Abstract
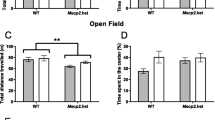
Stress during gestation increases vulnerability to disease and changes behavior in offspring. We previously reported that hypoxia and restraint during pregnancy sensitized the hypothalamic–pituitary–adrenal (HPA) axis and induced anxiety-like behavior in the adult offspring. Here, we report that gestational intermittent hypoxia (GIH) elicited a sex-dependent anxiety-like behavior in male P90 offspring and activation of corticotropin-releasing hormone (CRH) and CRH type-1 receptor (CRHR1) mRNA in the hypothalamic paraventricular nucleus (PVN) and in male E19 hypothalamus. These linked to demethylation at several specific sites of CpG island of Crhr1 promoter in P90 PVN and E19 embryo hypothalamus in GIH groups. Crhr1 DNA demethylation is more crucial in CpG island 1 than island 2 for activation of CRHR1 mRNA. DNMT3b is required for the Crhr1 DNA methylation than DNMT1 and DNMT3a in increased CRHR1 mRNA. We first address a novel hypothesis that GIH-induced male-sex-dependent demethylation at CpG sites of Crhr1 DNA in promoter triggers elevation of CRHR1 mRNA in PVN and anxiety-like behavior in adult offspring.







Similar content being viewed by others
References
Heim C, Nemeroff CB (2001) The role of childhood trauma in the neurobiology of mood and anxiety disorders: preclinical and clinical studies. Biol Psychiatry 49(12):1023–1039
de Kloet ER, Joels M, Holsboer F (2005) Stress and the brain: from adaptation to disease. Nat Rev Neurosci 6(6):463–475
Mueller BR, Bale TL (2008) Sex-specific programming of offspring emotionality after stress early in pregnancy. J Neurosci 28(36):9055–9065
Vale W, Spiess J, Rivier C, Rivier J (1981) Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 213(4514):1394–1397
Carrasco GA, Van de Kar LD (2003) Neuroendocrine pharmacology of stress. Eur J Pharmacol 463(1–3):235–272
Koob GF (1999) Corticotropin-releasing factor, norepinephrine, and stress. Biol Psychiatry 46(9):1167–1180
Dautzenberg FM, Hauger RL (2002) The CRF peptide family and their receptors: yet more partners discovered. Trends Pharmacol Sci 23(2):71–77
Bale TL, Vale WW (2004) CRF and CRF receptors: role in stress responsivity and other behaviors. Annu Rev Pharmacol Toxicol 44:525–557
Chang CP, Pearse RV 2nd, O'Connell S, Rosenfeld MG (1993) Identification of a seven transmembrane helix receptor for corticotropin-releasing factor and sauvagine in mammalian brain. Neuron 11(6):1187–1195
Chen R, Lewis KA, Perrin MH, Vale WW (1993) Expression cloning of a human corticotropin-releasing-factor receptor. Proc Natl Acad Sci U S A 90(19):8967–8971
Vita N, Laurent P, Lefort S, Chalon P, Lelias JM, Kaghad M, Le Fur G, Caput D, Ferrara P (1993) Primary structure and functional expression of mouse pituitary and human brain corticotrophin releasing factor receptors. FEBS Lett 335(1):1–5
Lovenberg TW, Liaw CW, Grigoriadis DE, Clevenger W, Chalmers DT, De Souza EB, Oltersdorf T (1995) Cloning and characterization of a functionally distinct corticotropin-releasing factor receptor subtype from rat brain. Proc Natl Acad Sci U S A 92(3):836–840
Perrin M, Donaldson C, Chen R, Blount A, Berggren T, Bilezikjian L, Sawchenko P, Vale W (1995) Identification of a second corticotropin-releasing factor receptor gene and characterization of a cDNA expressed in heart. Proc Natl Acad Sci U S A 92(7):2969–2973
Bale TL, Contarino A, Smith GW, Chan R, Gold LH, Sawchenko PE, Koob GF, Vale WW, Lee KF (2000) Mice deficient for corticotropin-releasing hormone receptor-2 display anxiety-like behaviour and are hypersensitive to stress. Nat Genet 24(4):410–414
Hauger RL, Risbrough V, Brauns O, Dautzenberg FM (2006) Corticotropin releasing factor (CRF) receptor signaling in the central nervous system: new molecular targets. CNS Neurol Disord Drug Targets 5(4):453–479
Kalaria RN, Spoors L, Laude EA, Emery CJ, Thwaites-Bee D, Fairlie J, Oakley AE, Barer DH, Barer GR (2004) Hypoxia of sleep apnoea: cardiopulmonary and cerebral changes after intermittent hypoxia in rats. Respir Physiol Neurobiol 140(1):53–62
Vannucci SJ, Hagberg H (2004) Hypoxia–ischemia in the immature brain. J Exp Biol 207(Pt 18):3149–3154
Golan H, Huleihel M (2006) The effect of prenatal hypoxia on brain development: short- and long-term consequences demonstrated in rodent models. Dev Sci 9(4):338–349
Patterson AJ, Chen M, Xue Q, Xiao D, Zhang L (2010) Chronic prenatal hypoxia induces epigenetic programming of PKC{epsilon} gene repression in rat hearts. Circ Res 107(3):365–373
Gozal D, Reeves SR, Row BW, Neville JJ, Guo SZ, Lipton AJ (2003) Respiratory effects of gestational intermittent hypoxia in the developing rat. Am J Respir Crit Care Med 167(11):1540–1547
Fan JM, Chen XQ, Jin H, Du JZ (2009) Gestational hypoxia alone or combined with restraint sensitizes the hypothalamic–pituitary–adrenal axis and induces anxiety-like behavior in adult male rat offspring. Neuroscience 159(4):1363–1373
Zohar I, Weinstock M (2011) Differential effect of prenatal stress on the expression of corticotrophin-releasing hormone and its receptors in the hypothalamus and amygdala in male and female rats. J Neuroendocrinol 23(4):320–328
Brunton PJ, Donadio MV, Russell JA (2011) Sex differences in prenatally programmed anxiety behaviour in rats: differential corticotropin-releasing hormone receptor mRNA expression in the amygdaloid complex. Stress 14(6):634–643
Day JJ, Sweatt JD (2010) DNA methylation and memory formation. Nat Neurosci 13(11):1319–1323
Elliott E, Ezra-Nevo G, Regev L, Neufeld-Cohen A, Chen A (2010) Resilience to social stress coincides with functional DNA methylation of the Crf gene in adult mice. Nat Neurosci 13(11):1351–1353
He XJ, Chen T, Zhu JK (2011) Regulation and function of DNA methylation in plants and animals. Cell Res 21(3):442–465
Breton CV, Byun HM, Wenten M, Pan F, Yang A, Gilliland FD (2009) Prenatal tobacco smoke exposure affects global and gene-specific DNA methylation. Am J Respir Crit Care Med 180(5):462–467
Paxinos G, Watson C, Pennisi M, Topple A (1985) Bregma, lambda and the interaural midpoint in stereotaxic surgery with rats of different sex, strain and weight. J Neurosci Methods 13(2):139–143
Li LC, Dahiya R (2002) MethPrimer: designing primers for methylation PCRs. Bioinformatics 18(11):1427–1431
Nesterenko TH, Aly H (2009) Fetal and neonatal programming: evidence and clinical implications. Am J Perinatol 26(3):191–198
Barnes SK, Ozanne SE (2011) Pathways linking the early environment to long-term health and lifespan. Prog Biophys Mol Biol 106(1):323–336
Li Y, Gonzalez P, Zhang L (2012) Fetal stress and programming of hypoxic/ischemic-sensitive phenotype in the neonatal brain: mechanisms and possible interventions. Prog Neurobiol 98(2):145–165
Zhang X, Li L, Xie W, Yang D, Heng X, Du Y, Doody RS, Le W (2013) Prenatal hypoxia may aggravate the cognitive impairment and Alzheimer's disease neuropathology in APP(Swe)/PS1(A246E) transgenic mice. Neurobiol Aging 34(3):663–678
Weinstock M (2007) Gender differences in the effects of prenatal stress on brain development and behaviour. Neurochem Res 32(10):1730–1740
Morgan CP, Bale TL (2011) Early prenatal stress epigenetically programs dysmasculinization in second-generation offspring via the paternal lineage. J Neurosci 31(33):11748–11755
Herman JP, Figueiredo H, Mueller NK, Ulrich-Lai Y, Ostrander MM, Choi DC, Cullinan WE (2003) Central mechanisms of stress integration: hierarchical circuitry controlling hypothalamo-pituitary-adrenocortical responsiveness. Front Neuroendocrinol 24(3):151–180
de Kloet ER (2008) About stress hormones and resilience to psychopathology. J Neuroendocrinol 20(6):885–892
Kokras N, Dalla C, Sideris AC, Dendi A, Mikail HG, Antoniou K, Papadopoulou-Daifoti Z (2012) Behavioral sexual dimorphism in models of anxiety and depression due to changes in HPA axis activity. Neuropharmacology 62(1):436–445
Orozco-Cabal L, Pollandt S, Liu J, Shinnick-Gallagher P, Gallagher JP (2006) Regulation of synaptic transmission by CRF receptors. Rev Neurosci 17(3):279–307
Bell KF, Claudio Cuello A (2006) Altered synaptic function in Alzheimer's disease. Eur J Pharmacol 545(1):11–21
Fischbach GD (2007) NRG1 and synaptic function in the CNS. Neuron 54(4):495–497
Gallagher JP, Orozco-Cabal LF, Liu J, Shinnick-Gallagher P (2008) Synaptic physiology of central CRH system. Eur J Pharmacol 583(2–3):215–225
Heinrichs SC, Lapsansky J, Lovenberg TW, De Souza EB, Chalmers DT (1997) Corticotropin-releasing factor CRF1, but not CRF2, receptors mediate anxiogenic-like behavior. Regul Pept 71(1):15–21
Arborelius L, Owens MJ, Plotsky PM, Nemeroff CB (1999) The role of corticotropin-releasing factor in depression and anxiety disorders. J Endocrinol 160(1):1–12
Chen XQ, Du JZ, Wang YS (2004) Regulation of hypoxia-induced release of corticotropin-releasing factor in the rat hypothalamus by norepinephrine. Regul Pept 119(3):221–228
Wu Y, Du JZ (2000) Effects of angiotensin II on release of CRH and AVP from hypothalamus during acute hypoxia. Acta Pharmacol Sin 21(11):1035–1038
He JJ, Chen XQ, Wang L, Xu JF, Du JZ (2008) Corticotropin-releasing hormone receptor 1 coexists with endothelin-1 and modulates its mRNA expression and release in rat paraventricular nucleus during hypoxia. Neuroscience 152(4):1006–1014
Reamon-Buettner SM, Borlak J (2007) A new paradigm in toxicology and teratology: altering gene activity in the absence of DNA sequence variation. Reprod Toxicol 24(1):20–30
Jirtle RL, Skinner MK (2007) Environmental epigenomics and disease susceptibility. Nat Rev Genet 8(4):253–262
Chalmers DT, Lovenberg TW, De Souza EB (1995) Localization of novel corticotropin-releasing factor receptor (CRF2) mRNA expression to specific subcortical nuclei in rat brain: comparison with CRF1 receptor mRNA expression. J Neurosci 15(10):6340–6350
Davis M (1992) The role of the amygdala in fear and anxiety. Annu Rev Neurosci 15:353–375
Schulkin J, Gold PW, McEwen BS (1998) Induction of corticotropin-releasing hormone gene expression by glucocorticoids: implication for understanding the states of fear and anxiety and allostatic load. Psychoneuroendocrinology 23(3):219–243
Brunson KL, Eghbal-Ahmadi M, Bender R, Chen Y, Baram TZ (2001) Long-term, progressive hippocampal cell loss and dysfunction induced by early-life administration of corticotropin-releasing hormone reproduce the effects of early-life stress. Proc Natl Acad Sci U S A 98(15):8856–8861
Gross C, Zhuang X, Stark K, Ramboz S, Oosting R, Kirby L, Santarelli L, Beck S, Hen R (2002) Serotonin1A receptor acts during development to establish normal anxiety-like behaviour in the adult. Nature 416(6879):396–400
Kolber BJ, Boyle MP, Wieczorek L, Kelley CL, Onwuzurike CC, Nettles SA, Vogt SK, Muglia LJ (2010) Transient early-life forebrain corticotropin-releasing hormone elevation causes long-lasting anxiogenic and despair-like changes in mice. J Neurosci 30(7):2571–2581
Brunton PJ, Russell JA (2010) Prenatal social stress in the rat programmes neuroendocrine and behavioural responses to stress in the adult offspring: sex-specific effects. J Neuroendocrinol 22(4):258–271
Khashan AS, Abel KM, McNamee R, Pedersen MG, Webb RT, Baker PN, Kenny LC, Mortensen PB (2008) Higher risk of offspring schizophrenia following antenatal maternal exposure to severe adverse life events. Arch Gen Psychiatry 65(2):146–152
Jones PA, Liang G (2009) Rethinking how DNA methylation patterns are maintained. Nat Rev Genet 10(11):805–811
Chen ZX, Riggs AD (2011) DNA methylation and demethylation in mammals. J Biol Chem 286(21):18347–18353
Acknowledgments
This work was supported by grants from the Ministry Science and Technology of China, National Basic Research Program “973” of China (No. 2012CB518200 and No. 2006CB504100), and the National Natural Science Foundation of China (No. 31171145 and No. 30770807). We would also like to thank Prof. Gareth Leng (Centre for Integrative Physiology, University of Edinburgh) and Prof. IC Bruce (Department of Physiology, School of Medicine, Zhejiang University, China) for editing and commenting on the manuscript.
Conflict of Interest
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, X., Meng, FS., Liu, ZY. et al. Gestational Hypoxia Induces Sex-Differential Methylation of Crhr1 Linked to Anxiety-like Behavior. Mol Neurobiol 48, 544–555 (2013). https://doi.org/10.1007/s12035-013-8444-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-013-8444-4