C. A. Jr. Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb. Symp. Quant. Biol. 54, 1–13 (1989).
Kurata, S. Peptidoglycan recognition proteins in Drosophila immunity. Dev. Comp. Immunol. 42, 36–41 (2014).
Vollmer, W., Blanot, D. & De Pedro, M. A. Peptidoglycan structure and architecture. FEMS Microbiol. Rev. 32, 149–167 (2008).
Royet, J., Gupta, D. & Dziarski, R. Peptidoglycan recognition proteins: modulators of the microbiome and inflammation. Nat. Rev. Immunol. 11, 837–851 (2011).
Kashyap, D. R. et al. Peptidoglycan recognition proteins kill bacteria by activating protein-sensing two-component systems. Nat. Med. 17, 676–683 (2011).
Kashyap, D. R. et al. Peptidoglycan recognition proteins kill bacteria by inducing oxidative, thiol, and metal Stress. PLoS Pathog. 10, e1004280–1004217 (2014).
Kashyap, D. R., Kuzma, M., Kowalczyk, D. A., Gupta, D. & Dziarski, R. Bactericidal peptidoglycan recognition protein induces oxidative stress in Escherichia coli through a block in respiratory chain and increase in central carbon catabolism. Mol. Microbiol. 105, 755–776 (2017).
Wang, Z. M. et al. Human peptidoglycan recognition protein-L is an N-acetylmuramoyl-L-alanine amidase. J. Biol. Chem. 278, 49044–49052 (2003).
De Marzi, M. C. et al. Peptidoglycan recognition protein-peptidoglycan complexes increase monocyte/macrophage activation and enhance the inflammatory response. Immunology 145, 429–442 (2015).
Dziarski, R., Platt, K. A., Gelius, E., Steiner, H. & Gupta, D. Defect in neutrophil killing and increased susceptibility to infection with nonpathogenic gram-positive bacteria in peptidoglycan recognition protein-S (PGRP-S)-deficient mice. Blood 102, 689–697 (2003).
Cho, J. H. et al. Human peptidoglycan recognition protein S is an effector of neutrophil-mediated innate immunity. Blood 106, 2551–2558 (2005).
Read, C. B. et al. Cutting Edge: identification of neutrophil PGLYRP1 as a ligand for TREM-1. J. Immunol. 194, 1417–1421 (2015).
Yashin, D. V. et al. Tag7 (PGLYRP1) in complex with Hsp70 induces alternative cytotoxic processes in tumor cells via TNFR1 receptor. J. Biol. Chem. 290, 21724–21731 (2015).
Kieser, K. J. & Kagan, J. C. Multi-receptor detection of individual bacterial products by the innate immune system. Nat. Rev. Immunol. 17, 376–390 (2017).
Yoshimura, A. et al. Cutting edge: recognition of Gram-positive bacterial cell wall components by the innate immune system occurs via Toll-like receptor 2. J. Immunol. 163, 1–5 (1999).
Takeuchi, O. et al. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11, 443–451 (1999).
Ozinsky, A. et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between Toll-like receptors. Proc. Natl Acad. Sci. USA 97, 13766–13771 (2000).
Travassos, L. H. et al. Toll-like receptor 2-dependent bacterial sensing does not occur via peptidoglycan recognition. EMBO Rep. 5, 1000–1006 (2004).
Dziarski, R. & Gupta, D. Staphylococcus aureus peptidoglycan Is a Toll-like receptor 2 activator: a reevaluation. Infect. Immun. 73, 5212–5216 (2005).
Asong, J., Wolfert, M. A., Maiti, K. K., Miller, D. & Boons, G. J. Binding and cellular activation studies reveal that Toll-like receptor 2 can differentially recognize peptidoglycan from Gram-positive and Gram-negative bacteria. J. Biol. Chem. 284, 8634–8644 (2009).
Iyer, J. K. et al. Inflammatory cytokine response to Bacillus anthracis peptidoglycan requires phagocytosis and lysosomal trafficking. Infect. Immun. 78, 2418–2428 (2010).
Volz, T. et al. Natural Staphylococcus aureus-derived peptidoglycan fragments activate NOD2 and act as potent costimulators of the innate immune system exclusively in the presence of TLR signals. FASEB J. 24, 4089–4102 (2010).
Caruso, R., Warner, N., Inohara, N. & Nuñez, G. NOD1 and NOD2: signaling, host defense, and inflammatory disease. Immunity 41, 898–908 (2014).
Bertin, J. et al. Human CARD4 protein is a novel CED-4/Apaf-1 cell death family member that activates NF-κB. J. Biol. Chem. 274, 12955–12958 (1999).
Inohara, N. et al. Nod1, an Apaf-1-like activator of caspase-9 and nuclear factor-κB. J. Biol. Chem. 274, 14560–14567 (1999).
Ogura, Y. et al. Nod2, a Nod1/Apaf-1 family member that is restricted to monocytes and activates NF-κB. J. Biol. Chem. 276, 4812–4818 (2001).
Girardin, S. E. et al. Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J. Biol. Chem. 278, 8869–8872 (2003).
Inohara, N. et al. Host recognition of bacterial muramyl dipeptide mediated through NOD2. Implications for Crohn's disease. J. Biol. Chem. 278, 5509–5512 (2003).
Chamaillard, M. et al. An essential role for NOD1 in host recognition of bacterial peptidoglycan containing diaminopimelic acid. Nat. Immunol. 4, 702–707 (2003).
Girardin, S. E. et al. Nod1 detects a unique muropeptide from gram-negative bacterial peptidoglycan. Science 300, 1584–1587 (2003).
Grimes, C. L., Ariyananda, L. D. Z., Melnyk, J. E. & O'Shea, E. K. The innate immune protein Nod2 binds directly to MDP, a bacterial cell wall fragment. J. Am. Chem. Soc. 134, 13535–13537 (2012).
Mo, J. et al. Pathogen sensing by nucleotide-binding oligomerization domain-containing protein 2 (NOD2) is mediated by direct binding to muramyl dipeptide and ATP. J. Biol. Chem. 287, 23057–23067 (2012).
Laroui, H. et al. L-Ala-γ-D-Glu-meso-diaminopimelic acid (DAP) interacts directly with leucine-rich region domain of nucleotide-binding oligomerization domain 1, increasing phosphorylation activity of receptor-interacting serine/threonine-protein kinase 2 and its interaction with nucleotide-binding oligomerization domain 1. J. Biol. Chem. 286, 31003–31013 (2011).
Lee, J. et al. pH-dependent internalization of muramyl peptides from early endosomes enables Nod1 and Nod2 signaling. J. Biol. Chem. 284, 23818–23829 (2009).
Sasawatari, S. et al. The solute carrier family 15A4 regulates TLR9 and NOD1 functions in the innate immune system and promotes colitis in mice. Gastroenterology 140, 1513–1525 (2011).
Nakamura, N. et al. Endosomes are specialized platforms for bacterial sensing and NOD2 signalling. Nature 509, 240–244 (2014).
Charriere, G. M. et al. Identification of Drosophila Yin and PEPT2 as evolutionarily conserved phagosome-associated muramyl dipeptide transporters. J. Biol. Chem. 285, 20147–20154 (2010).
Ismair, M. G. et al. hPepT1 selectively transports muramyl dipeptide but not Nod1-activating muramyl peptides. Can. J. Physiol. Pharmacol. 84, 1313–1319 (2006).
Vavricka, S. R. et al. hPepT1 transports muramyl dipeptide, activating NF-κB and stimulating IL-8 secretion in human colonic Caco2/bbe cells. Gastroenterology 127, 1401–1409 (2004).
Paik, D. et al. SLC46 family transporters facilitate cytosolic innate immune recognition of monomeric peptidoglycans. J. Immunol. 199, 263–270 (2017).
Schwechheimer, C. & Kuehn, M. J. Outer-membrane vesicles from Gram-negative bacteria: biogenesis and functions. Nat. Rev. Micro. 13, 605–619 (2015).
Bielig, H., Dongre, M., Zurek, B., Wai, S. N. & Kufer, T. A. A role for quorum sensing in regulating innate immune responses mediated by Vibrio cholerae outer membrane vesicles (OMVs). Gut Microbes 2, 274–279 (2011).
Bielig, H. et al. NOD-like receptor activation by outer membrane vesicles from Vibrio cholerae non-O1 non-O139 strains is modulated by the quorum-sensing regulator HapR. Infect. Immun. 79, 1418–1427 (2011).
Kaparakis, M. et al. Bacterial membrane vesicles deliver peptidoglycan to NOD1 in epithelial cells. Cell. Microbiol. 12, 372–385 (2010).
Keestra-Gounder, A. M. et al. NOD1 and NOD2 signalling links ER stress with inflammation. Nature 532, 394–397 (2016). This paper highlights a role for NOD1 and NOD2 that is independent of peptidoglycan sensing.
Shimada, T. et al. Staphylococcus aureus evades lysozyme-based peptidoglycan digestion that links phagocytosis, inflammasome activation, and IL-1β secretion. Cell Host Microbe 7, 38–49 (2010).
Martinon, F., Agostini, L., Meylan, E. & Tschopp, J. Identification of bacterial muramyl dipeptide as activator of the NALP3/cryopyrin inflammasome. Curr. Biol. 14, 1929–1934 (2004).
Lamkanfi, M. & Dixit, V. M. Mechanisms and functions of inflammasomes. Cell 157, 1013–1022 (2014).
Pan, Q. et al. MDP-induced interleukin-1β processing requires Nod2 and CIAS1/NALP3. J. Leukoc. Biol. 82, 177–183 (2007).
Marina-Garcia, N. et al. Pannexin-1-mediated intracellular delivery of muramyl dipeptide induces caspase-1 activation via cryopyrin/NLRP3 independently of Nod2. J. Immunol. 180, 4050–4057 (2008).
Hsu, L. C. et al. A NOD2-NALP1 complex mediates caspase-1-dependent IL-1β secretion in response to Bacillus anthracis infection and muramyl dipeptide. Proc. Natl Acad. Sci. USA 105, 7803–7808 (2008).
Ferwerda, G. et al. Engagement of NOD2 has a dual effect on proIL-1β mRNA transcription and secretion of bioactive IL-1β. Eur. J. Immunol. 38, 184–191 (2008).
Faustin, B. et al. Reconstituted NALP1 inflammasome reveals two-step mechanism of caspase-1 activation. Mol. Cell 25, 713–724 (2007).
Hedl, M. & Abraham, C. NLRP1 and NLRP3 inflammasomes are essential for distinct outcomes of decreased cytokines but enhanced bacterial killing upon chronic Nod2 stimulation. Am. J. Physiol. Gastrointest. Liver Physiol. 304, G583–596 (2013).
Wolf, A. J. et al. Hexokinase is an innate immune receptor for the detection of bacterial peptidoglycan. Cell 166, 624–636 (2016). This paper describes the innate immune sensing of the peptidoglycan sugar NAG through perturbation of the glycolytic metabolism of a phagocytic cell.
Vollmer, W. Structural variation in the glycan strands of bacterial peptidoglycan. FEMS Microbiol. Rev. 32, 287–306 (2008).
Bera, A., Herbert, S., Jakob, A., Vollmer, W. & Gotz, F. Why are pathogenic staphylococci so lysozyme resistant? The peptidoglycan O-acetyltransferase OatA is the major determinant for lysozyme resistance of Staphylococcus aureus. Mol. Microbiol. 55, 778–787 (2005).
Wolf, A. J. et al. Phagosomal degradation increases TLR access to bacterial ligands and enhances macrophage sensitivity to bacteria. J. Immunol. 187, 6002–6010 (2011).
Ip, W. K. E. et al. Phagocytosis and phagosome acidification are required for pathogen processing and MyD88-dependent responses to Staphylococcus aureus. J. Immunol. 184, 7071–7081 (2010).
de Jonge, B. L. M. & Tomasz, A. Abnormal peptidoglycan produced in a methicillin-resistant strain of Staphylococcus aureus grown in the presence of methicillin: functional role for penicillin-binding protein 2a in cell wall synthesis. Antimicrob. Agents Chemother. 37, 342–346 (1993).
Müller, S. et al. Poorly cross-linked peptidoglycan in MRSA due to mecA induction activates the inflammasome and exacerbates immunopathology. Cell Host Microbe 18, 604–612 (2015). This reference describes how antibiotics can induce changes in peptidoglycan structure, leading to increased degradation sensitivity that affects the overall inflammatory response to S. aureus.
Wolf, A. J., Liu, G. Y. & Underhill, D. M. Inflammatory properties of antibiotic-treated bacteria. J. Leukoc. Biol. 101, 127–134 (2017).
Fritz, J. H. et al. Nod1-mediated innate immune recognition of peptidoglycan contributes to the onset of adaptive immunity. Immunity 26, 445–459 (2007).
Schaffler, H. et al. NOD2 stimulation by Staphylococcus aureus-derived peptidoglycan is boosted by Toll-like receptor 2 costimulation with lipoproteins in dendritic cells. Infect. Immun. 82, 4681–4688 (2014).
Tada, H., Aiba, S., Shibata, K., Ohteki, T. & Takada, H. Synergistic effect of Nod1 and Nod2 agonists with toll-like receptor agonists on human dendritic cells to generate interleukin-12 and T helper type 1 cells. Infect. Immun. 73, 7967–7976 (2005).
van Heel, D. A. et al. Synergistic enhancement of Toll-like receptor responses by NOD1 activation. Eur. J. Immunol. 35, 2471–2476 (2005).
Magalhaes, J. G. et al. Nod2-Dependent Th2 Polarization of Antigen-Specific Immunity. J. Immunol. 181, 7925–7935 (2008).
Watanabe, T., Kitani, A., Murray, P. J. & Strober, W. NOD2 is a negative regulator of Toll-like receptor 2-mediated T helper type 1 responses. Nat. Immunol. 5, 800–808 (2004).
Netea, M. G. et al. Nucleotide-binding oligomerization domain-2 modulates specific TLR pathways for the induction of cytokine release. J. Immunol. 174, 6518–6523 (2005).
Netea, M. G. et al. NOD2 mediates anti-inflammatory signals induced by TLR2 ligands: implications for Crohn's disease. Eur. J. Immunol. 34, 2052–2059 (2004).
Kelly, B. & O'Neill, L. A. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res. 25, 771–784 (2015).
Hedl, M., Yan, J. & Abraham, C. IRF5 and IRF5 disease-risk variants increase glycolysis and human M1 polarization by regulating proximal signaling and Akt2 activation. Cell Rep. 16, 2442–2455 (2016).
Rabot, S. et al. Germ-free C57BL/6J mice are resistant to high-fat-diet-induced insulin resistance and have altered cholesterol metabolism. FASEB J. 24, 4948–4959 (2010).
Chan, K. L. et al. Circulating NOD1 activators and hematopoietic NOD1 contribute to metabolic inflammation and insulin resistance. Cell Rep. 18, 2415–2426 (2017). This paper highlights a role for circulating peptidoglycan fragments that activate NOD1 signalling and alter the body's metabolism.
Denou, E. et al. Defective NOD2 peptidoglycan sensing promotes diet-induced inflammation, dysbiosis, and insulin resistance. EMBO Mol. Med. 7, 259–274 (2015). In this paper, Nod2−/− mice on a HFD show increased intestinal permeability linked to worse glucose tolerance.
Schertzer, J. D. et al. NOD1 activators link innate immunity to insulin resistance. Diabetes 60, 2206–2215 (2011).
Cavallari, J. F. et al. Muramyl dipeptide-based postbiotics mitigate obesity-induced insulin resistance via IRF4. Cell Metab. 25, 1063–1074.e3 (2017). This paper shows that the NOD2 ligand MDP activates IRF4 and leads to decreased levels of inflammation in adipose tissue and improved overall glucose tolerance.
Luckey, T. D. Introduction to intestinal microecology. Am. J. Clin. Nutr. 25, 1292–1294 (1972).
Sender, R., Fuchs, S. & Milo, R. Revised estimates for the number of human and bacteria cells in the body. PLoS Biol. 14, e1002533 (2016).
Sender, R., Fuchs, S. & Milo, R. Are we really vastly outnumbered? Revisiting the ratio of bacterial to host cells in humans. Cell 164, 337–340 (2016).
Hugot, J. P. et al. Prevalence of CARD15/NOD2 mutations in Caucasian healthy people. Am. J. Gastroenterol. 102, 1259–1267 (2007).
Eckmann, L. & Karin, M. NOD2 and Crohn's disease: loss or gain of function? Immunity 22, 661–667 (2005).
van Heel, D. A. et al. Muramyl dipeptide and toll-like receptor sensitivity in NOD2-associated Crohn's disease. Lancet 365, 1794–1796 (2005).
Al Nabhani, Z., Dietrich, G., Hugot, J. P. & Barreau, F. Nod2: the intestinal gate keeper. PLOS Pathog. 13, e1006177 (2017).
Ostaff, M. J., Stange, E. F. & Wehkamp, J. Antimicrobial peptides and gut microbiota in homeostasis and pathology. EMBO Mol. Med. 5, 1465–1483 (2013).
Ramanan, D. et al. Helminth infection promotes colonization resistance via type 2 immunity. Science 352, 608–612 (2016).
Rehman, A. et al. Nod2 is essential for temporal development of intestinal microbial communities. Gut 60, 1354–1362 (2011).
Chassaing, B. et al. Crohn disease-associated adherent-invasive E. coli bacteria target mouse and human Peyer's patches via long polar fimbriae. J. Clin. Invest. 121, 966–975 (2011).
Mondot, S. et al. Altered gut microbiota composition in immune-impaired Nod2−/− mice. Gut 61, 634–635 (2012).
Petnicki-Ocwieja, T. et al. Nod2 is required for the regulation of commensal microbiota in the intestine. Proc. Natl Acad. Sci. USA 106, 15813–15818 (2009).
Robertson, S. J. et al. Nod1 and Nod2 signaling does not alter the composition of intestinal bacterial communities at homeostasis. Gut Microbes 4, 222–231 (2013).
Shanahan, M. T. et al. Mouse Paneth cell antimicrobial function is independent of Nod2. Gut 63, 903–910 (2014).
Al Nabhani, Z. et al. Nod2 deficiency leads to a specific and transmissible mucosa-associated microbial dysbiosis which is independent of the mucosal barrier defect. J. Crohns Colitis 10, 1428–1436 (2016).
Ramanan, D., Tang, M. S., Bowcutt, R., Loke, P. a. n. & Cadwell, K. Bacterial sensor Nod2 prevents inflammation of the small intestine by restricting the expansion of the commensal Bacteroides vulgatus. Immunity 41, 311–324 (2014).
Bouskra, D. et al. Lymphoid tissue genesis induced by commensals through NOD1 regulates intestinal homeostasis. Nature 456, 507–510 (2008).
Tsuji, M., Suzuki, K., Kinoshita, K. & Fagarasan, S. Dynamic interactions between bacteria and immune cells leading to intestinal IgA synthesis. Semin. Immunol. 20, 59–66 (2008).
Sidiq, T., Yoshihama, S., Downs, I. & Kobayashi, K. S. Nod2: a critical regulator of ileal microbiota and Crohn's disease. Front. Immunol. 7, 367 (2016).
Barnich, N., Aguirre, J. E., Reinecker, H. C., Xavier, R. & Podolsky, D. K. Membrane recruitment of NOD2 in intestinal epithelial cells is essential for nuclear factor-κB activation in muramyl dipeptide recognition. J. Cell Biol. 170, 21–26 (2005).
Marina-Garcia, N. et al. Clathrin- and dynamin-dependent endocytic pathway regulates muramyl dipeptide internalization and NOD2 activation. J. Immunol. 182, 4321–4327 (2009).
Viala, J. et al. Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island. Nat. Immunol. 5, 1166–1174 (2004).
Leung, C. H., Lam, W., Ma, D. L., Gullen, E. A. & Cheng, Y. C. Butyrate mediates nucleotide-binding and oligomerisation domain (NOD) 2-dependent mucosal immune responses against peptidoglycan. Eur. J. Immunol. 39, 3529–3537 (2009).
Cooney, R. et al. NOD2 stimulation induces autophagy in dendritic cells influencing bacterial handling and antigen presentation. Nat. Med. 16, 90–97 (2010).
Travassos, L. H. et al. Nod1 and Nod2 direct autophagy by recruiting ATG16L1 to the plasma membrane at the site of bacterial entry. Nat. Immunol. 11, 55–62 (2010).
Homer, C. R., Richmond, A. L., Rebert, N. A., Achkar, J. P. & McDonald, C. ATG16L1 and NOD2 interact in an autophagy-dependent antibacterial pathway implicated in Crohn's disease pathogenesis. Gastroenterology 139, 1630–1641.e2 (2010).
Irving, A. T. et al. The immune receptor NOD1 and kinase RIP2 interact with bacterial peptidoglycan on early endosomes to promote autophagy and inflammatory signaling. Cell Host Microbe 15, 623–635 (2014). References 103–106 demonstrate how NOD1 and NOD2 signalling can activate the autophagy pathway to protect against bacterial invasion.
Stappenbeck, T. S., Hooper, L. V. & Gordon, J. I. Developmental regulation of intestinal angiogenesis by indigenous microbes via Paneth cells. Proc. Natl Acad. Sci. USA 99, 15451–15455 (2002).
Ayabe, T. et al. Secretion of microbicidal α-defensins by intestinal Paneth cells in response to bacteria. Nat. Immunol. 1, 113–118 (2000).
Kobayashi, K. S. et al. Nod2-dependent regulation of innate and adaptive immunity in the intestinal tract. Science 307, 731–734 (2005).
Vaishnava, S. et al. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011).
Ogura, Y. et al. Expression of NOD2 in Paneth cells: a possible link to Crohn's ileitis. Gut 52, 1591–1597 (2003).
Duerr, C. U. et al. Control of intestinal Nod2-mediated peptidoglycan recognition by epithelium-associated lymphocytes. Mucosal. Immunol. 4, 325–334 (2011).
Macho Fernandez, E. et al. Anti-inflammatory capacity of selected lactobacilli in experimental colitis is driven by NOD2-mediated recognition of a specific peptidoglycan-derived muropeptide. Gut 60, 1050–1059 (2011).
Clarke, T. B. et al. Recognition of peptidoglycan from the microbiota by Nod1 enhances systemic innate immunity. Nat. Med. 16, 228–231 (2010). This paper uses radiolabelled bacteria to demonstrate that peptidoglycan fragments from gut microbes enter the circulation and enhance the capacity of neutrophils to kill bacteria through a NOD1 signal.
Arentsen, T. et al. The bacterial peptidoglycan-sensing molecule Pglyrp2 modulates brain development and behavior. Mol. Psychiatry 22, 257–266 (2017). In this paper, the authors show that peptidoglycan fragments can reach the brain and, through recognition by PGLYRP, alter neuronal development and cause behaviourial changes.
Hergott, C. B. et al. Peptidoglycan from the gut microbiota governs the lifespan of circulating phagocytes at homeostasis. Blood 127, 2460–2471 (2016). This reference highlights the need for a gut microbiota for the proper development of phagocytic cells and also shows that phagocytic cell development is dependent on NOD1 signalling.
Martinic, M. M. et al. The bacterial peptidoglycan-sensing molecules NOD1 and NOD2 promote CD8+ thymocyte selection. J. Immunol. 198, 2649–2660 (2017).
Lin, G. H., Wortzman, M. E., Girardin, S. E., Philpott, D. J. & Watts, T. H. T cell intrinsic NOD2 is dispensable for CD8 T cell immunity. PLoS ONE 8, e56014 (2013).
Saha, S. et al. Peptidoglycan recognition proteins protect mice from experimental colitis by promoting normal gut flora and preventing induction of interferon-gamma. Cell Host Microbe 8, 147–162 (2010).
Jing, X. et al. Peptidoglycan recognition protein 3 and Nod2 synergistically protect mice from dextran sodium sulfate-induced colitis. J. Immunol. 193, 3055–3069 (2014).
Diaz Heijtz, R. et al. Normal gut microbiota modulates brain development and behavior. Proc. Natl Acad. Sci. USA 108, 3047–3052 (2011).
Goldman, S. M. et al. Peptidoglycan recognition protein genes and risk of Parkinson's disease. Mov. Disord. 29, 1171–1180 (2014).
Vollmer, W. & Tomasz, A. The pgdA gene encodes for a peptidoglycan N-acetylglucosamine deacetylase in Streptococcus pneumoniae. J. Biol. Chem. 275, 20496–20501 (2000).
Boneca, I. G. et al. A critical role for peptidoglycan N-deacetylation in Listeria evasion from the host innate immune system. Proc. Natl Acad. Sci. USA 104, 997–1002 (2007).
Melnyk, J. E., Mohanan, V., Schaefer, A. K., Hou, C.-W. & Grimes, C. L. Peptidoglycan modifications tune the stability and function of the innate immune receptor Nod2. J. Am. Chem. Soc. 137, 6987–6990 (2015).
Weadge, J. T., Pfeffer, J. M. & Clarke, A. J. Identification of a new family of enzymes with potential O-acetylpeptidoglycan esterase activity in both Gram-positive and Gram-negative bacteria. BMC Microbiol. 5, 49 (2005).
Raymond, J. B., Mahapatra, S., Crick, D. C. & Pavelka, M. S. Jr. Identification of the namH gene, encoding the hydroxylase responsible for the N-glycolylation of the mycobacterial peptidoglycan. J. Biol. Chem. 280, 326–333 (2005).
Vijayrajratnam, S. et al. Bacterial peptidoglycan with amidated meso-diaminopimelic acid evades NOD1 recognition: an insight into NOD1 structure-recognition. Biochem. J. 473, 4573–4592 (2016).
Girardin, S. E. et al. Peptidoglycan molecular requirements allowing detection by Nod1 and Nod2. J. Biol. Chem. 278, 41702–41708 (2003).
Cava, F., de Pedro, M. A., Lam, H., Davis, B. M. & Waldor, M. K. Distinct pathways for modification of the bacterial cell wall by non-canonical D-amino acids. EMBO J. 30, 3442–3453 (2011).
Radkov, A. D. & Moe, L. A. Bacterial synthesis of D-amino acids. Appl. Microbiol. Biotechnol. 98, 5363–5374 (2014).
Bradshaw, W. J. et al. Molecular features of the sortase enzyme family. FEBS J. 282, 2097–2114 (2015).
Author information
Authors and Affiliations
Contributions
Both authors contributed to the writing of this Review.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Pattern-recognition receptors
-
(PRRs). Innate immune receptors expressed at the cell surface and in the cytoplasm that sense small molecules that are common to several different microorganisms.
- Lipoproteins
-
Molecules embedded in the cell wall of both Gram-positive and Gram-negative bacteria that play diverse functional roles, including adhesion, signalling and nutrient uptake and that activate signalling through Toll-like receptor 2 (TLR2) in combination with either TLR1 or TLR6.
- Staphylococcus aureus
-
A Gram-positive bacterium that behaves as an important commensal bacteria and opportunistic pathogen.
- Lipopolysaccharide
-
(LPS). A component of the cell wall of most Gram-negative bacteria and the innate immune ligand for the membrane receptor Toll-like receptor 4 (TLR4).
- Lipoteichoic acids
-
(LTAs). LTAs and teichoic acids are hydrophilic polyphosphate polymers anchored by a glycolipid link to the bacterial membrane or covalently bound to the cell wall peptidoglycan, respectively. They are specific to Gram-positive bacteria and have been reported to activate Toll-like receptor 2 (TLR2).
- Bacillus anthracis
-
Anthrax. A Gram-positive, spore-forming bacterium that is a common livestock pathogen and an occasional human pathogen.
- Lysostaphin
-
A zinc-dependent endopeptidase with bacteriolysin activity that is produced by certain Staphylococci strains. It specifically cleaves the pentaglycine bridge found only in peptidoglycan generated by Staphylococcus aureus and a few other Staphylococci strains.
- Mutanolysin
-
An N-acetylmuramidase that cleaves the β(1–4)-linkage between N-acetylmuramic acid and N-acetylglucosamine in the sugar backbone of peptidoglycan. Mutanolysin treatment of several strains of Gram-positive bacteria results in bacterial lysis.
- PAM3CSK4
-
A small synthetic triacylated lipopeptide ligand for TLR2–TLR1 heterodimers of Toll-like receptors. It functions as a mimic of the acylated amino terminus of lipoproteins found in the cell wall of both Gram-positive and Gram-negative bacteria that activate TLR2–TLR1.
- NOD-like receptors
-
(NLRs). A large family of related proteins that function as cytosolic innate immune sensors and share a general three domain structure. All NLRs contain a nucleotide- binding and oligomerization domain (NACHT). Most have a C-terminal leucine-rich repeat domain, and the N-terminus is composed of various combinations of effector domains that can include the caspase recruitment domain (CARD), PYRIN domains, baculoviral inhibitor of apoptosis repeat domains or the transactivator domain.
- Tracheal cytotoxin
-
(TCT). A small fragment of peptidoglycan from the Gram-negative bacteria Bordetella pertussis and Neisseria gonorrhoeae composed of a disaccharide and a diaminopimelic acid (DAP)-containing tetrapeptide that is recognized by nucleotide-binding oligomerization domain-containing protein 1 (NOD1).
- Thapsigargin
-
An inhibitor of the sarcoplasmic reticulum Ca2+ ATPase (SERCA) channels in the membrane of the endoplasmic reticulum (ER) responsible for transporting Ca2+ from the cytoplasm into the ER. Thapsigargin treatment results in an elevation in cytosolic Ca2+ while depleting ER Ca2+ stores, leading to ER stress and activation of Ca2+-dependent cell signalling pathways.
- Inflammasome
-
A large multiprotein complex that contains certain NOD-like receptors, retinoic acid RIG-I-like receptors and IFI200 proteins, the adaptor protein ASC and pro-caspase 1. Assembly of the inflammasome leads to the activation of caspase 1, which cleaves pro-interleukin-Iβ (pro-IL-1β) and pro-IL-18 to generate the active forms of these pro-inflammatory cytokines.
- Lysozyme
-
Also known as muramidase. A small antibacterial enzyme produced by innate immune phagocytes. A glycoside hydrolase that cleaves the β(1–4) linkage between N-acetylmuramic acid (MurNAc) and N-acetylglucosamine (NAG) in bacterial peptidoglycan.
- β-Lactam antibiotics
-
β-Lactam antibiotics inhibit bacterial cell wall peptidoglycan synthesis, leading to a build-up of peptidoglycan intermediates and induction of bacterial autolysis. This class of broad spectrum antibiotics includes penicillins, cephalosporins, monobactams and carbapenems.
- Hexokinase
-
The enzyme responsible for phosphorylating glucose, which is the first step in glycolysis.
- Warburg effect
-
Describes the metabolic state of a cell that primarily produces ATP by elevating its rate of glycolysis even in the presence of sufficient levels of oxygen, which could be used to produce ATP by mitochondrial respiration. This is known as aerobic glycolysis and is a common feature of many cancer cells.
- Paneth cells
-
Specialized epithelial cells found in the crypts of the small intestine epithelium that are responsible for producing large amounts of antimicrobial peptides (for example, α-defensins) and antimicrobial enzymes (for example, lysozyme) that help keep the gut microbiome in check and protect the epithelium from pathogenic bacteria.
- Cryptopatches
-
Small aggregates of lymphocytes in the lamina propria of the small intestine thought to be the site of extrathymic T cell development; they contain lymphoid tissue inducer cells (LTis) and Group 3 innate lymphoid cells (ILC3s).
Rights and permissions
About this article
Cite this article
Wolf, A., Underhill, D. Peptidoglycan recognition by the innate immune system. Nat Rev Immunol 18, 243–254 (2018). https://doi.org/10.1038/nri.2017.136
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri.2017.136
This article is cited by
-
Effects of dietary Lactobacillus postbiotics and bacitracin on the modulation of mucosa-associated microbiota and pattern recognition receptors affecting immunocompetence of jejunal mucosa in pigs challenged with enterotoxigenic F18+ Escherichia coli
Journal of Animal Science and Biotechnology (2024)
-
Peptidoglycan in osteoarthritis synovial tissue is associated with joint inflammation
Arthritis Research & Therapy (2024)
-
The individual response to antibiotics and diet — insights into gut microbial resilience and host metabolism
Nature Reviews Endocrinology (2024)
-
Bioaerosol Exposures and Respiratory Diseases in Cannabis Workers
Current Allergy and Asthma Reports (2024)
-
Effect of peptidoglycan on hepatopancreas of female Chinese mitten crabs (Eriocheir sinensis)
Aquaculture International (2024)